INTRODUCTION
Nonhuman primates (NHP) hold a unique position in biomedical research related to their close phylogenetic proximity to humans. Several NHP species are used in research every year, although they represent only a small proportion of the vertebrate animals used for such purposes. Rodents represent the vast majority of models used in biomedical research, and NHP account for around 0,2% to 0,5%1,2. The constant social concern about their use leads to an unremitting reduction of their use, according to the last agreements in Europe and the United States, with the revision of documents, normative and guidelines3. Due to ethical considerations, the use of NHP in research is constantly sparing, and results are invaluable given the reduction of their use together with their increased predictive value for the clinical scenario. Consequently, the stress levels and responsibilities carried over by NHP researchers in different fields have been significantly intensified throughout the last decades.
The careful planning of experiments involving NHP raises issues far beyond the experimental design and statistical power. Many other important aspects must be taken into consideration for reaching the most feasible species to answer the proposed questions guaranteeing the best possible translational potential for human clinical advances. Choosing the species for a given investigation demands a thorough analysis, still commonly devoid of careful concern during the experimental planning for several reasons. As large, intelligent, social, long-lived, and non-domesticated animals, NHP are the most challenging species in biomedical research, and issues such as convenience, practical and economic approaches should not be determinant for the choice of the species to be used in a given experiment design; however, characteristics such as phylogenetic position, telencephalization coefficients, morphological, functional, and neurobehavioral aspects should be considered to guarantee the use of the best possible model to answer the intended experimental questions.
Parkinson's disease (PD) is a research challenge in which a model with anatomic structures, pathways, cognitive abilities, and behaviors closer to humans is pivotal to develop novel therapeutic approaches and determine their clinical potential. Historically, the most successful studies in PD, which allowed the comprehension of functional connectivity within the basal ganglia-thalamocortical circuit and provided evidence that the subthalamic nucleus (STN) manipulations could relieve motor symptoms, were performed in monkeys4,5. Furthermore, the demonstration that high-frequency electric stimulation of STN led to significant improvement of PD symptoms in monkeys6, providing the fundamental underpinning of understanding which paved the way to develop successful surgical therapy for PD, giving rise to subsequent clinical trials7 and leading to the most advanced treatment option to PD available nowadays, the deep brain stimulation.
Research with NHP is crucial for discovering new treatments for human diseases, essential for medical progress, and will still be necessary for the foreseeable future1. Their use is highly relevant to the advancement of knowledge with considerable potential benefits to human health. When considering the appropriate model for answering an experimental question, several features will be determinant of to what extent the results can be extrapolated to humans, therefore choosing the most appropriate species is a relevant step for the experimental plan. This paper discusses the use of Sapajus apella monkeys as a model for the investigation of the pathophysiology and the development of novel therapeutic approaches for PD, based on their phylogenetic, neurological, immunological, genetic, and behavioral characteristics.
PARKINSON'S DISEASE AND RESEARCH IN NHP
Western society has been facing a huge challenge of brain diseases, burgeoning because of our aging population and PD figures as the second most prevalent neurodegenerative disorder8. PD is a life-threatening progressive disorder of the nervous system that affects the brain region responsible for initiating voluntary movements. The most common features of PD are tremor, rigidity, bradykinesia, postural instability, and balance and gait disorders, but it also shows a myriad of non-motor symptoms that can be even more disabling than the motor ones9. Clinical manifestations of the disease are a consequence of dopamine deficiency resulting from the degeneration of pigmented neurons in the mesencephalic substantia nigra, starting in most patients between the fifth and sixth decades of life. PD affects around 1-2 per 1,000 of the population at any time10, and the burden of the disease is very high for the individuals with PD, caregivers, and the health system, with the societal burden per capita around US$ 6,000 per year11. Despite an increase in the arsenal of available treatments and intensive research, the optimum management for this situation remains controversial. Nevertheless, the finest treatment options available for PD are the same for more than five decades, and there were no significant changes in the life expectancy of PD patients for the same period12. Furthermore, the pathophysiological and etiological mechanisms and possibilities for early diagnosis are still to be elucidated. Therefore, the development of novel and improved therapeutic options - with prospective to translate efficiently and as soon as possible to clinical practice - is critical to change this scenario.
