Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2337-9622
Epidemiol. Serv. Saúde v.25 n.3 Brasília jul./set. 2016
http://dx.doi.org/10.5123/S1679-49742016000300008
ORIGINAL ARTICLE
Cross-sectional study on case fatality rate due to hantavirus infection in Goiás State, Brazil, 2007-2013*
1Universidade Federal de Goiás, Instituto de Ciências da Saúde, Jataí-GO, Brasil
2Universidade Federal de Goiás, Faculdade de Enfermagem, Goiânia-GO, Brasil
OBJECTIVE:
to describe the epidemiological and clinical profile of hantavirus infection and to analyze factors associated with case-fatality in Goiás, Brazil.
METHODS:
this was a cross-sectional study on case fatality due to hantavirus infection from 2007 to 2013 using data from the Notifiable Diseases Information System (SINAN) for Goiás State. Logistic regression analysis was performed to estimate odds ratios (OR) with 95% confidence intervals (95%CI).
RESULTS:
1,171 suspected cases were reported, of which 73 (6.2%) were confirmed. Among the confirmed cases, greater frequency was found among males (n=50), fever was the most common symptom (n=70), and there was a high proportion of hospitalization (n=68). The case-fatality rate was 57.5% . The following factors were associated with death: acute respiratory failure (ARF) (OR=3.6; 95%CI 1.2;10.6), hemoconcentration (OR=3.3; 95%CI 1.1;7.9) and not using a mechanical ventilator (OR=3.4; 95%CI 1.3;9.1). After adjustment, the ARF was still associated with death (OR=3.4; 95%CI 1.0;11.6).
Key words: Hantavirus Infections; Hantavirus; Case fatality; Cross-Sectional Studies.
Introduction
The hantavirus infection is an acute viral zoonosis caused by viruses of the family Bunyaviridae, genus Hantavirus, transmitted to humans by rodents and marsupials secretions. This disease is associated with two clinical forms: hantavirus hemorrhagic fever with renal syndrome (HFRS), known since 1930s and endemic in Europe and Asia; and hantavirus cardiopulmonary syndrome (HCPS), first described in the United States in 1993 and restricted to the Americas, which presents cardiogenic shock and cardiopulmonary involvement, followed by severe lung disease.1,2
The hantavirus was first described in Brazil in November 1993 in the municipality of Juquitiba, in São Paulo State, with the first case of cardiopulmonary syndrome. The infection has been endemic in the country since then. The first case reported in the Brazilian Savanna (Cerrado) was in 1996, and the first confirmed case in Goiás State occurred in 2000.3-7
In addition to Juquitiba and Araraquara subtypes, Anajatuba, Castelo de Sonhos, and Laguna Negra were identified in the country and associated with some species of rodents. Three other subtypes - Rio Mearim, Jaborá and Rio Mamoré - were considered as nonpathogenic, despite being found in some rodent species.8
Hantavirus case fatality rate is high and its severity has been studied according to the different virus subtypes.2 Other factors such as age, sex, clinical and laboratory results, mechanical ventilation and treatment characteristics may be involved in the prognosis. The different case fatality rates found in Brazil can be related to variations in the virulence of hantaviruses circulating subtype, as well as to the patients' different genetic characteristics and immunity. Furthermore, it is important to consider the epidemiological surveillance system and all medical support and assistance to cases identified in the different regions.2 Patients with sepsis, dyspnea, requiring mechanical ventilation and hemoconcentration present the worst prognosis.8,9
Although case fatality rates are high and its severity recognized, Brazilian studies relating the prevalence of hantavirus with its case fatality rate are still scarce. This study aims to describe the clinical and epidemiological profile of hantavirus infection and to analyze factors associated with its case fatality in Goiás State, Brazil.
Methods
This is a cross sectional study on hantavirus infection in Goiás State, from 2007 to 2013, using data from the Notifiable Diseases Information System (SINAN).
Goiás State is almost entirely covered by the Brazilian savanna, the second largest Brazilian biome. This is one of the most endangered biomes in the country, due to the agricultural expansion and the population growth that began in the 1970s.13
The selection of the period 2007-2013 was based on the need for obtaining standardized data for the longest time possible, given that the hantavirus notification forms have been modified with the implementation of the surveillance system in Brazil, which reflected in the database composition.14
All confirmed cases recorded in the notification system in Goiás in the period studied were included, according to the definition of case.14 First, the database was organized manually, checking for inconsistencies and repetitions. For the analysis, blank data or data recorded as ignored in the spreadsheet were excluded.
The following variables (dependent and independent) were used, as recorded in SINAN or as required in the study:
Epidemiological: notification year (from 2007 to 2013), State region (North, South, Central, Southeast, Southwest) and season (dry - October to March, or rainy - April to September).
Sociodemographic: sex (male and female); age, in years (considering the date of birth and the date of notification); education level (illiterate, incomplete elementary school I (1st to 4th grade), complete elementary school I (4th grade), incomplete elementary school II (5th to 8th grade); complete elementary school II; incomplete High School; complete High School; incomplete higher education; complete higher education; unknown); ethnicity/skin color (white, brown, black, indigenous, yellow, unknown); area of residence (urban or rural); type of environment where the infection probably occurred (household, work, leisure places, other, unknown); and occupation. The variable occupation was recorded as reported and encoded according to the Brazilian Classification of Occupations of the Brazilian Institute of Geography and Statistics (IBGE), using the code table to identify the individual's occupation for the confirmed cases.15
Clinical: clinical form (prodrome or unspecific, hantavirus cardiopulmonary syndrome); signs and symptoms (fever, headache, hypotension, chest pain, neurological symptoms, dry cough, generalized myalgia, shock, dizziness/vertigo, asthenia, dyspnea, low back pain - kidney area, nausea/vomiting, heart failure, petechiae, acute respiratory failure, abdominal pain, diarrhea, renal failure, other haemorrhage).
