Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2337-9622
Epidemiol. Serv. Saúde v.25 n.3 Brasília jul./set. 2016
http://dx.doi.org/10.5123/S1679-49742016000300009
ORIGINAL ARTICLE
Yellow fever outbreaks in São Paulo State, Brazil, 2000-2010*
1Faculdade de Ciências Médicas da Santa Casa de São Paulo, Mestrado Profissional em Saúde Coletiva, São Paulo-SP, Brasil
2Faculdade de Ciências Médicas da Santa Casa de São Paulo, Departamento de Medicina Social, São Paulo-SP, Brasil
OBJECTIVE:
to describe yellow fever occurrence in the state of São Paulo, Brazil, in the period 2000-2010.
METHODS:
this was a descriptive study using data regarding human cases, epizootics in non-human primates and virus vector isolation in São Paulo State, between 2000 and 2010.
RESULTS:
three sylvatic yellow fever outbreaks were registered in the state with 32 human cases and 15 deaths; the cases occurred in unvaccinated individuals exposed to the sylvatic form of transmission; epizootics was confirmed before the virus began circulating in humans in the region of São José do Rio Preto and the virus was isolated twice in vectors.
CONCLUSION:
three sylvatic yellow fever outbreaks occurred in the state between 2000 and 2010, two of them in a transition area and the other in an area considered to be unaffected; vaccination and maintaining immunization coverage are necessary for preventing the disease.
Key words: Yellow Fever; Zoonosis; Epidemiological Surveillance
Introduction
Yellow fever is an acute infectious, febrile, hemorrhagic and non contagious disease, which has been responsible for decimating South American and African populations since the seventeenth century.1,2 Two different epidemiological patterns of transmission are described: a sylvatic pattern and an urban cycle of transmission by the Aedes aegypti.2
Although previously considered as one of the safest vaccine containing the live attenuated virus, the yellow fever vaccine can induce adverse events after immunization, which can be classified into three levels: mild, moderate and severe. Neurotropic and viscerotropic diseases are two severe adverse events associated with the vaccine.1-3
Brazil has a large enzootic area for sylvatic yellow fever, where every year cases of the disease are reported in unvaccinated individuals that have contact with vectors and sources of the disease sylvatic cycle.1,2
The sylvatic cycle is transmitted between non-human primates and sylvatic mosquitoes, mainly those belonging in the genera Haemagogus and Sabethes that live in treetops.3 On the other hand, the urban cycle has the mosquitoes in the genus Aedes as the main transmitters, being the human the only infection source to these vectors.3,4
Nowadays, in cyclical intervals from three to seven years, there can be outbreaks of the sylvatic yellow fever, which result from the epizootics in non-human primates. In these animals, the disease is periodically spread with intervals that contribute to the appearance of new vulnerable populations.3
About half century after an epidemiological silence, the state of São Paulo recorded again autochthonous cases of the disease.5 Despite the efforts to vaccinate the population in risk areas of the state, the virus continues to spread.6 The risk areas - and, consequently, with vaccine recommendation - increased due to the detection of new areas with yellow fever virus.
Taking into account the spread of yellow fever virus in São Paulo State, this study aimed to describe the occurrence of yellow fever since its reintroduction and detection in the state, in 2000, up to 2010.
Methods
This is a descriptive study using secondary data. The study was conducted in São Paulo State, located in the Southeast of Brazil. This state has 645 municipalities over a territorial area of 248,209.3 km², representing 2.91% of Brazilian territory. With regard to the surveillance actions, São Paulo's territory was divided into 27 groups of epidemiological surveillance.
Concerning the epidemiological surveillance for yellow fever in the state, the occurrence of a unique autochthonous case of the disease is already considered as an outbreak, initiating the recommended containment measures, which are eco-epidemiological and entomological evaluations, followed by actions to contain the virus circulation.
The study information was obtained from previous data related to epidemiological investigation of human and animal confirmed cases of yellow fever in São Paulo State, conducted by the agents of the municipal and state surveillance, which are available at the Immunization and Zoonosis Division of Epidemiological Surveillance Center "Prof. Alexandre Vranjac" of the State Health Department. The reports of the surveillance conducted were also revised.
Data on human cases, human deaths, epizootics in non-human primates and virus vector isolation were included.
The study was submmited to the Committee on Ethics in Research of the College of Medical Sciences of the Santa Casa of São Paulo, being dispensed from evaluation, because the available data did not have personal identification.
Since this study is part of the Training Program in Epidemiology Applied to the Services of the Brazilian National Health System in São Paulo State, and this program belongs to São Paulo State Health Department, the data has been made available, and its use and publishing have been authorized.