PD as a complex multisystem disorder represents a research challenge of great proportions, for which most raised questions cannot be fully addressed using cellular, mathematical, or computational models, which precludes the appliance of the ethical principle of replacement. For such purposes, due to the complexity of the neural networks and symptoms to investigate, full organisms having both peripheral and central nervous systems are required. Rodents are the dominant mammalian animal species used in this scenario and have been proven valuable models on the elucidation of several molecular mechanisms, risk factors, and therapeutic approaches. They are inexpensive models, with a short lifespan, easy management, abundant genetic resources, and good reproductive performance in captivity, that together with anatomical, physiological, and genetic similarities to humans results on the impact of this model in translational science, however, limited and under permanent discussion13,14,15,16.
From a translational medicine standpoint, it is particularly critical to choose appropriate research models because a tremendous amount of money is spent testing drugs and therapies that fail at various stages of pre-clinical and clinical trials. Especially on investigations dedicated to understanding PD, some points on the experimental models become highly relevant for the discussion. As an illustration, although the neuroanatomical substrates underlying motor control are similar for humans and rodents, the behavioral repertoire mediated by those circuits is not. Furthermore, regardless of whether a PD model is based on toxins, lesions or genome modifications, to date, no single rodent model for PD was able to reproduce all key symptoms of the disease17.
There are critical differences in the organization of the motor system and behaviors among rodents, NHP, and humans18. Namely, major differences in the organization of the corticospinal system include the number of fibers and neural trajectories in motor tracts of primates. They correlate with dexterity and highly developed cortico-motoneuronal projections observed in all dexterous primates that use tools in the wild18,19. Such factors can be relevant when investigating a complex neurological condition that affects the motor system such as PD.
Furthermore, as PD shows a myriad of symptoms that includes relevant cognitive and psychiatric changes that may be worsened as adverse effects on pharmacological and surgical treatments20,21, the use of NHP in its research greatly enhances the range of behaviors and symptoms that can be studied, helping identify the crucial circuit elements and activity patterns involved in each cluster of symptoms, for which rodent models cannot provide sufficient data for translational needs22,23.
It is important to note that refinement does not always lead to the use of a lower species24. NHP can be trained for more elaborate tasks than rodents, and essays in NHP are recognized to attain improved predictive value due to their close similarity to humans in genomics1,25, neuroanatomic, neurophysiologic26,27,28, immunogenetic, and age-related dopaminergic changes22,29,30,31,32, representing the most relevant model for studies to determine the efficacy and safety of therapeutic approaches to PD.
Among the animal models that have proven effective in predicting the symptomatic efficacy of the antiparkinsonian drugs in current clinical use, the NHP arise as the highest predictive of both therapeutic and adverse effects subsequently observed in clinical trials and almost all known antiparkinsonian medication in current use had the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) primate as a predictive model of the effects in humans (see table 1 of Duty and Jenner23 for reference). Therefore, the parkinsonian NHP remains as a model through which drugs must almost inevitably be tested during the process of selection for clinical trial programs in PD23.