Treatment-related: laboratory tests (hematocrit >45%, thrombocytopenia, atypical lymphocytes, increased urea and creatinine, SGOT, SGPT, leukopenia, leukocytosis, IgM, immunohistochemistry); hospitalization (yes, no); and therapeutic support (mechanical respirator - invasive mechanical ventilation, vasoactive drugs, antivirals, antibiotics, corticosteroids, continuous positive airway pressure [CPAP] or bi-level positive airway pressure [BiPAP] - noninvasive mechanical ventilation).
Case Evolution: cure; death from another cause; death due to hantavirus infection; and unknown.
The case fatality rate was calculated by dividing the absolute number of deaths by the number of confirmed cases of the disease, multiplied by 100. Because this is a low-frequency disease, population-based mortality indicators were not used in order to prevent data dilution.
Statistical analysis was performed using the IBM(r) SPSS(r) Statistics, version 20.0. Qualitative variables were expressed in absolute numbers; and proportions and quantitative variables were expressed in average, median and standard deviation values. Pearson's chi-square test or Fisher's exact test, when appropriate were performed to evaluate associations between variables. We used logistic regression to estimate odds ratio [OR] and confidence intervals [CI] to investigate the association between clinical variables and variables related to the treatment with death outcome among confirmed cases. Variables that presented significance level <0.20 in the crude analysis were included in the adjusted analysis. The model worked with forced input to avoid suppressive effect between variables. In the final model, we adopted a significance level of 0.05, according to Wald chi-square test.
The project was approved by the Ethics Committee of the Federal University of Goiás on July 9, 2014 - Report No. 713,807/2014.
Results
In the period from 2007 to 2013, 1,171 suspected cases of hantavirus infection were reported in Goiás State, with an annual average of 167.3 notifications (standard deviation [SD]±95.7). Of the total of reported cases, 73 were confirmed (6.2%) with average of 10.4 (SD±5.1) cases per year. The year with the lowest number of confirmed cases was 2007, with six cases, whilst 2009 and 2011 had the highest numbers, both with 14 confirmed cases (Figure 1).
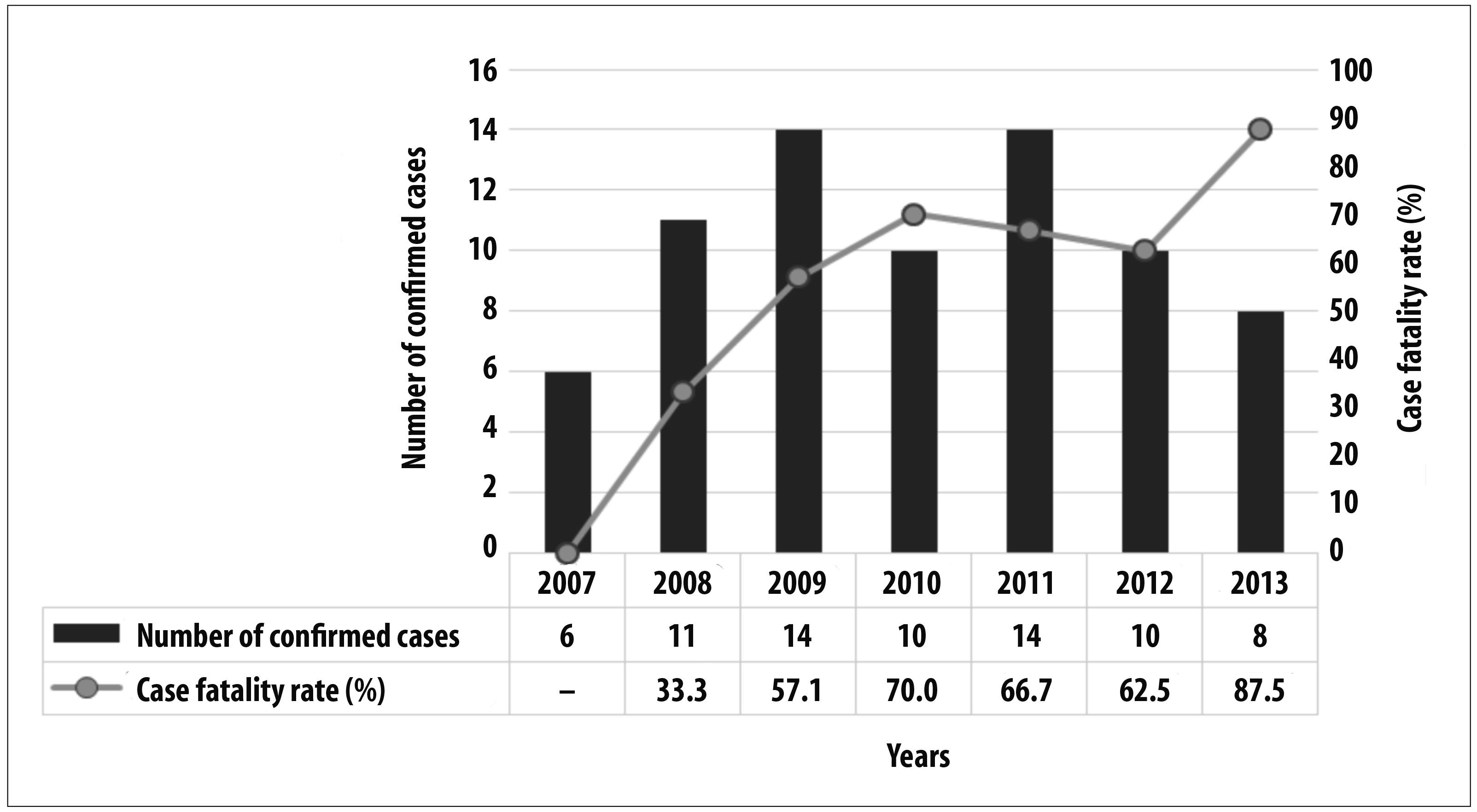
Figure 1 - Distribution of the number of confirmed cases of hantavirus infection (n=73) and case fatality rate per year, Goiás, 2007-2013
The average age of confirmed cases was 34.9 years (SD±13.8 years) considering both sexes; it was 39.0 years (SD±14.0) for women and 33.0 years (SD±13.4) for men (p>0.50). Over half of the individuals (62/73) were under 50 years old and were male (50/73) (Table 1). Considering education level, the highest frequency of hantavirus infection cases occurred in individuals with elementary or high school (19 and 15, respectively). The ethnicity/skin color brown was the most frequent (37/73), followed by white (26/73). About a third of the cases (24/73) reported contact with rodents and the others denied or were unable to inform; out of the total, 24 cases were characterized as work-related disease, but without statistically significant association (p>0.05). The main occupations identified were: rural worker or agriculturalist (n=4), construction worker (n=5), truck driver (n=3) and wholesalers (n=3) (Table 1).
