Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.27 no.1 Brasília mar. 2018 Epub 11-Jan-2018
http://dx.doi.org/10.5123/s1679-49742018000100002
ORIGINAL ARTICLE
Microcephaly in Piauí, Brazil: descriptive study during the Zika virus epidemic, 2015-2016
1Ministério da Saúde, Secretaria de Vigilância em Saúde, Brasília, DF, Brasil
2Secretaria de Estado da Saúde do Piauí, Superintendência de Atenção Integral à Saúde, Teresina, PI, Brasil
3Secretaria de Estado da Saúde do Piauí, Instituto de Doenças Tropicais Natan Portella, Teresina, PI, Brasil
OBJECTIVE:
to describe the occurrence and characteristics of microcephaly cases in Piauí, Brazil, during an epidemic of Zika virus infection in 2015-2016.
METHODS:
descriptive study using data of live births from January/2015 to January/2016, obtained from the Information System on Live Births (Sinasc), the Public Health Events Registry (RESP) and the active search for medical records; mothers and live births were tested for dengue, chikungunya, and Zika, besides syphilis, toxoplasmosis, rubella, cytomegalovirus, and herpes (STORCH).
RESULTS:
of the 75 microcephaly cases, 34 were related to congenital infectious process; microcephaly prevalence was of 13.6/10 thousand live births; imaging exams confirmed that 34 live births presented calcifications, 23 had cerebral atrophies, 14 had lissencephaly, 12 had ventriculomegaly and 6 had dysgenesis; none tested positive for STORCH, dengue or chikungunya; 1 was IgM positive for Zika.
CONCLUSION:
there was an outbreak of microcephaly in Piauí, possibly related to infection during pregnancy Zika virus.
Keywords: Microcephaly; Zika Virus Infection; Epidemiology, Descriptive
Introduction
Congenital microcephaly is a neurologic anomaly in which the brain does not develop properly and the head circumference (HC) measured at birth is below than expected for the corresponding gestational age at birth and sex.1 The World Health Organization (WHO) defines microcephaly as an HC below minus two standard deviations (HC<-2SD), according to sex and gestational age at birth.2 Such parameters are determined through population-based analyses and standardization of measurement techniques, followed by application of reference calculations and charts, as follows: Fenton chart is used for preterm births (gestational age lower than 37 weeks of pregnancy); and WHO 2006 Child Growth Standards for full-term and post-term births (from 37 weeks of pregnancy onwards).1,3
Microcephaly may be related to numerous environmental and genetic factors. Perinatal hypoxia, congenital infections caused by TORCH (syphilis, toxoplasmosis, rubella virus, cytomegalovirus and herpes simplex virus type 2), intrauterine exposure to ionizing radiation, alcohol, drug abuse, and maternal phenylketonuria (maternal PKU) are among the main environmental factors. Single-gene disorders (Mendelian disorders), chromosome abnormalities or multifactorial disorders (polygenic disorders) are among the main genetic factors. Some examples of genetic disorders related to microcephaly are Seckel, Smith-Lemli-Opitz, Apert, Crouzon, Saethre-Chotzen, Pfeiffer and Carpenter syndromes.4,5
In cases of infection-related microcephaly, the most frequent neurological signs described are brain calcification, cerebral atrophy, dysgenesis of the corpus callosum, ventriculomegaly and lissencephaly. The imaging methods used to identify such alterations are ultrasonography, computed tomography (CT scan), and magnetic resonance imaging (MRI).1,6-8 Most cases of microcephaly are accompanied by motor and cognitive alterations, which vary according to brain damage.2
From 1995 to 2008, the worldwide prevalence of severe microcephaly, including newborns with HC lower than minus three standard-deviations (HC<-3SD) for age and sex, was of 5.9 cases per 100 thousand live births.9,10 In the same period, the prevalence was of 3.7/100 thousand in Latin America and 5.1/100 thousand in Brazil, according to the Latin American Collaborative Study of Congenital Malformations (ECLAMC).9,10 A study using data from the Information System on Live Births (Sinasc) estimated an annual average of 164 cases of microcephaly from 2000 to 2014; in 2015, however, 1,608 cases were reported, with a prevalence of 54.6/100 thousand live births.11