Table 1 - Comparison of some common marmosets (Callithrix jacchus) and capuchin monkeys (Sapajus apella) relevant characteristics as models for investigations in PD
| Callithrix jacchus | Sapajus apella | |
|---|---|---|
| General biologic characteristics | Small body size (~400 g) | Larger body size (~4 kg) |
| Rapid development (~12-15 months to sexual maturity) | Slower development (~5-7 years to sexual maturity) | |
| Old age reached early (~8 years) | * | |
| Short-lived (~5-7 years in captivity, with a maximum of 16-17 years) | Longer-lived (40-50 years in captivity) | |
| Twin births are common | Single births are common | |
| Shorter gestation (~4-5 months) | Longer gestation (6 months) | |
| Shorter inter-litter interval (~5-7 months) | Longer inter-litter interval (~21-24 months) | |
| Greater phylogenetic distance to humans | Smaller phylogenetic distance to humans | |
| Morphofunctional characteristics of the nervous system | Absolutely and relatively small unconvoluted brains (among the smallest relative to body size) | Absolutely and relatively larger convoluted brains |
| Higher-order parietal, temporal, and prefrontal areas | Higher-order parietal, temporal, and prefrontal areas | |
| Lack of monosynaptic projections of corticospinal neurons onto the motor neurons of the spinal cord ventral horn | Presence of monosynaptic projections of corticospinal neurons onto the motor neurons of the spinal cord ventral horn | |
| Motor and behavioral threats | No developed digital opposability and grasping abilities (grip with the claws) | Well-developed digital opposability and grasping abilities |
| * | Complex motor planning abilities | |
| No use of tools | Only new world monkey species that show the use of tools on the natural environment | |
| PD-related characteristics | Natural A53T mutation in α-syn | Exact human copy of 51-60 amino acid residues of α-syn |
| * | Pattern of development of basal ganglia structures similar to humans |
Sources: Kishi et al., 201448; Preuss, 201948; Kimberley et al., 201050; Vermilyea and Emborg, 201541; AnAge/The Animal Ageing and Longevity Database (genomics.senescence.info).
* Information not available.
Additionally, the commonalities between human and NHP brains are manifold, and similarities in the cellular composition, encephalization, and structural organization make the primate brain a scaled-down human brain33,34. Such similarities include safety-relevant characteristics of the primate immune systems and immunocompetent brain cells, which show relevant differences compared to rodents35,36 and can lead to important differences in the effects of several therapeutic approaches over neurons and glial cells. NHP have proven to be the best models for acquiring a closer prediction of human neural and immune responses not only on the pharmacological field but also on more invasive approaches such as implantable biomedical devices, neural grafts, and optogenetics that have shown a high potential for developing new therapeutic approaches for PD37,38,39.
Nevertheless, the NHP PD models also present their own drawbacks. Hitherto, there is no available model that fully recapitulates PD pathological and functional progression, therefore the recent emergence of new animal models based on α-synuclein (α-syn) changes has received growing attention40. Accumulation of α-syn and formation of Lewy bodies are proposed to have a significant role in PD pathology. Like humans, NHP show ubiquitous expression of α-syn in the central and peripheral nervous system. The protein sequence and localization are well conserved among mammalian species, with some Old World monkeys displaying an exact copy of the human α-syn and New World species showing few altered residues41. However, there are no reports of spontaneous PD in NHP and no post-mortem Lewy bodies identification in any NHP PD model22, even in marmosets that naturally carry the A53T α-syn gene mutation, which in humans is associated with aggressive early-onset PD40,41. Therefore, a better understanding of α-syn changes in NHP models of PD may help elucidate its role in PD pathology and progression and unravel the obscure genetic basis for major differences in brain aging between humans and NHP.
Several reports have outlined the scientific case for continued use of NHP in biomedical research pointing to neuroscience as an area where the evidence supports their use due to the high relevance of the amount and precision of data that can be collected, and the potential to promote the translation of basic science into clinical practice to improve neurorehabilitation42. Recital 17 of European Union Directive 2010/63 states that "the use of NHPs in scientific procedures is still necessary on biomedical research", and that "the use of NHPs should be permitted only in those biomedical areas essential for the benefit of human beings, for which no other alternative replacement methods are yet available"43. Therefore, NHP should be used only in those areas that are likely to be of ultimate benefit for humans. Neuroscientific research on PD is one example in which NHP can help us obtain insights into the mechanisms and neural basis of several symptoms, allowing us to investigate new therapeutic strategies and, besides that, provide an indispensable tool for basic research.
NHP SPECIES AND SCIENTIFIC INVESTIGATIONS IN PD
Testing potential therapies for PD requires an animal model that simulates the conditions of human disease as closely as possible. NHP have a highly developed nervous system with the same anatomic structures and pathways as humans, and, due to the close phylogenetic relationship, parkinsonian NHP models show similar symptoms, making them the best currently available animal models for PD in the preclinical context44.