Table 1 - Epidemiological profile of reported cases of confirmed hantavirus infection (N=73), according to the occurrence of death, Goiás, 2007-2013
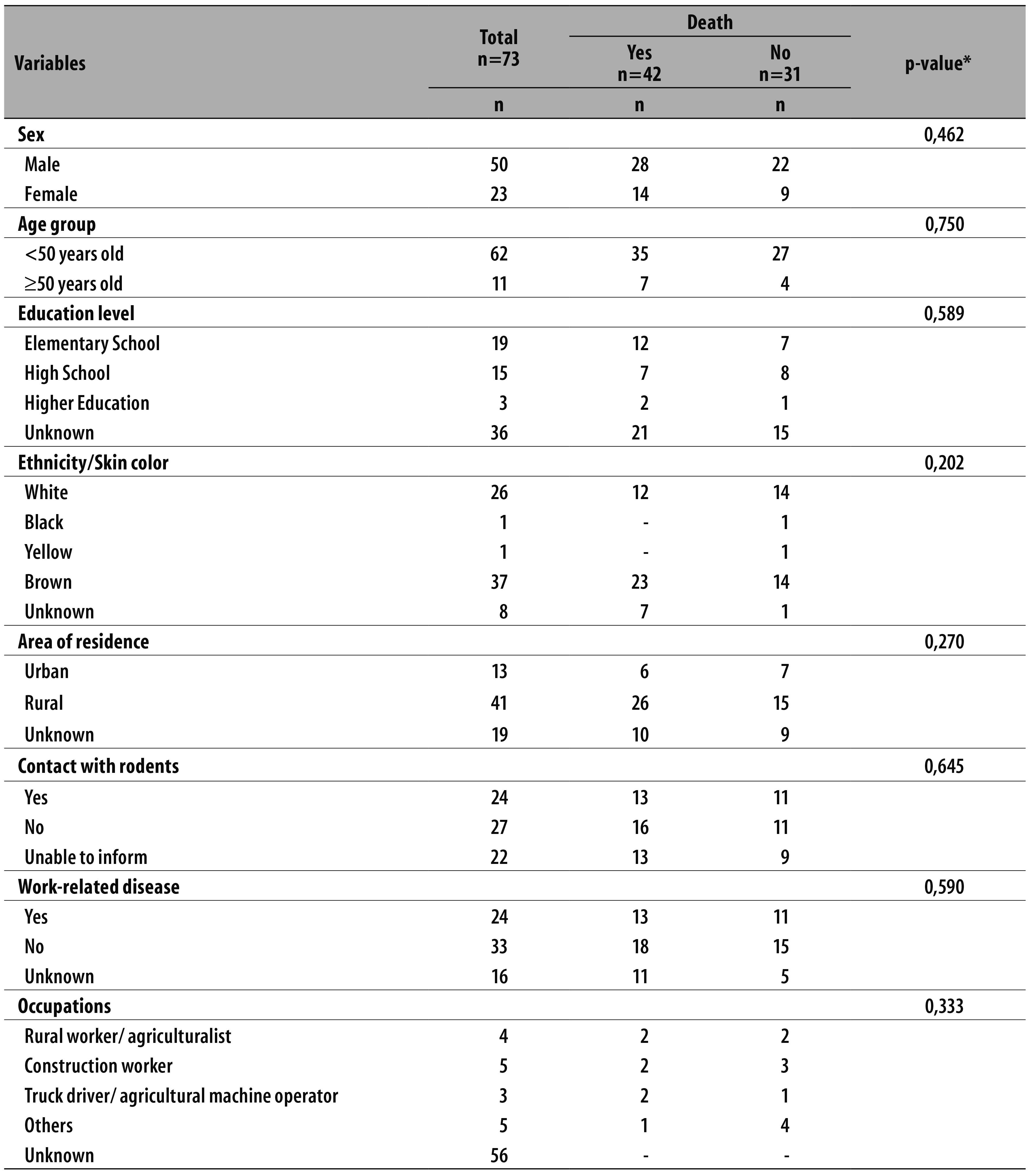
* Pearson's chi-square test.
Most of the notifications occurred in the Central and Southwest regions of the state, with the highest number in Goiânia (the capital city), where 795 suspected cases of hantavirus infection were reported and 32 (4.0%) cases were confirmed. The second municipality with the highest number of cases was Anápolis, which is near the capital, with 173 suspected cases and 14 (8.1%) confirmed (data not presented in the table).