Results
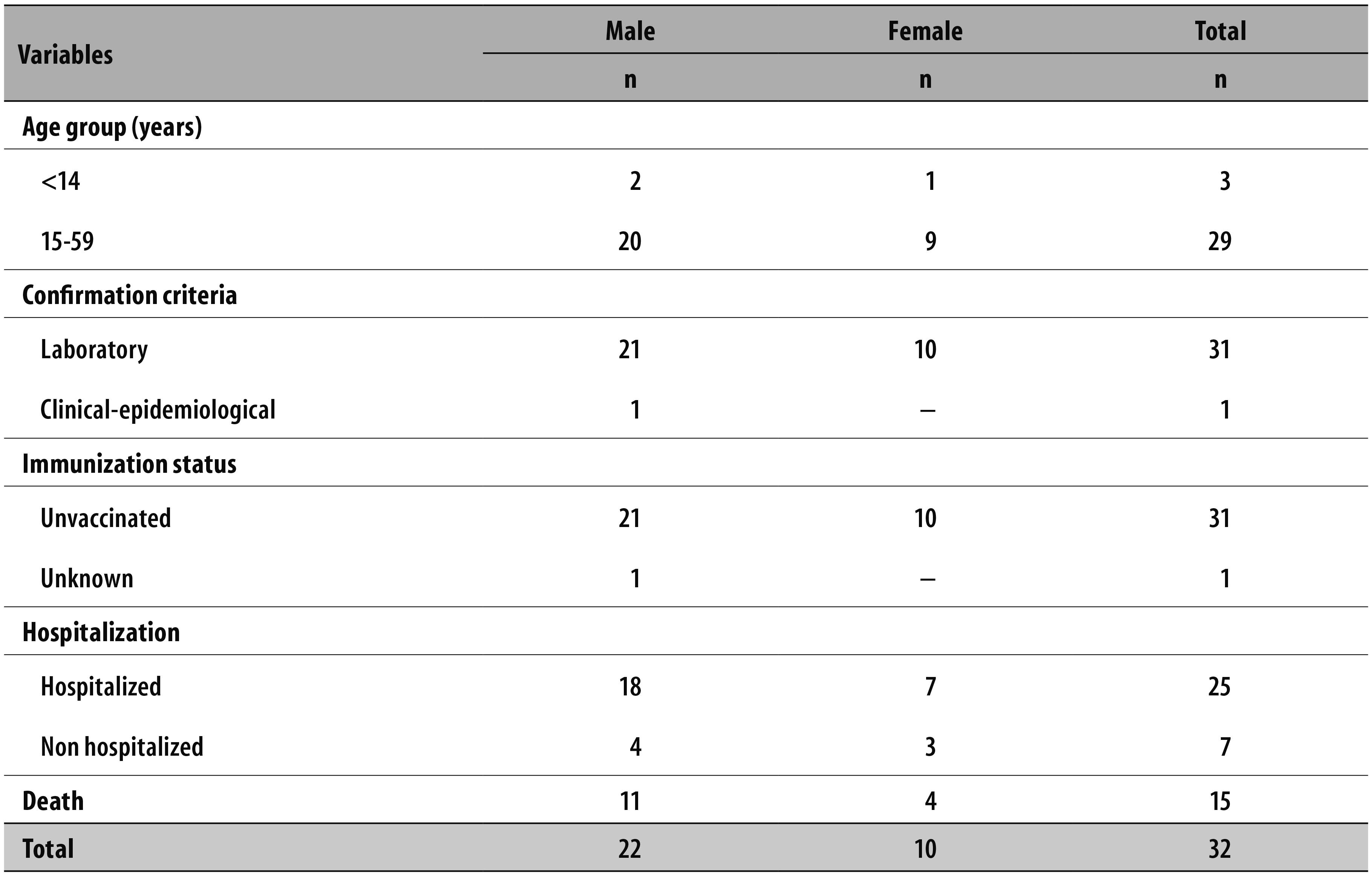
São Paulo State reported three outbreaks of autochthonous sylvatic yellow fever from 2000 to 2010. Thirty-two cases were registered with a global case-fatality rate of 46.8% (Tables 1 and 2). The disease affected mainly men between 15 and 59 years old. All the cases occurred in non-vaccinated individuals, who had contact with the sylvatic form of transmission of the disease (Tables 1 and 2).
Table 1 - Distribution of the yellow fever outbreaks, according to year of occurence, number of cases and the case-fatality rate in São Paulo State, 2000-2010
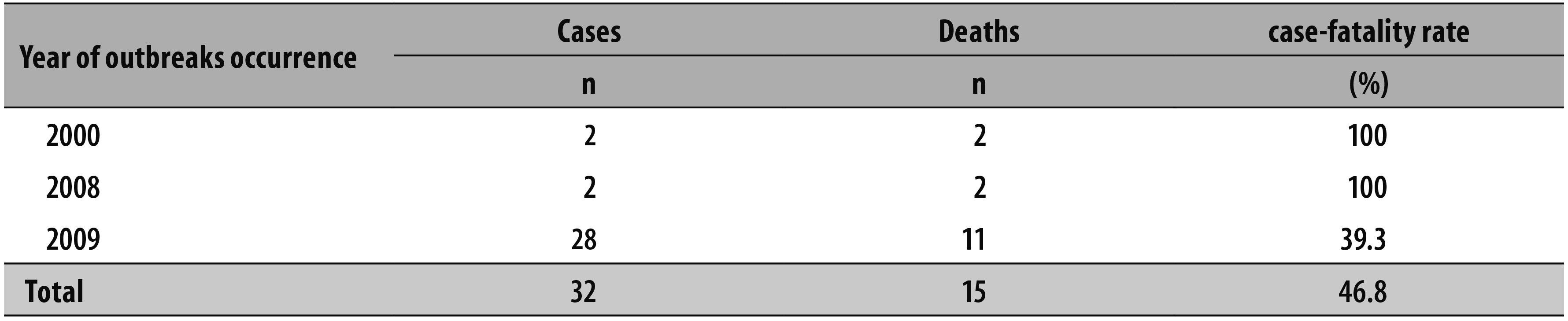
Table 2 - Distribution of yellow fever cases according to sex, age group, confirmation criteria, immunization status, hospitalization, number of deaths and activity performed in the probable place of infection in São Paulo State, 2000-2010
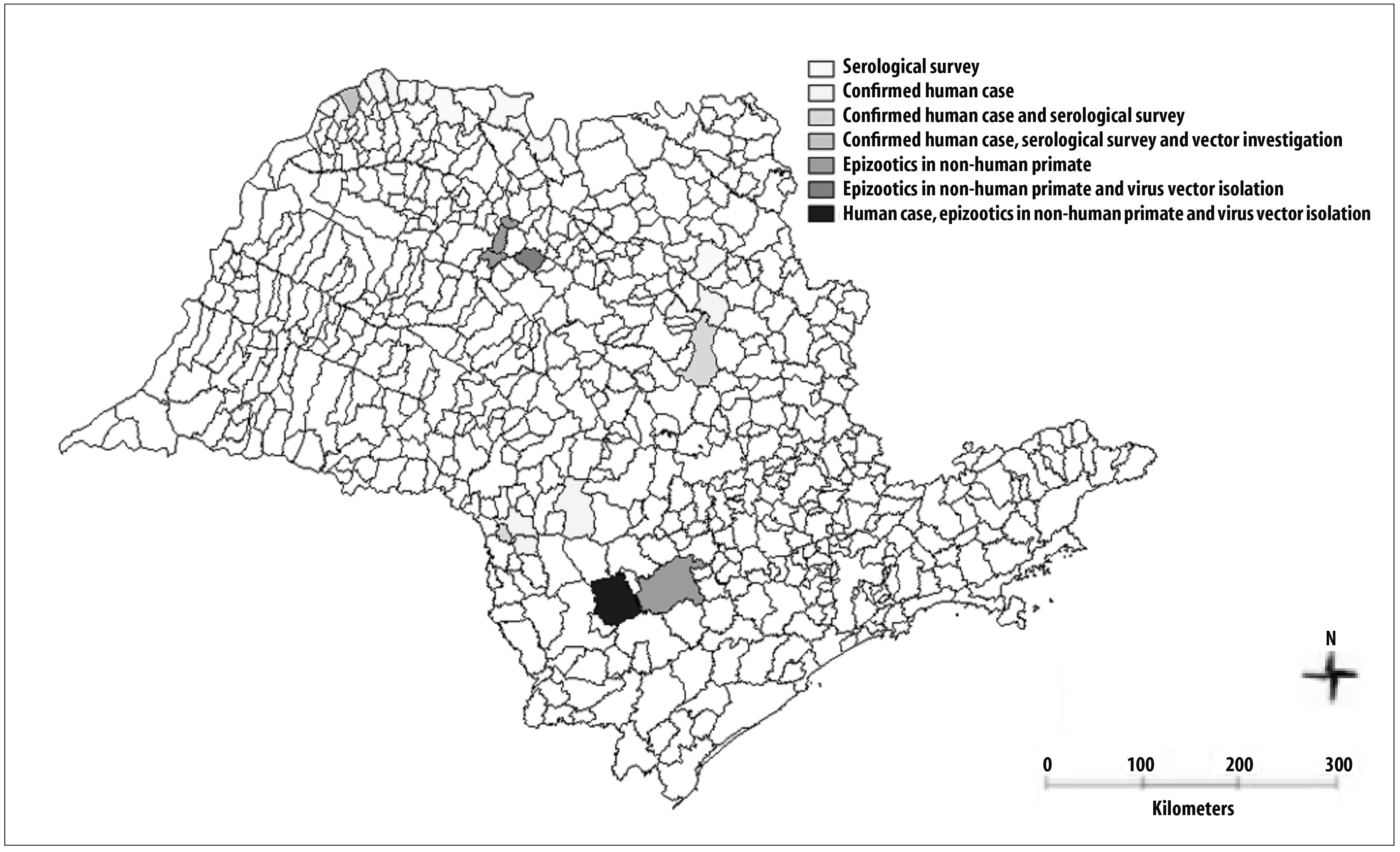
In March 2000, the São Paulo State Health Department received two notifications of sylvatic transmission of yellow fever in the Northwest region of the state, in the municipalities of Santa Albertina and Ouroeste, located at the margins of Rio Grande, with possible autochthonous transmission.
The patients were two men, aged 43 and 44, who lived in Dolcinópolis and Ribeirão Preto, respectively. These two individuals were unvaccinated and contracted the severe form of the disease, progressing to death 10 and 5 days - respectively - after the onset of symptoms. The confirmation criterion of the disease was laboratorial, with positive result in the immunohistochemistry test, which was carried out by the reference laboratory of the state, the Adolfo Lutz Institute (Table 2). An epidemiological investigation was conducted in the probable places of infection, pointing out that in both cases there were reports of fishing activity next to the river Rio Grande, on the border with the states of Minas Gerais and Goiás.
A serological investigation was performed with the riverside populations in 13 municipalities situated close to Rio Grande. The entomological evaluation was conducted by capturing the mosquitoes in the municipality of Santa Albertina. Dead or ill non-human primates were not found in that region. Six samples of serological investigation had a monotypic response to yellow fever virus; however, it was not possible to conclude that those cases were autochthonous, nor was the virus isolated from the vector7 (Table 2 and Figure 1).