Research using NHP models for studying PD has been developed in several different species including all four superfamilies of primates, and a great diversity of primate species is observed in neuroscience research. In many cases, however, the species of choice is based on practical reasons given that research with NHP has become increasingly difficult to sustain in many countries45. Regardless of that, the most important characteristics to be considered for choosing which NHP species to use in neuroscientific experiments should include the phylogenetic characteristics, telencephalization coefficients, neuroanatomic structures and pathways, cognitive abilities, and behavioral repertoires getting to the model with closer similarities to the human case under investigation, which is indispensable to realistically determine the clinical potential of the possible observed results.
However, only in very few studies we can find the rationale on the phylogenetic similarities or behavioral, neurophysiological or morphological features considered for the choice of the species. Practical aspects may influence the choices, such as animal lifespan and reproductive performance, with a reduction in the use of some species due to inability to keep pace of the production with the research demands in self-sustaining NHP research sources46. Also, accelerated efforts to reduce and replace the use of NHP with alternative research models led to severe restrictions to use the most close-related NHP species to humans in research such as the great apes. In consequence, Old World monkeys, which were the most commonly superfamily used in research, has been constantly replaced by New World primate species45.
Traditionally, the most used species in PD research is the common marmoset (Callithrix jacchus), a New World monkey native to east-central Brazil. Research using marmosets has grown exponentially in the last decade, due to several practical reasons, such as their small size, fast growth, sophisticated social life, and easy management in captivity. Marmosets mature and age more quickly than bigger monkeys, speeding up studies of diseases that affect development and aging. Also, they give birth twice a year aiding multigeneration genetic experiments. They have recently been genetically engineered to make their brains easier to image and to serve as models for several neurological disorders47. Though, there are important factors to be considered about their use that advocate in favor of the use of other bigger NHP species, especially concerning PD therapeutic approaches as a target for investigation. The marmoset characteristics that make them attractive as experimental subjects, such as their rapid development and short lifespan, are what differ them from humans, while bigger NHP species more closely resemble humans48.
In this scenario, capuchin monkeys (Sapajus apella) represent an interesting species for investigations in PD. They share several important characteristics of development and aging with humans, and neuroanatomic features with special interest for PD such as age-related changes in basal ganglia structures49 that together with cognitive abilities, motor, and behavioral repertoire make them important comparative models for understanding age-related changes and neurodegenerative conditions. Table 1 provides a comparative overview of many research-related relevant characteristics of capuchin monkeys and marmosets.
THE SAPAJUS APELLA AS A MODEL FOR STUDYING PD
Traditionally, capuchin monkeys have been lumped into the genus Cebus51 with four species, Cebus albifrons, Cebus olivaceus, Cebus capucinus, and Cebus apella, and these designations remained for decades. The genus has a large area of occurrence, from the north of Colombia to the south of Argentina, from the Coastal Plain Atlantic to the Andes mountains, distributing geographically throughout most of Brazil, especially in tropical forests spread both to north of the south of the Brazilian Legal Amazonia and in the Cerrado52. More recent shreds of evidence from morphological, phylogenetic, and biogeographic studies have shown that capuchin monkeys are diversified in two clades distinct enough to give rise to two genera: the gracile (untufted) capuchins, which represent the genus Cebus, and the robust (tufted) capuchins, representing the genus Sapajus53.
The Sapajus apella, also known as brown capuchin, is a New World primate from the genus Sapajus of the Cebidae family, living in South America, endemic to the Amazonian region, and represents one of the most widespread primates in the Neotropics54,55. It is a non-endangered species and shows good reproductive performance in captivity, being one of the species that most easily adapt to human contact. Because of its high reproductive potential and habitat flexibility, it was not devastated by hunting and habitat destruction as observed in other species of primates, being the most seen monkey in Amazonia56.