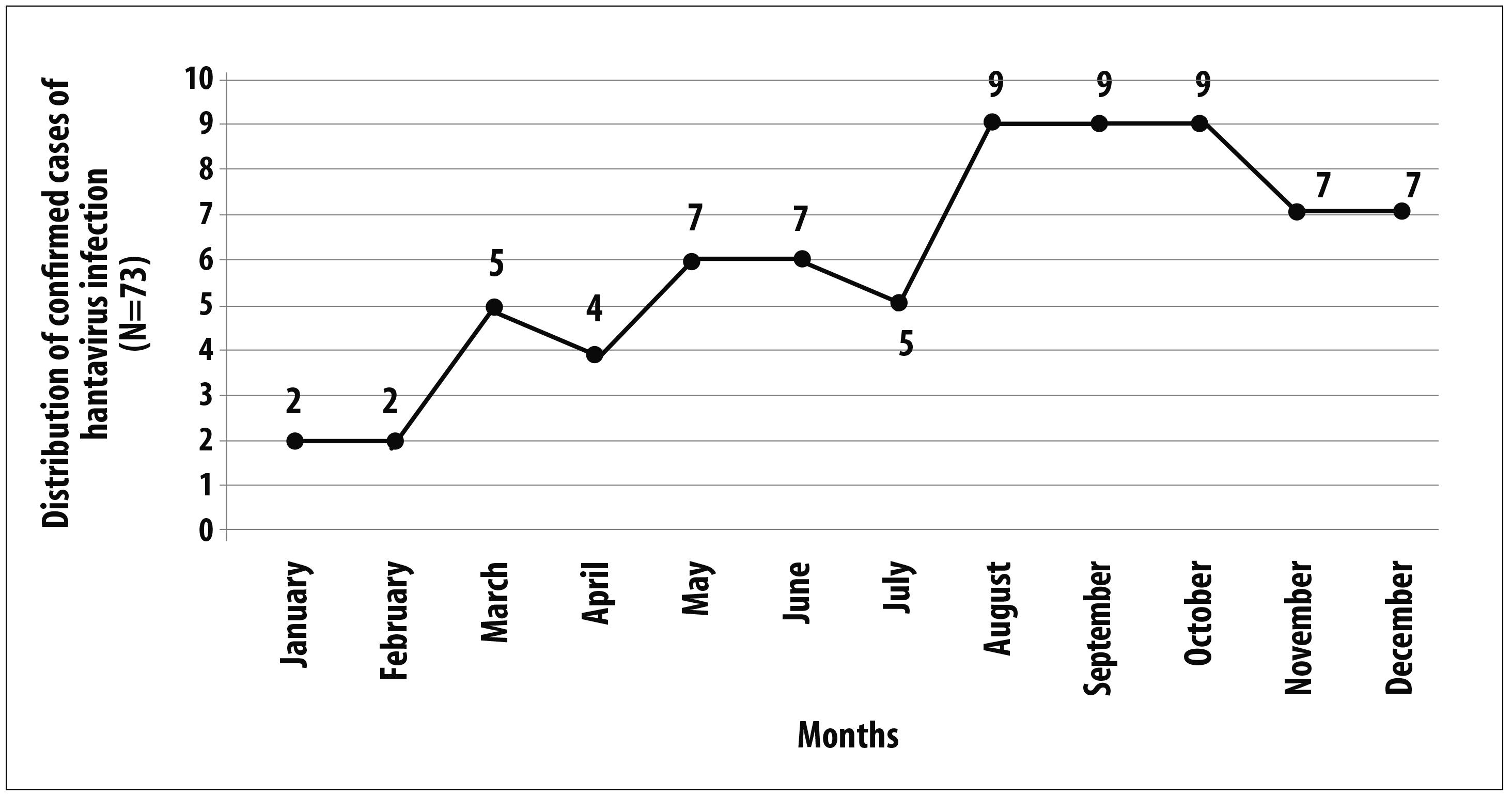
There was no association (p=0.406) between the frequency of confirmed cases of the disease, considering the notification date, and the climate seasonality of the region or the season in Cerrado (dry and rainy) (Figure 2).
With regard to signs and symptoms, fever was one of the most frequent, being present in 70 cases (70/73), followed by dyspnea (63/73) and dry cough (52/73). Besides those, the signs or symptoms that occurred in more than half the cases were IgM positive (n=57), respiratory failure (n=52), dry cough (n=52), myalgia (n=51), headache (n=45), nausea (n=41), chest pain (n=40) and diffuse pulmonary infiltrate (n=40). Renal failure was diagnosed in 51 cases and acute respiratory failure (ARF), in 53 cases. Among the confirmed cases, 32 presented hematocrit above 45% (Table 2).
Table 2 - Clinical profile of confirmed cases of hantavirus infection (N=73), according to the occurrence of death, Goiás, 2007-2013
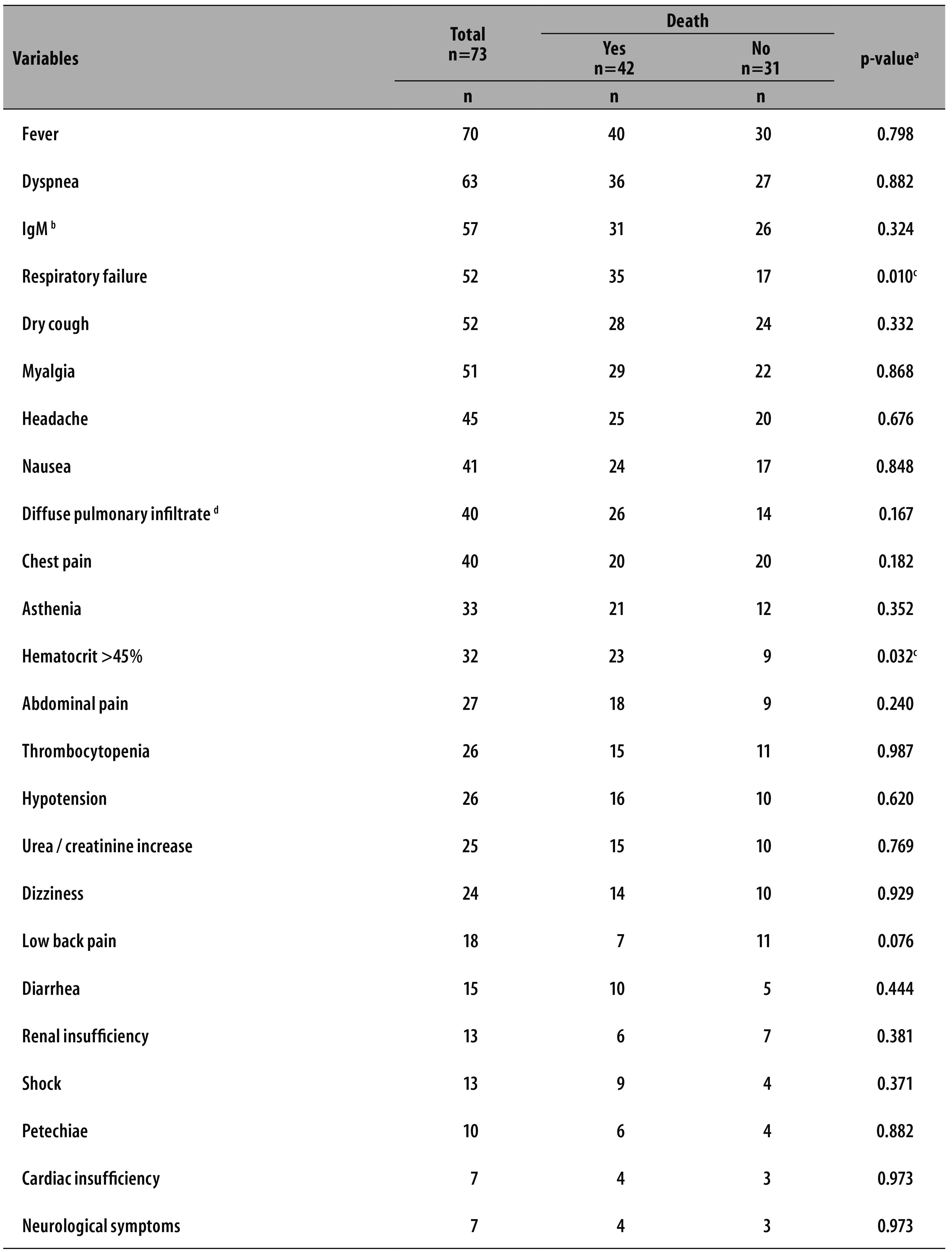
a) Wald chi-square test.