The state broadened the areas with vaccine recommendation due to the virus reintroduction in the municipalities of Ouroeste and Santa Albertina. From 2001 on, 277 municipalities were in the recommendation area, which covers nine groups of epidemiological surveillance (Figure 2).
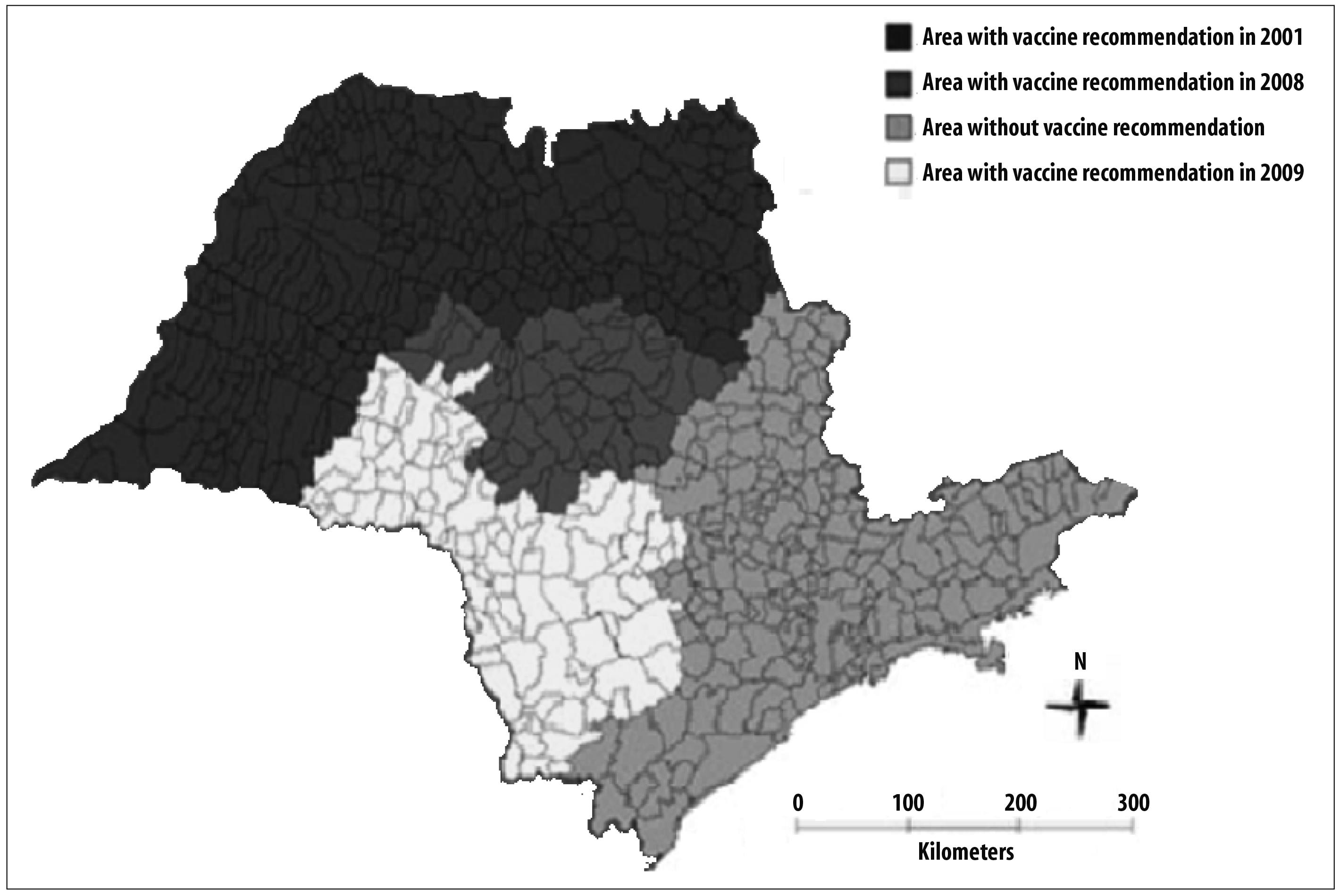
Figure 2 - Expansion of recommendation areas of the yellow fever vaccine in São Paulo State, 2000-2010
In 2003, the State Health Department created the passive death surveillance of non-human primates in areas with risk of yellow fever transmission in the state, in a partnership with the Municipality Zoonosis Control Centers, the Municipality Health Departments, the Superintendence for Endemic Control, the Environmental Police and the Schools of Veterinary Medicine.
Between 2004 and 2007, there was no report of in non-human primates' deaths in the state. In 2008, in the period from January to June, the death of 140 monkeys was reported in transition areas of yellow fever, allowing the laboratory investigation of 96 (68.6%) samples at the Adolfo Lutz Institute. The positive result in the samples from the municipalities of Mendonça, Nova Aliança and Urupês (01/14/2008, 02/18/2008 and 02/18/2008, respectively) confirmed, through laboratory tests, the epizootics occurrence of yellow fever virus in the region of São José do Rio Preto.8,9 Besides that, the death of a non-human primate due to yellow fever was confirmed by clinical-epidemiological test, in the municipality of Bady Bassitt, located in the same region.8-10
After confirming the etiologic diagnosis, 13 municipalities were defined as priority to prevent and control the disease. This definition was based on a 30-kilometer radius from where the carcasses of the primates were found. In addition to the vaccine intensification, since the municipalities that registered epizootics cases were already in the area of vaccine recommendation since 2001 (after the virus reintroduction in the state), other actions were developed in three phases: (i) vaccination and search for symptomatic individuals visiting all the households, situated both in rural and urban areas, (ii) expansion of the vaccine recommendation area to 16 municipalities, including the region of São José do Rio Preto, and (iii) assessment of immunization coverage in the other municipalities of the groups of epidemiological surveillance.8-10 Apparently, these actions were able to prevent the occurrence of human cases during that period, which shows the importance of the epizootics surveillance.