Sapajus apella monkeys have a long life expectancy, a high encephalization rate, and a great tendency toward exploitation and manipulation; they share food, follow an omnivorous diet, and have complex social behaviors such as cooperation and coalition formation57. They are also notably pro-social and exhibit social cognitive abilities, such as imitation, that are rare outside the apes. Also, they are the only New World monkey species that show the use of tools on the natural environment, and the variety of tool-tasks in which capuchins are successful together with their complex manipulative abilities are similar to those reported for apes58. Therefore, the motor, psychologic, and cognitive complexity of the capuchin monkey offers abundant sources for the analysis of different symptomatic and neurophysiological aspects on the course of PD and experimental therapeutic approaches closer to a human perspective. In an evolutionary context, capuchins are the most phylogenetically related to humans among New World primates, based on morphological, behavioral, and genetic characteristics59 (Figure 1).
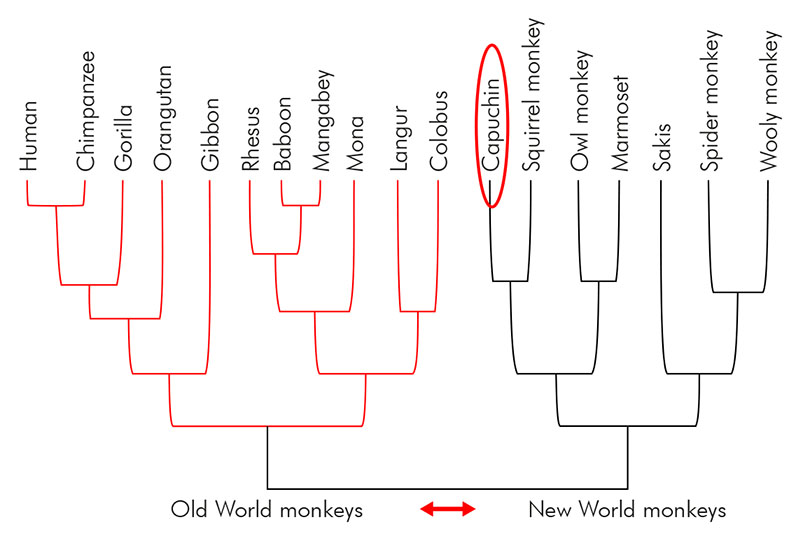
Source: Based on Baum et al., 200559; Chatterjee et al., 200960; and Surridge et al., 200361. Available at EvoEd phylogenetics (https://www.evo-ed.org/Pages/Primates/phylogenetics.html).
The position of capuchin monkeys is highlighted.
Figure 1 - Rooted phylogenetic representative tree of Old World and New World primate groups based on morphological, behavioral, and genetic characteristics
Although hitherto there are no reports of spontaneous PD in NHP, they present age-related dysfunctions of the dopaminergic nigrostriatal system, associated with motor impairments22, pointing to a vulnerability to changes that may be related to the development of neurodegenerative diseases during ageing62. The New World primates contain some of the short-lived as well as some of the long-lived monkey species, ranging from small species such as marmosets (quick development, relatively short-lived, and plentiful reproduction) to capuchin monkeys, which by contrast have the longest reported captive lifespan of any monkey, only slightly exceeded by the great apes63 (Table 1). Due to developmental and cytoarchitectonic patterns similar to humans in the basal ganglia structures49,64, the capuchin monkeys may be the most suitable options as models to provide a framework to study PD pathology and therapeutic approaches in an aged system.
The brain morphology in New World monkeys evolved with the occupation of several ecological niches and is strongly associated with the evolutionary increase of social behaviors65. The high encephalization rate of Sapajus apella also advocates their genetic proximity to humans, since human-specific genes are responsible for cortical expansion by the promotion of basal progenitor amplification, driving the formation of gyrencephalic cortex, differently from other NHP species such as marmosets that are quite lissencephalic66.
The diversification of brain morphology comprises one of the most prominent features of the primate adaptive radiation and therefore the evolutionary success, and particularly the convergent brain phenotypes may be related to ecological and behavioral factors that directly interfere with complex abilities such as social complexity65 and can be reflected also in other range of behaviors as well as an anatomical and functional organization.