In October 2015, six months after the confirmation of autochthonous transmission of the Zika virus in the Northeast region of Brazil,12 an unexpected increase in the occurrence of microcephaly was observed, especially in Pernambuco State, with the confirmation of 40 children born since August 2015.13 Studies carried out during the same period have also detected an increase in microcephaly occurrence in Brazil, in comparison to previous years, mainly in the Northeast.1,14 Based on evidence gathered from epidemiologic investigation, the Brazilian Ministry of Health was pioneer in indicating the Zika virus implication in the microcephaly outbreak and, in November 2015, declared it as a Public Health Emergency of National Concern (PHENC).1,13,14
On January 17th, 2016, the Pan American Health Organization (PAHO) informed that other 17 countries in the region of the Americas had confirmed local transmission of Zika virus.15 In light of strong suspicion about the connection between the Zika virus and the increase in microcephaly cases, the International Health Regulations Emergency Committee, convened by WHO, gathered on February 1st, 2016 and declared the situation as a Public Health Emergency of International Concern (PHEIC).15
The objective of this study was to describe the occurrence and the characteristics of microcephaly cases in the state of Piauí, Brazil, during the Zika virus epidemic in 2015-2016.
Methods
This is a descriptive study conducted with infection-related microcephaly cases among live births in Piauí, from January 1st, 2015 to January 26th,2016. Teams from the Brazilian Ministry of Health and from Piauí State Health Department conducted the investigation of the suspected cases from January 25th to February 15th 2016.
Piauí State, located in the Brazilian Northeast region, is composed by 224 municipalities. In 2015, it had a population of 3,204,028 inhabitants, according to data from the Brazilian Institute of Geography and Statistics (IBGE). In that same year, 49,253 live births were registered in the Information System on Live Births (Sinasc).1,11
Data from Sinasc and from the Public Health Events Registry (RESP) were used. Since 1990, Sinasc has been the Ministry of Health’s official source of information on birth records, and is based on the Certificate of Live Birth (CLB). Since its establishment, it presents high specificity for severe microcephaly, defined as HC<-3SD for age and sex, according to the appropriate curves. The physician who accompanied the child birth is responsible for filling out the CLB, in which there is a field reserved to describe congenital malformations, which is later coded according to the International Statistical Classification of Diseases and Related Health Problems (ICD-10) (field 34 of CLB). Logging this information into Sinasc, however, is restricted to professionals registered in the system. RESP was created in November 2015 and allows detecting possible microcephaly cases and investigating the occurrence of infection during pregnancy. It consists of an online form available to any health care professional, allowing them to notify microcephaly cases, and it has more sensitive criteria for notification than Sinasc (HC lower than or equal to 32cm, for both sexes). Professionals were advised to register in the system microcephaly cases detected since January 1st 2015, before the creation RESP.1,2
Aiming to increase the coverage of microcephaly case records, medical records were reviewed at Dona Evangelina Rosa Maternity Hospital (MDER), a reference center for the diagnosis and treatment of microcephaly cases, and at Santa Fé Clinic (CSF), the largest private maternity hospital in Piauí.
In both hospitals, a pediatrician measured the HC at birth, using a tape measure. Upon microcephaly detection, they performed a second measurement within 48 hours. Once confirmed, the case was registered in Sinasc and in RESP,1 sent to follow-up at MDER (reference hospital), and evaluated by pediatricians and neurologists trained to apply the Ministry of Health criteria.1 Confirmed cases were also referred for early stimulation and family psychosocial monitoring.