In April and May 2008, two autochthonous human cases of the disease were reported in the region of Ribeirão Preto. Both cases occurred with 39 year-old males, unvaccinated, residents in the municipalities of Cravinhos and Luiz Antônio, and who had been exposed to risk activities (fishing). Both patients contracted the severe type of the disease, progressing to death within 5 and 4 days - respectively - after the onset of symptoms.9
The first person to develop the symptoms lived in Cravinhos and the infection occurred at the Ecological Station of Jataí, in the municipality of Luís Antônio: the symptoms began on April 22, 2008 and the death occurred on April 26, 2008. In the second case, the person lived in the municipality of São Carlos, and developed the symptoms on May 23, 2008 and died on May 26, 2008. Both cases occurred in the rural area of São Carlos, on the border with the municipality of Rincão, and next to the Mogi-Guaçu River and the Ecological Station of Jataí. The confirmation criterion for the disease was laboratorial, and both cases had positive result in the immunohistochemistry test, which was carried out by the Adolfo Lutz Institute (Table 2).
The positive results in the laboratory samples confirmed the virus circulation and initiated an epidemiological investigation, in which samples of humans, monkeys and vectors were collected (Table 3).
Table 3 - Positivity for yellow fever virus in samples collected in the outbreak years in São Paulo State, 2000, 2008 and 2009
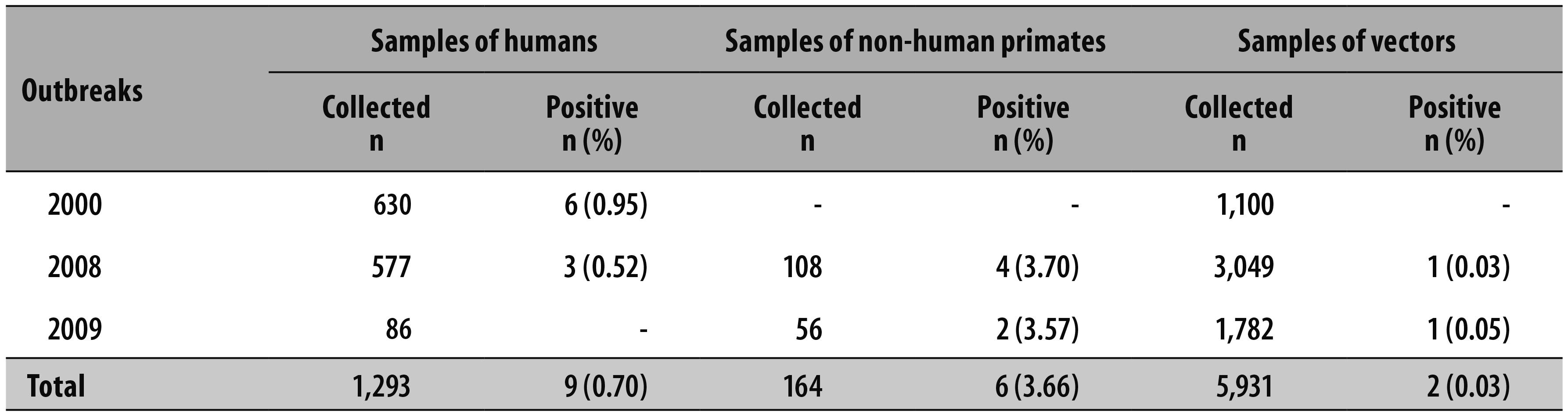
During the investigations, a serologic survey was conducted with 128 human samples, from the municipalities of São Carlos, Rincão and Ribeirão Preto, (close to the likely locations of infection); other 10 samples of patients' contacts were collected. Three samples were positive for IgM antibodies of the yellow fever virus, four monkeys tested had a positive diagnosis, and the virus of mosquitoes from Urupês region was isolated. (Table 3 and Figure 1).8-10
This spread of virus circulation resulted in the increase of vaccine recommendation areas for residents and travelers. In 2008, the state included other 62 municipalities in the vaccine recommendation area, in the groups of epidemiological surveillance of the municipalities of Araraquara and Bauru9,11,12 (Figure 2).