It is still unclear if or how the genetic and morphofunctional differences in primate brains interfere on the presentation of pathologic and clinical features in PD. The 6-OHDA and MPTP-based models are the most used experimental NHP models of PD and replicate the human disease regarding neurotransmitter loss, neurochemical, morphological, and behavioral changes, but with lack of presentation of proteinaceous aggregates or typical Lewy bodies, which is one of the cardinal hallmarks of the pathophysiology of PD67. Moreover, even though all primates develop neurochemical and morphologic effects of MPTP and 6-OHDA toxins, the degree of susceptibility is variable among species68, and this may be related to changes in the α-syn41.
Nonetheless, the genetic basis for major differences in brain aging between humans and other primates is as obscure as is the basis for differences in life span. Aging monkeys and humans accumulate deposits of α-syn which is ubiquitously expressed in the nervous system of humans and NHP with a well-conserved protein sequence among species41. For example, gorillas, orangutans, and bonobos have exact copies of the human protein sequence, and New World NHP have few altered sites of α-syn protein sequence compared to humans, with the presence of threonine in the 53rd amino acid location (A53T) naturally occurring in marmosets, which in humans is a missense mutation associated with aggressive and early onset PD. The Sapajus apella monkey, in this aspect, shows the similar human sequence of the residues 51-60 of α-syn69. For the other side, as the marmosets, Sapajus apella monkeys present a total of four variations in the α-syn amino acid sequence compared to humans (Table 2). For marmosets, the alterations are A53T, S87N, Q99H, and N103S41. For Sapajus apella, the alterations are S87N, K96R, Q99H, and N103S69. The role of such alterations on PD pathology, if present, is still not clear.
Table 2 - α-syn amino acid sequence residues 51-60 in different primate species. The α-syn amino acid sequence is highly conserved in humans and NHP
| Species | 53 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Human WT | G | V | A | T | V | A | E | K | T | K |
| Human A53T | G | V | T | T | V | A | E | K | T | K |
| Sapajus apella | G | V | A | T | V | A | E | K | T | K |
| Callithrix jacchus | G | V | T | T | V | A | E | K | T | K |
| Natural 53A | PD A53T | Natural 53T | ||||||||
Source: Vermilyea and Emborg, 201541; and NCBI protein database.
Color represents amino acid variation (red) compared to human wild type (WT) sequence (yellow)
Brains have substantial ecological and adaptive importance because they underlie the behavior that allows an animal to successfully interact with their environment. In this sense, primates constitute a notable example, as the evolution of brain morphology is one of the most prominent features of their diversification70. Sapajus apella have relatively large brains and their brain development, as in humans, is widely influenced by the experiences in early childhood. The use of Sapajus apella monkeys may allow a more detailed analysis of PD symptomatology in both motor and non-motor aspects for several reasons and some of them are pointed out below.
MOTOR SKILLS
From an evolutionary point of view, exquisite manual dexterity is largely considered a prerogative of primates, as other mammalian orders do not exhibit such a high degree of manual dexterity. The Sapajus apella monkeys, as humans and Old World monkeys, show highly advanced manual dexterity, with a precision grip between the thumb and index finger due to the development of direct cortico-motoneuronal connections observed only in higher primates71. Since PD comprises several changes affecting global and fine motor coordination in different levels, the observance of the effects of experimental conditions over fine motor coordination skills can bring the analysis to a new level. Marmosets, despite their capacities of pre-shaping hand for grasping objects based on visuomanual coordination, do not present a precision grip, a critical ability for important advances involving fine motor behavior including tool use72. Sapajus apella monkeys show a highly developed precision grip, manual dexterity, and complex fine motor skills which, for PD motor studies, can allow a careful investigation beyond the rotational behaviors commonly analysed on rodent and marmoset models of PD, for which the most used motor tasks only evaluate the global motor coordination of large muscular groups73. It has been recently reported that Sapajus apella monkeys also present complex motor planning abilities74 and are able to modify a behavior to anticipate the future consequences of a given action, which may be related to the presence of multiple premotor areas in the frontal lobe75 as well as a proprioceptive cortical area 2 and a well-developed cortical area 5, which is associated with motor planning, visually guided reaching, grasping, and manipulation76.