b) IgM: Immunoglobulin M
c) Significant value (p <0.05).
d) Radiography was performed in 55 patients.
Diffuse pulmonary infiltrate occurred in 40 cases among those who underwent chest radiography (n=55). Both immune histochemical analysis and results of the white series and liver transaminases were affected by underreporting, not being possible to identify the data.
Of the total of confirmed cases, 68 individuals were hospitalized, 60 of them diagnosed with the clinical form HCPS, and 36 used mechanical respirator. The average interval between hospitalization and death was 5.8 (SD±17.6) days. Nearly all the cases had diagnostic confirmation through laboratory tests (data not presented in the table). Among the hospitalizations, 39 individuals died (Table 3).
Table 3 - Crude and adjusted analysis of factors associated with death from hantavirus infection in Goiás, 2007-2013
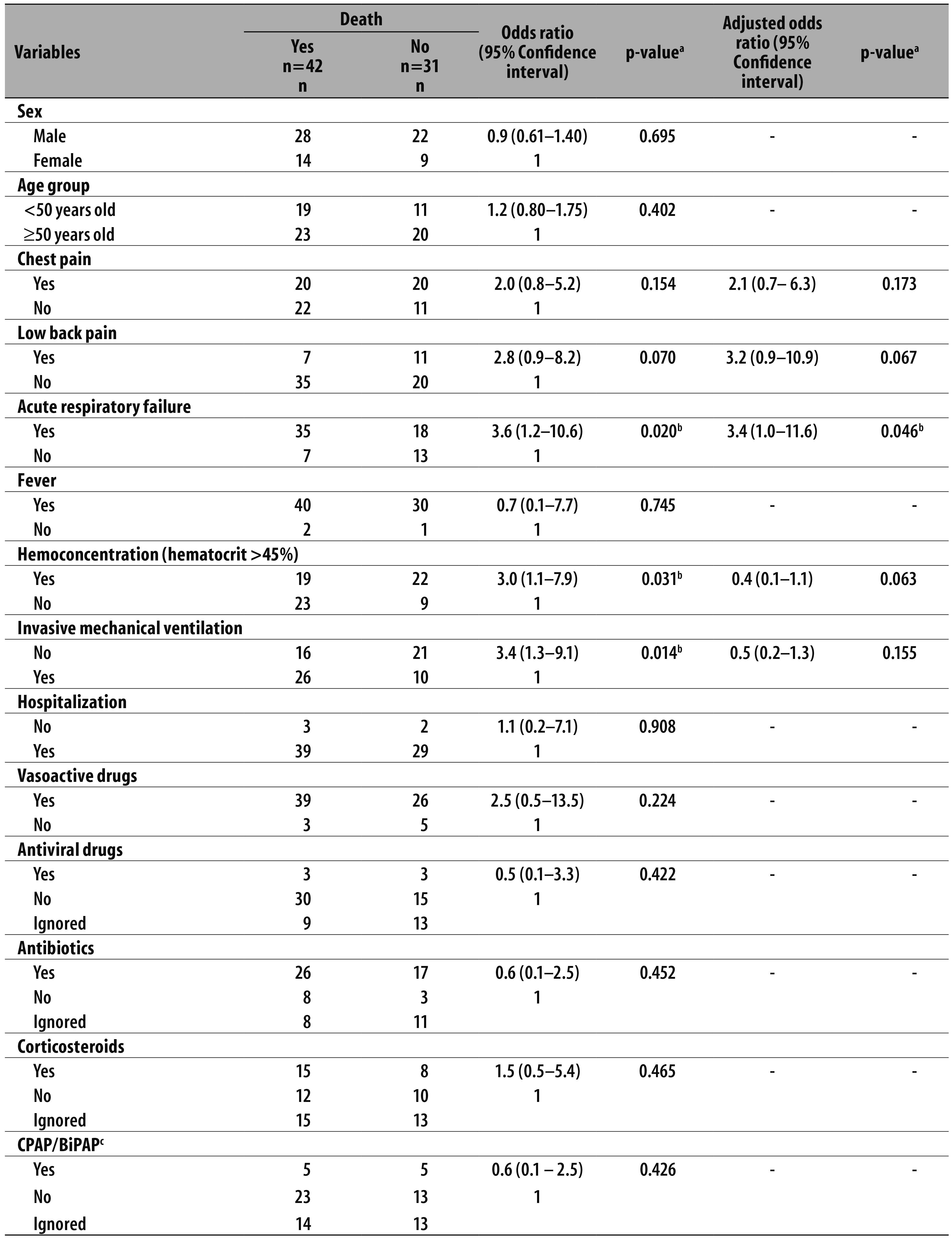
a) Wald chi-square test.
b) Significant P-value (p <0.05).
c) Continuous positive airway pressure [CPAP] ou bi-level positive airway pressure [BiPAP].