In 2009, the disease presented an epidemic characteristic: 138 suspected human cases were reported in Botucatu region, from February to April. This situation was considered an Public Health emergence of National Importance, according to the International Health Regulations 2005. From the analyzed cases, 110 were negative and 28 were confirmed in five municipalities - Sarutaiá, Piraju, Avaré, Buri and Tejupá -, none of them is in the vaccine recommendation area.11,13,14
From the reported cases, 11 progressed to death, with an average of 7 days between the onset of symptoms and the death (case-fatality rate: 39.3%). The probable places of infection were the rural areas of the municipalities of Sarutaiá, Piraju, Tejupá, Avaré and Buri. Ten cases occurred in agricultural workers with activities in the forest, and 18 cases in individuals who had participated in leisure activities. Among the 28 confirmed cases, 18 (64.3%) cases occurred in males, and the age varied from 3 days of life to 52 years old.11,13
The confirmation of yellow fever virus circulation in the municipalities of Avaré, Buri, Itapetininga, Piraju, Sarutaiá and Tejupá resulted, once more, in the increase of the vaccine recommendation area in São Paulo State. From 2009 on, 49 municipalities were included in the areas with vaccine recommendation, including more five groups of epidemiological surveillance (Figure 2).11,12,14,16
The immunization actions immediately began after the confirmation of the first case in Sarutaiá and they were expanded to the probable places of virus circulation. A total of 1,018,705 doses of the vaccine were distributed, reaching vaccination coverage of 86.8% in the period from March and April 2009. During immunization campaign, three cases of acute neurotropic disease and one case of immediate hypersensitivity were confirmed, and all of them progressed to death, and there were five cases of acute viscerotropic disease that also progressed to death - approximately 1 death per 200,000 doses and 1 severe adverse event case per 113,000 doses administered.11
Discussion
Although the virus has reemerged in São Paulo State after decades without cases, the disease continues to affect mostly men, in the economically active segment of the population, the rural workers and individuals who expose themselves to risk activities (fishing, hunting, ecotourism, among others) in the warmer months of the year, in areas with high levels of rainfall and, consequently, higher vector density. Previously, yellow fever was considered an occupational disease; however, nowadays, a new type of risk groups can be identified: unvaccinated individuals who expose themselves to ecotourism activities in river banks or in areas with fragments of preserved forest.4
Throughout history, outbreaks and epizootics have been reported in Brazil (and in other endemic countries), from time to time. Similar to Brazil, after decades of silence, yellow fever outbreaks reemerged in Africa, representing risk for the entire population of the continent.17 The increase in migration, consequently in urbanization, and the travel infrastructure improvement are global tendencies that expand the risk of the disease circulation in part of the world where the disease had disappeared.17,18
Some experts have been claiming that the epidemics repeat in the East of Africa because its savannah is relatively dry and, consequently, the population density of mosquitoes is lower than the most humid savannah in Central Africa. Thus, the human population of the savannah in the East of Africa can be exposed to the virus less often than the population of Central Africa, and a large number of vulnerable young people, just enough to maintain an outbreak, grow over the years, between the epidemics. On the other hand, epidemiological studies suggest that a persistent "silence transmission" (without outbreaks of perceived diseases) can occur among the inhabitants of the most humid savannah in Central Africa, creating a type of herd immunity.17,18
In the Amazon region and other endemic areas in Brazil, the outbreaks and epizootics were reported every 5 to 7 years. This frequency is probably related to the renewal of non-human primates' population, which is essential to expand the virus; however this situation does not occur with the vectors' population, even considering the possibility of a transovarial or vertical transmission.4,5,19
In the current outbreaks registered in different regions of the country, in the states of Minas Gerais20 and Rio Grande do Sul,21 men are the most affected by the disease. In São Paulo State, the disease incidence was twice higher in men than in women. The most affected age group was the economically active population, which is similar to the pattern described for Brazil.4 In the largest outbreak, registered in 2009, the affected age group varied; three cases were reported in children under 14 years old, and one unusual situation occurred: a case of perinatal transmission was diagnosed.22
The case-fatality can be an indicator of the surveillance sensitivity to detect new cases, as well as the severity of the disease. The expected case-fatality rate of the disease, in all of its forms, ranges from 5 to 10% and the values above indicate low surveillance capacity to detect mild cases.20 In the three outbreaks explained before, the surveillance conducted an active search for oligosymptomatic and asymptomatic cases. There was no conclusion if the nine cases confirmed by the serological tests were autochthonous.
The limitations of this study were related to the use of secondary data, which aimed to describe the outbreaks. This study did not have access to the individual data on adverse reactions to the vaccine and in the one case of perinatal disease.
Even though the epizootics surveillance of non-human primates was implemented in the state in 2003, the occurrence of those events was already considered as a sentinel of yellow fever outbreaks in the country. Between 1999 and 2000, after the period of high transmission of sylvatic yellow fever in the Brazilian Midwest region, involving a record of human cases and monkeys' deaths in the same period, Brazil began to register the death of primates as a warning to the risk of yellow fever. Consequently, there was a fast adoption of measures to prevent human cases and to control the transmission.1
This tool was only useful in the 2008 epizootics, in São José do Rio Preto region; probably, because that region was already considered a risk area for the virus transmission, so the surveillance for these events was strong and the health agents were prepared, and the human population had already been vaccinated. Therefore, the measures of prevention and control were adopted before the virus started circulating in the human population or infected urban areas - with high population density and infested by Aedes aegypti -, with risk of the disease re-urbanization, just like happened in 2008, in the metropolitan region of Asunción, Paraguay.23 That outbreak was immediately controlled by the immunization of approximately one million people and through vector control activities.17,23,24 This was the first urban yellow fever outbreak reported in the South America since 1942 and raised concerns about the potential spread of the virus to non-endemic areas, without immunization coverage and with vectors for virus transmission, such as the Caribbean, Central and North America.23,24
Although three outbreaks have been confirmed in the analyzed period, the virus was able to infect a large number of people only when that population was not previously vaccinated. It is acknowleged the combination of key-factors in the epidemiology of disease transmitted by vectors: the ecology and host's behavior, the ecology and vector's behavior and the level of the population's immunity.2 In the first two outbreaks, only two cases were registered, probably, they were introduced by the presence of human or non-human infection sources and vectors in the probable places of infection. The high immunization coverage must have enabled herd immunization (stopping the transmission through high vaccine coverage in the population), restricting the transmission risk of the rest of the population. In the last outbreak, in 2009, even though the early start of immunization after the identification of the first autochthonous case, the population had not been previously vaccinated and it led to the occurrence of 28 cases. It is clear that the barrier imposed by the vaccine and the continuous and high rates of immunization coverage are, even today, the most important method to prevent and control the disease in areas where ecological conditions are favorable to the establishment of the transmission cycle.