BASAL GANGLIA AND BRAIN DEVELOPMENT
As observed in humans, the brain development of Sapajus apella is markedly influenced by early life events, with the presentation of brain growth spurt during early childhood77, and relatively big brain size after complete development. In the same direction, the capuchin monkey newborn, like all primates, is considered a relatively early mammal, and they show altricial behavior78. The high rate of brain development after birth leads to significant individual behavioral diversification as observed in humans79, and they show well-developed social intelligence compared to other platyrrhines primate species80.
Outputs of basal ganglia structures are not only involved in planning, initiation, and regulation of movement, but also in a variety of non-motor and cognitive functions such as working memory, visual perception, and attention switching. Basal ganglia are involved in both complex cognitive and motor acts, and imaging studies have revealed a similar pattern of development of basal ganglia structures similar to humans in capuchin monkeys, notably the Sapajus apella lineage, suggesting them as a suitable model for studying neurodevelopmental and age-related disorders49.
NON-MOTOR SKILLS
Unlike what is observed in simpler models such as rodents, primates exhibit intricate cognitive processes, complex social interactions, and a diverse behavioral repertoire, therefore they are more susceptible to behavioral, psychiatric, and psychological disorders81. Sapajus apella monkeys, as other primates, share with human core features of brain architecture and function, and complex social and cognitive behaviors typical of the primate order82. Many behavioral aspects of Sapajus apella point that this species can be particularly promising as a model for studying the neural circuits disturbances in PD and their behavioral and cognitive consequences. Sapajus apella presents social behavior and cognition features more similar to humans in several aspects, such as stable social relationships, dynamic social structure, and maintenance of affiliative relationships with cooperation in various ways83.
When it comes to studying cognitive processes and other complex behaviors, some procedures you can only perform with a primate model, for rodents do not present the complex psychological and behavioral features that are intertwined and mutually influenced by the disruption of dopaminergic systems that occur in PD. Sapajus apella cognitive task performance and personality evolved in behavioral and cognitive traits qualitatively similar to those of great apes, and individual differences in behavior and cognition often reflect personality differences82. Analyzing PD symptomatic profiles in such models can provide outcomes that might lead to new scientific and therapeutic approaches to PD. Recent studies are pointing to promising therapeutic approaches such as stem cell-based replacement therapies for which investigations using NHP have played an instrumental role in the process of preclinical optimization to guarantee safety and efficacy and facilitate the translation for humans27,39,84.
CONCLUSION
There are some complex and clinically relevant aspects of the symptomatology of Parkinson's disease that simply cannot be investigated with simple experimental models such as rodents in a relevant way to human patients. It is important to note that the appropriateness of each model and species for the development and assessment of treatments or basic science investigations depends on several factors, including the experimental aim of the study and whether the emphasis is placed on the analysis of behavioral, cognitive, and fine motor deficits, for which larger and more complex species are better to bridge the gap between rodent studies and clinical applications. Primates present ethical and practical challenges as animal models; however, NHP seem to be inevitable models for acquiring a closer prediction of human neural and immune responses due to their highly developed neural systems that base the cluster of symptoms observed in PD, together with safety-relevant differences between rodent and primate immune systems that can influence the outcomes of novel therapeutic strategies.
Sapajus apella monkey is a very promising NHP model for research on PD, for its complex behavioral and motor repertoire, and cognitive abilities, together with the neuroanatomical similarities of basal ganglia structural organization and age-related changes to humans, pointing that they comprise very suitable models for the study of new therapeutic approaches for PD. Their appropriate use as experimental models for PD might help elucidate several mechanisms of the disease and help to bridge the gap between rodent studies and clinical applications.