The case fatality rate in the analyzed period was 57.5% (42/73); 2013 was the year with the highest rate (87.5%). No deaths by hantavirus were recorded in Goiás in 2007 (Figure 1).
In all municipalities where there were cases reported and deaths, the case fatality rate represented more than 50.0% of the confirmed cases. In the capital, Goiânia, the mortality rate was of 56.3%, higher than in Anápolis, where it was 50.0%. In the municipality of Jataí, in the southwest of the state, which presented one of the highest frequencies of hantavirus, the case fatality rate was higher: 62.5%. Other municipalities, such as Campo Alegre de Goiás, Cidade Ocidental, Corumbá de Goiás, Mineiros, Rio Verde and Nerópolis, even with one case each, had high case fatality rate (100.0%); in Aparecida de Goiânia, two cases were confirmed, and the case fatality rate was also 100% (data not presented in the table).
The case fatality rate was 56.3% in the Central region and 62.5% in the Southwest region, being similar among women (60.9%; 14/23) and men (56.0%; 28/50), and there was no association observed between different age groups (data not presented in the table).
The associated variables in the crude analysis (p<0.05) with death caused by hantavirus were: respiratory failure (OR=3.6; 95%CI 1.2-10.6), not using a mechanical respirator (OR=3.4; 95%CI 1.3-9.1) and hematocrit above 45% (OR=3.0; 95%CI 1.1;7.9) (Table 3).
In the logistic regression analysis model, acute respiratory failure, mechanical respirator use, low back pain, chest pain, and hemoconcentration were included. After adjustment, only the presence of acute respiratory failure remained associated with deaths due to hantavirus, with risk of death among patients with ARF increased by more than three times (OR=3.4; 95%CI 1.0;11.6) (Table 3).
Regarding therapeutic support, 65 patients received vasoactive drugs, 43 received antibiotics and 23 corticosteroids, and in 6 cases antiviral drugs were administered. As for mechanical ventilation, in addition to those 36 patients undergoing invasive mechanical ventilation (mechanical ventilator), other 10 patients underwent non-invasive ventilation methods such as CPAP and BiPAP (Table 3).
Discussion
About 4 thousand cases of hantavirus infection have been recorded in the Americas since the first case was reported, in 1993. In the Brazilian Midwest region the subtypes identified were Araraquara and Juquitiba.16,17 This study showed that, among the confirmed cases of hantavirus infection in Goiás State, young men, with low education levels were the most affected. The case fatality rate was high and the presence of respiratory failure was associated with mortality. The frequency of notification on hantavirus infection in the period studied (2007-2013) increased over the years, however, the proportion of confirmed cases of the disease did not raise. For each confirmed case, 16 suspected cases were reported.
A study conducted in Oklahoma, United States, in 2005, tried to correlate the number of HCPS notifications and deaths whose cause was acute respiratory failure, and concluded that there might be an underreporting of cases by misdiagnosis.18
In Brazil, an epidemiological study using the same database as here, SINAN, and retrospectively assessing 2001-2011 data nationwide, found 1,486 reported cases, with increase trend.16
We can imply that the data notification found in this study reflect the effort of epidemiological surveillance in disease control with evident attention showed by professionals when notifying the cases. On the other hand, the differential diagnosis between hantavirus infection and other infectious diseases that are also present in the region, such as leptospirosis, influenza, dengue, yellow fever, malaria, pneumonia, and non-infectious diseases, also common in the Goiás, such as chronic obstructive pulmonary disease, systemic lupus erythematous and rheumatoid arthritis is clearly difficult.3,19,20
The number of confirmed cases showed no proportional growth in relation to reported cases, i.e., more reports did not imply in more confirmations.
The total number of confirmed cases was concentrated in the South-Central region of the state, especially in Anápolis and Jataí as the cities with the highest frequency of the disease, after the capital, Goiânia. This region of the state is characterized by intensive agricultural and livestock production, as well as a recent agro-industrial development in grain production.13
In Cerrado, this disease is present since 1996, i.e., three years after the first national case was identified in the southeastern region of the country. Considering this biome in a 16-year analysis (1996-2008), the average frequency of cases of hantavirus infection was 26.6 cases per year, which was higher than the number reported in this study.19
Most of cases in Goiás occurred in men. Some studies reported a higher incidence among young men, associating the fact with the interaction between occupation and sex.10 The profile of cases was similar to what was found in other studies in Brazil and in the Americas, with predominance of young men in the productive phase. Other studies also report increased frequency of HCPS cases among young individuals under 50 years old. A study conducted in Brazil, from 1993 to 2006, identified 855 reported cases - most of them occurred in young men, workers in the rural area.10 In Argentina, in the period from 1995 to 2008, the results were similar, showing higher frequency of this disease among young adult men, workers, indicating also more severe cases among women.20,21
No association was found with the occupation or that could relate the disease to labor activities. It is important to highlight that the level of education identified was mainly Elementary School, which features, besides the young population, people with low education level and with occupations related to activities in the countryside or even in the city, but that do not require higher qualification, as construction workers and drivers. These occupations favor the exposure to rodents and increase the possibility of hantavirus infection.7,21-24
No difference in the frequency of hantavirus infection according to the ethnicity/skin color was observed. Most studies link the presence of hantavirus with social, economic and environmental issues, and not solely to ethnicity/skin color. A study conducted in the United States identified the presence of pulmonary hantavirus, with distribution of 63% in Caucasians, 35% in Native Americans and 2% in American Afro-descendants, however, with no association with the incidence of the disease.12,25,26
Contact with rodents or risk exposure, like sleeping in tents, house cleaning, grinding, freight transport, deforestation and rural tourism were also reported. Such situations are proven to be associated with the increased likelihood of infection by hantavirus and are not only related to the countryside, but also have been shown in the outskirts of cities in low and middle income countries, where the uncontrolled growth of the population and the low sanitation coverage promote the proliferation of rodents which invade households.15,19,21
The distribution of cases did not demonstrate seasonality of the infection, since the number of cases did not change expressively in the two main climatic periods of the region - dry and rainy. This result was different from other studies, which found greater concentration of cases in the dry season, with the largest population of rodents in Cerrado; or even, in the Atlantic Rainforest, where the largest record frequency was registered between the months of September and December.27,28
The number of suspected cases has increased each year in Goiás, but the number of confirmed cases has not increased as well. On the other hand, the case fatality rate has increased. The case fatality rate was higher than the one found in a Brazilian study on Cerrado and was also higher than the hantavirus fatality rate in Brazil last year.9,19
The surveillance system should allow early case identification, still in the initial phase (prodrome), as well as efficient intervention and treatment.16 The case fatality rate in this study was higher among those diagnosed in the severe clinical form of HCPS, and almost all patients required hospital care.