In Africa, two different situations are reported: in several West African countries, mass immunization campaigns had resulted in the disappearance of the disease over the following 40 years, whereas countries, such as Nigeria, where there is no immunization routine, suffered large outbreaks of the disease in the 1980s. Actually, countries with immunization activities reported sporadic cases in rural and isolated communities, where the population had not been vaccinated. These data confirm the importance of the vaccine for controlling the endemics.25,26
The virus reintroduction in São Paulo State increased the vaccine recommendation area every time there was new identification of virus circulation. The state passed from 277 municipalities and 9 groups of epidemiological surveillance in 2001 to 429 municipalities and 15 groups with vaccine recommendation, in 2010.
Although it was previously regarded as one of the safest vaccine containing the live attenuated virus, the yellow fever vaccine can induce adverse events after immunization, which are classified into three levels: mild, moderate and severe.1,27 The frequent record of these events has forced the health authorities to be more careful before expanding the vaccine coverage, trying to establish a favorable balance between the risk of acquiring the natural infection and the risk of developing severe medical conditions after exposure to the vaccine virus.
Mass immunization campaigns have been associated with the increase in detection of the adverse events post-immunization.28 During these campaigns, the individuals' medical history may not be investigated properly. In addition, the vaccine in adults without previous immunity can increase the rates of severe events, because the risk is greater in the first immunization and appears to increase with the age.28,29 In this sense, performing campaigns after cases detection may represent an option that involves greater risk than the previous expansion of vaccine coverage.
The outbreaks occurred in Brazil between 2008 and 2009 were the first in the country to identify rates of neurotropic disease associated to the vaccine, similar to cases in the United States of America (0.8 per 100.000 doses administered), suggesting that the increase in the surveillance and more sensitive laboratory tests (specifically, real-time PCR for RNA detection of yellow fever virus in the cerebrospinal fluid) cooperated to detect these events - although the diagnosis of neurotropic disease can be conclusive only with the presence of specific IgM antibodies to yellow fever in the cerebrospinal fluid.29
Finally, in the period analyzed, three yellow fever outbreaks took place in São Paulo State with 32 confirmed cases. Almost all of the cases occurred in unvaccinated individuals and none of the cases were observed in vaccinated individuals. Even though the cases of adverse reaction can be associated with the vaccine, this is still the best way to prevent the disease and the virus circulation.
REFERENCES
1. Monath TP. Yellow fever: an update. Lancet Infect Dis.2001 Aug;1(1):11-20. [ Links ]
2. Reiter P. Yellow fever and dengue: a threat to Europe? Euro Surveill. 2010 Mar;15(10):19509. [ Links ]
3. Vasconcelos PFC. Febre amarela. Rev Soc Bras Med Trop. 2003 mar-abr;36(2):275-93. [ Links ]
4. Vasconcelos PFC. Yellow fever in Brazil: thoughts and hypotheses on the emergence in previously free areas. Rev Saude Publica. 2010 Dec;44(6):1144-9. [ Links ]
5. Tauil PL. Aspectos críticos do controle da febre amarela no Brasil. Rev Saude Publica. 2010 mai;44(3):555-8. [ Links ]
6. Rocco IM, Katz G, Tubaki RM. Febre amarela silvestre no Estado de São Paulo, Brasil: casos humanos autóctones. Rev Inst Adolfo Lutz. 2003;62(3):201-6. [ Links ]
7. Moreno ES,Rocco IM, Bergo ES, Brasil RA, Siciliano MM, Suzuki A, et al. Reemergence of yellow fever: detection of transmission in the State of São Paulo, Brazil, 2008. Rev Soc Bras Med Trop. 2011 mai-jun;44(3):290-6. [ Links ]
8. São Paulo. Secretaria de Estado de Saúde. Divisão de Zoonoses do Centro de Vigilância Epidemiológica Professor Alexandre Vranjac. Coordenadoria de Controle de Doenças da Secretaria de Estado da Saúde de São Paulo. Casos de febre amarela silvestre em residentes do Estado de São Paulo, 2007-2008. BEPA. 2008 jul;5(55):12. [ Links ]
9. Coimbra TL, Souza RP, Moreno ES. Febre Amarela: Epizootias e casos humanos no Estado de São Paulo, 2008. In: Anais da 21º Reunião Anual do Instituto Biológico; 2008 nov 3-6; São Paulo. São Paulo: Instituto Biológico; 2008. [ Links ]
10. Mascheretti M, Tengan CH, Sato HK, Suzuki A, Souza RP, Maeda M, et al. Febre amarela silvestre: reemergência de transmissão no estado de São Paulo, Brasil, 2009. Rev Saude Publica. 2013 out;47(5):881-9. [ Links ]
11. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Coordenação Geral de Doenças Transmissíveis. Áreas com recomendação de vacina contra a febre amarela no Brasil 2008 e 2009. Brasília: Fundação Nacional de Saúde; 2008. [ Links ]
12. São Paulo. Secretaria de Estado de Saúde. Divisão de Zoonoses do Centro de Vigilância Epidemiológica Professor Alexandre Vranjac.Coordenadoria de Controle de Doenças da Secretaria de Estado da Saúde de São Paulo. Febre Amarela Silvestre no Estado de São Paulo, 2009. BEPA. 2009;6(63) [ Links ]
13. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Febre amarela silvestre, Brasil, 2009: boletim de atualização - dezembro/2009: Emergências em Saúde Pública de Importância Nacional (ESPIN) de febre amarela Silvestre em São Paulo e no Rio Grande do Sul e a situação epidemiológica atual no Brasil (2008/2009) [Internet]. Brasília: Ministério da Saúde; 2009 [citado 2016 mar 18]. Disponível em: Disponível em: http://www.salto.sp.gov.br/arquivos/boletim_febre_amarela.pdf [ Links ]
14. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Guia de vigilância de epizootias em primatas não humanos e entomologia aplicada à vigilância da febre amarela. 2. ed. Brasília: Ministério da Saúde; 2014. [ Links ]
15. Moreno ES, Barata RC. Municipalities of higher vulnerability to sylvatic yellow fever occurrence in the São Paulo State, Brazil. Rev Soc Bras Med Trop. 2011 Nov-Dec;53(6):335-9. [ Links ]
16. Markoff L. Yellow fever outbreak in Sudan. N Engl J Med. 2013 Feb;368:689-69 [ Links ]
17. Ellis BR, Barrett AD. The enigma of yellow fever in East Africa. Rev Med Virol. 2008 Sep-Oct;18:331-46. [ Links ]
18. Saraiva MDG, Amorim RDS, Moura MAS, Santos ECS, Sampaio LS, Barbosa MGV, et al. Historical analysis of the records of sylvan yellow fever in the State of Amazonas, Brazil, from 1996 to 2009. Rev Soc Bras Med Trop. 2013 Mar-Apr;46(2):223-6. [ Links ]
19. Ribeiro M, Antunes CMF. Febre Amarela: estudo de um surto. Rev Soc Bras Med Trop. 2009 set-out;42(5):523-31. [ Links ]
20. Almeida MAB, Cardoso JC, Santos E, Fonseca DF, Cruz LL, Faraco FJC, et al. (2014) Surveillance for yellow fever virus in non-human primates in southern Brazil, 2001- 2011: a tool for prioritizing human populations for vaccination. PLoS Negl Trop Dis. 2014 Mar;8(3):e2741 [ Links ]
21. Bentilin MR, Almeida RAB, Coelho KI, Ribeiro AF, Siciliano MM, Suzuki A, et al. Perinatal transmisson of yellow fever, Brazil 2009. Emerg Infect Dis. 2011 Sep;17(9):1779-80 [ Links ]
22. Pan American Health Organization. Outbreak of yellow fever in Paraguay [Internet]. Washigton: Pan American Health Organization; 2008 [citado 2016 Mar 18]. Avaliable from: Avaliable from: http://www.paho.org/bulletins/index.php?option=com_content&view=article&id=161%3Aoutbreak-of-yellow-fever-in-paraguay&lang=en [ Links ]
23. Johansson MA, Arana-Vizcarrondo N, Biggerstaff BJ, Gallagher N, Marano N, Staples JE. Assessing the risk of international spread of yellow fever virus: a mathematical analysis of an urban outbreak in Asunción, 2008. Am J Trop Med Hyg. 2012 Feb;86(2):349-58 [ Links ]
24. World Health Organization. Emerging and other Communicable Disease, Surveillance and Control. Yellow fever: technical consensus meeting [Internet]. Geneva: World Health Organization; 1998 [citado 2016 Mar 18]. Avaliable from: http://www.who.int/csr/resources/publications/yellowfev/en/whoepigen9808.pdf [ Links ]
25. Garske T, Van Kerkhove MD, Yactayo S, Ronveaux O, Lewis RF, Staples JE, et al. Yellow fever in Africa: estimating the burden of disease and impact of mass vaccination from outbreak and serological data. PloS Med. 2014 May;11(5):e1001638 [ Links ]
26. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Manual de vigilância epidemiológica de febre amarela. Brasília: Ministério da Saúde; 2004. [ Links ]
27. Waldman EA, Luhm KR, Monteiro SAMG, Freitas FRM.Vigilância de eventos adversos pós-vacinação e segurança de programas de imunização. Rev Saude Publica. 2011 fev;45(1):173-84. [ Links ]
28. Camacho LAB, Freire MS, Leal MLF, Aguiar SG, Nascimento JP, Iguchi T, et al. Immunogenicity of WHO-17D and Brazilian 17 DD yellow fever vaccines: a randomized trial. Rev Saude Publica .2004 Oct;38(5):671-8. [ Links ]
29. Romano AP, Costa ZG, Ramos DG, Andrade MA, Jayme VS, Almeida MA, et al. Yellow fever outbreaks in unvaccinated populations, Brazil, 2008-2009. PLoS Negl Trop Dis. 2014 Mar;8(3):e2740 [ Links ]
Received: December 28, 2015; Accepted: March 02, 2016











 texto em
texto em