The case fatality of hantavirus in the Brazilian Midwest and in Cerrado has been higher than in the other Brazilian regions and biomes, such as the Atlantic Rainforest.19 The high case fatality rate can be associated to the variants of hantavirus that present different virulence.18,28 This was one of the limitations of this study, since the identification of the different genotypes of the etiologic agent was not performed.
The hantavirus infection in Goiás has been diagnosed in its advanced stage (HCPS), when the respiratory impairment is very common. This fact, associated with the high virulence of hantavirus, might explain the high fatality rates found. A study conducted in Brazil, from 1993 to 2007,17 pointed that the two main strains present in the Brazilian Southeast, South and Midwest were Araraquara and Juquitiba, and that the fatal HCPS were more frequent with Araraquara virus.12,14,19
In addition to hantavirus virulence and its late diagnosis, it is important to relate the health care conditions in many municipalities, especially in those that are smaller and far from the major centers and that do not have facilities for intensive treatment of the most severe cases of the disease with respiratory compromise. These associated conditions may be influencing the high fatality rates. Almost all confirmed cases were hospitalized, and most of them evolved to death.
The main clinical manifestations of hantavirus infection in the Americas are related to respiratory compromise such as cough and dyspnea.1 In this study, the ARF is associated with fatal cases. Those individuals who had access to treatment and were also assisted in therapeutic support by mechanical respirator had almost 30% more chances to survive. Meanwhile, hematocrit (>45%) was associated with death caused by hantavirus.
In multiple regression analysis, respiratory failure remained associated with the risk of death independent of other variables, and those with ARF were almost four times more likely to progress to death. A study conducted in Brazil, considering data from 1993 to 2006 identified hemoconcentration as an independent variable associated with high risk of death, and suggests that this variable can be used as a marker to identify severe cases of HCPS earlier, enabling better therapeutic support.1,9-11
The limitations of this study are related to some weaknesses of the database provided by SINAN, which still presents inconsistencies in filling the clinical records, limiting data analysis, such as onset of symptoms, date of notification/investigation, treatment and examinations conducted. Moreover, the possibility of underreporting of cases due to the difficulty in the differential diagnosis of this disease should also be considered as a limitation.
Another limitation was the absence of genotyping information of the hantaviruses involved in confirmed cases, which could be the subject of future studies, allowing the knowledge of the most common strain of the virus in Goiás State.
The points to be considered are the expansion of knowledge of an emerging disease in a region where hantavirus has received little attention from health authorities, and also the identification of weaknesses in the epidemiological surveillance system, demonstrated by the growing disproportion between the number of suspected cases reported and the number of confirmed cases and diagnosis in late clinical form of the disease.
The results of the study enabled to reveal the epidemiological and clinical profile of the hantavirus infection in Goiás, which can alert the health authorities to prioritize surveillance actions, particularly in regions of the state where the disease's frequency has been higher and high mortality rates are found. This information can also be useful in the formulation of clinical reasoning, so that health professionals are able to timely think this diagnostic hypothesis, which can help patients' prognosis.
REFERENCES
1. Figueiredo LT, Campos GM, Rodrigues FB. Hantavirus pulmonary and cardiovascular syndrome: epidemiology, clinical presentation, laboratory diagnosis and treatment. Rev Soc Bras Med Trop. 2001 Jan-Feb;34(1):13-23. [ Links ]
2. Teixeira BR, Loureiro N, Strecht L, Gentile R, Oliveira RC, Guterres A, et al. Population ecology of hantavirus rodent hosts in Southern Brazil. Am J Trop Med Hyg. 2014 Aug;91(2):249-57. [ Links ]
3. Badra SJ, Maia FGM, Figueiredo GG, Santos Júnior GS, Campos GM, Figueiredo LTM, et al. A retrospective serologic survey of hantavirus infections in the county of Cassia dos Coqueiros, State of Sao Paulo, Brazil. Rev Soc Bras Med Trop. 2012 jul-ago;45(4):468-70. [ Links ]
4. Secretaría Regional Ministerial de Salud de la Región del Bobío (Chile). Informações de saúde [Internet]. Concepción: SEREMI de la Región del Bobío; 2015. [citado 2016 mar 29]. Disponível em: Disponível em: http://www.seremidesaludbiobio.cl/sd/web/ . [ Links ]
5. Oliveira SV, Escobar LE, Peterson AT, Gurgel-Gonçalves R. Potential geographic distribution of hantavirus reservoirs in Brazil. PLoS One. 2013 Dec;8(12):e85137. [ Links ]
6. Oliveira RC, Guterres A, Fernandes J, D'Andrea PS, Bonvicino CR, Lemos ERS. Hantavirus reservoirs: current status with an emphasis on data from Brazil. Viruses. 2014 May;6(5):1929-73. [ Links ]
7. Guterres A, Oliveira RC, Fernandes J, D'Andrea PS, Bonvicino CR, Bragagnolo C, et al. Phylogenetic analysis of the S segment from Juquitiba hantavirus: identification of two distinct lineages in Oligoryzomys nigripes. Infect Genet Evol. 2013 Aug;(18):262-8. [ Links ]
8. Oliveira RC, Sant'ana MM, Guterres A, Fernandes J, Hillesheim NL, Lucini C, et al. Hantavirus pulmonary syndrome in a highly endemic area of Brazil. Epidemiol Infect. 2016 Apr;144(5):1096-106. [ Links ]
9. Elkhoury MR, Mendes WS, Waldman EA, Dias JP, Carmo EH, Vasconcelos PFC. Hantavirus pulmonary syndrome: prognostic factors for death in reported cases in Brazil. Trans R Soc Trop Med Hyg. 2012 May;106(5):298-302. [ Links ]
10. Kaya S. Prognostic factors in hantavirus infections. Mikrobiyol Bul. 2014 Jan;48(1): 179-87. [ Links ]
11. Campos GM, Borges AA, Badra SJ, Figueiredo GG, Souza RL, Moreli ML, et al. Pulmonary and cardiovascular syndrome due to hantavirus: clinical aspects of an emerging disease in southeastern Brazil. Rev Soc Bras Med Trop. 2009 May-Jun;42(3): 282-9. [ Links ]
12. Limongi JE, Costa FC, Paula MB, Pinto RM, Oliveira ML, Pajuaba Neto AA, et al. Hantavirus cardiopulmonary syndrome in the Triangulo Mineiro and Alto Paranaiba regions, State of Minas Gerais, 1998-2005: clinical-epidemiological aspects of 23 cases. Rev Soc Bras Med Trop. 2007 May-Jun;40(3):295-9. [ Links ]
13. Silva GB, Mendes EPP, Santos DP. As transformações do cerrado goiano: o processo histórico, da mineração aos dias atuais. VI Congresso Iberoamericano de Estudios Territoriales y Ambientales; 2014; São Paulo. São Paulo: Universidade de São Paulo; 2014:2026-38. [ Links ]
14. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de vigilância em saúde [Internet]. Brasília: Ministério da Saúde; 2014 [citado 2016 Jul 8]. Disponível em: Disponível em: http://portalsaude.saude.gov.br/images/pdf/2014/novembro/27/guia-vigilancia-saude-linkado-27-11-14.pdf [ Links ]
15. Instituto Brasileiro de Geografia e Estatística. Comissão Nacional de Classificação. Classificação Brasileira de Ocupações. Classificações Brasileira de Ocupações [Internet]. Rio de Janeiro: Instituto Brasileiro de Geografia e Estatística; 2014 [citado 2016 maio 7]. Disponível em Disponível em http://concla.ibge.gov.br/estrutura/ocupacao-estrutura.html [ Links ]
16. Pinto Junior VL, Sousa AI, Lemos ERS. Regional variations and time trends of hantavirus pulmonary syndrome in Brazil. Epidemiol Infect. 2014 Oct;142(10):2166-71. [ Links ]
17. Figueiredo LTM, Moreli ML, Sousa RLM, Borges AA, Figueiredo GG, Machado AM, et al. Hantavirus pulmonary syndrome, central plateau, southeastern, and southern Brazil. Emerg Infect Dis. 2009 Apr;15(4):561-7. [ Links ]
18. Smithee L, Bos J, Mallonee S, Nisbett RA, Crutcher JM. Update on hantavirus in Oklahoma: are we missing cases? J Okla State Med Assoc. 2007 May;100(5):145-8. [ Links ]
19. Nunes ML, Maia-Elkhoury ANS, Peliscari DM, Elkhoury MR. Caracterização clínica e epidemiológica dos casos confirmados de hantavirose com local provável de infecção no bioma Cerrado Brasileiro, 1996 a 2008. Epidemiol Serv Saude. 2011 out-dez;20(4):537-45. [ Links ]
20. Suzuki K, Mutinelli LE. Knowledge and practices about hantavirus pulmonary syndrome in a cluster of Japanese communities in Argentina. Rev Panam Salud Publica. 2009 Feb;25(2):128-33. [ Links ]
21. Jonsson CB, Figueiredo LT, Vapalahti O. A global perspective on hantavirus ecology, epidemiology, and disease. Clin Microbiol Rev. 2010 Apr;23(2):412-41. [ Links ]
22. Travassos da Rosa ES, Sampaio de Lemos ER, Medeiros DBA, Smith DB, Pereira AS, Elkhoury MR et al. Hantaviruses and hantavirus pulmonary syndrome, Maranhão, Brazil. Emerg Infect Dis. 2010 Dec;16(12):1952-5. [ Links ]
23. Watson DC, Sargianou M, Papa A, Chra P, Starakis I, Panos G. Epidemiology of Hantavirus infections in humans: a comprehensive, global overview. Crit Rev Microbiol. 2014 Aug;40(3):261-72. [ Links ]
24. Simpson SQ, Spikes L, Patel S, Faruqi I. Hantavirus pulmonary syndrome. Infect Dis Clin North Am. 2010 Mar;24(1):159-73. [ Links ]
25. Donalisio MR, Vasconcelos CH, Pereira LE, Avila AMH, Katz G. Climatic aspects in hantavirus transmission areas in Sao Paulo State, Brazil. Cad Saude Publica. 2008 May;24(5):1141-50. [ Links ]
26. Katz G, Williams RJ, Burt MS, Souza LTM, Pereira LE, Mills JN, et al. Hantavirus pulmonary syndrome in the State of Sao Paulo, Brazil, 1993-1998. Vector Borne Zoonotic Dis. 2001 Sep;1(3):181-90. [ Links ]
27. Martinez VP, Bellomo CM, Cacace ML, Suarez P, Bogni L, Padula PJ. Hantavirus pulmonary syndrome in Argentina, 1995-2008. Emerg Infect Dis. 2010 Dec;16(12): 1853-60. [ Links ]
28. Casapia M, Mamani E, Garcia MP, Miraval ML, Valencia P, Quino AH, et al. Hantavirus pulmonary syndrome (Rio Mamore virus) in the Peruvian Amazon region. Rev Peru Med Exp Salud Publica. 2012 Jul-Sep;29(3):390-5. [ Links ]
*Article based on Hélio Ranes de Menezes Filho's master's thesis, entitled Epidemiologic-clinic profile of hantavirus infection in the Goiás State between 2007-2013, presented to the Graduate Program in Applied Health Science (PPGCAS) of the Federal University of Goiás, in Jataí campus, defended on 2015.
Received: February 21, 2016; Accepted: April 03, 2016











 texto em
texto em 
 Curriculum ScienTI
Curriculum ScienTI