The medical records data were registered in a semi-structured form, with information regarding both mother and infant. The following variables were included:
mother’s socioeconomic, demographic and clinical information - name; age (in years); first antenatal care visit; education (last completed school grade); occupation (open field); household address; number of previous pregnancies, miscarriages and fetal death; presence of rash or fever during pregnancy (yes or no), with the respective dates of symptoms onset;
infant’s demographic and clinical information - name, sex (male or female); date of birth; gestational age (in weeks); Apgar score of first and fifth minutes; head circumference at birth and 48 hours after birth (in centimeters); thoracic circumference (in centimeters); weight at birth (in grams); length at birth (in centimeters); moment of detection of microcephaly (intrauterine or after birth); place of birth (MDER or CSF); type of delivery (vaginal or cesarean section) -;
mother’s and infant’s laboratory tests results - serological markers and antigens detection for dengue, chikungunya, Zika and TORCH (positive, negative, not performed); antibodies testing (IgM, IgG, immunohistochemistry [IHC] and plaque-reduction neutralization test [PRNT]), antigen testing (virus isolation and bacterial culture) and molecular biology testing (Polymerase Chain Reaction [PCR]) in human blood and cerebrospinal fluid (CSF) samples-;
signs detected in infants through imaging tests (ultrasonography, CT or MRI) performed in all suspected cases - calcification, ventriculomegaly, lissencephaly, cerebral atrophy and dysgenesis of the corpus callosum (presence or absence) -;
The following case definitions were considered:
Microcephaly case: live birth with less than 37 weeks of gestational age (GA), with HC below percentile 3 of the Fenton chart or newborn with 37 weeks or more of GA, presenting HC lower than or equal to 32cm at birth.1-3
Suspected case of infection-related microcephaly: newborn with diagnosed microcephaly and registered in medical records, regardless of the underlying cause of lesion, assisted at one of the main state reference centers (MDER or CSF) from November 24th 2015 to January 26th 2016.
Confirmed case of infection-related microcephaly: suspected case with presence of alterations that suggest infection in any imaging exam AND/OR with specific and conclusive laboratory diagnosis for Zika virus or any other infectious agent (TORCH), identified in samples from the infant or from the mother.
Case discarded for infection-related microcephaly: suspected case with laboratory or imaging results without alterations that suggest infection in any imaging method OR based on clinical criteria after investigation (reclassification of microcephaly case after new clinical assessment).
Inconclusive case for infection-related microcephaly: suspected case that, after investigation, presented no laboratory, imaging or post-clinical evaluation records that would allow relating the case with infectious process.
To calculate the monthly microcephaly prevalence in Piauí, the numerator was the total number of microcephaly cases registered in Sinasc (Code Q02 in ICD-10), in RESP and those identified through medical records review. The denominator was the number of live births registered in Sinasc by month. The prevalence was calculated per 10 thousand live births.
To describe the characteristics of microcephaly cases related to infectious process, we used data from the medical records of the confirmed microcephaly cases and their mothers, obtained from the aforementioned hospitals. The softwares Epi Info™ 7.1.5.0, Microsoft Excel® 2010 and Tabwin® 3.6b were used.
This study was exempted from submission to the Ethics Research Committee, because it refers to epidemiological surveillance conducted by professionals in the health services, in the context of a Public Health emergency situation. The ethical aspects included in the Resolution No. 510 of the National Health Council (CNS), dated April 7th, 2016 were observed. Consolidated results are presented, which ensures individual anonymity. The involved institutions agreed to provide the data.
Results
The estimated prevalence of microcephaly for the entire period was of 13.6 cases/10 thousand live births. The occurrence of microcephaly cases increased from September 2015 onwards, reaching a peak in December 2015 for the cases recorded in Sinasc and RESP, and a peak in January 2016 was observed for the cases identified through medical records review. In December 2015, the prevalence reached 75.9/10 thousand according to Sinasc and 91.6/10 thousand, according to RESP; and in January 2016, it was of 84.3/10 thousand, based on active hospital search (Figure 1).
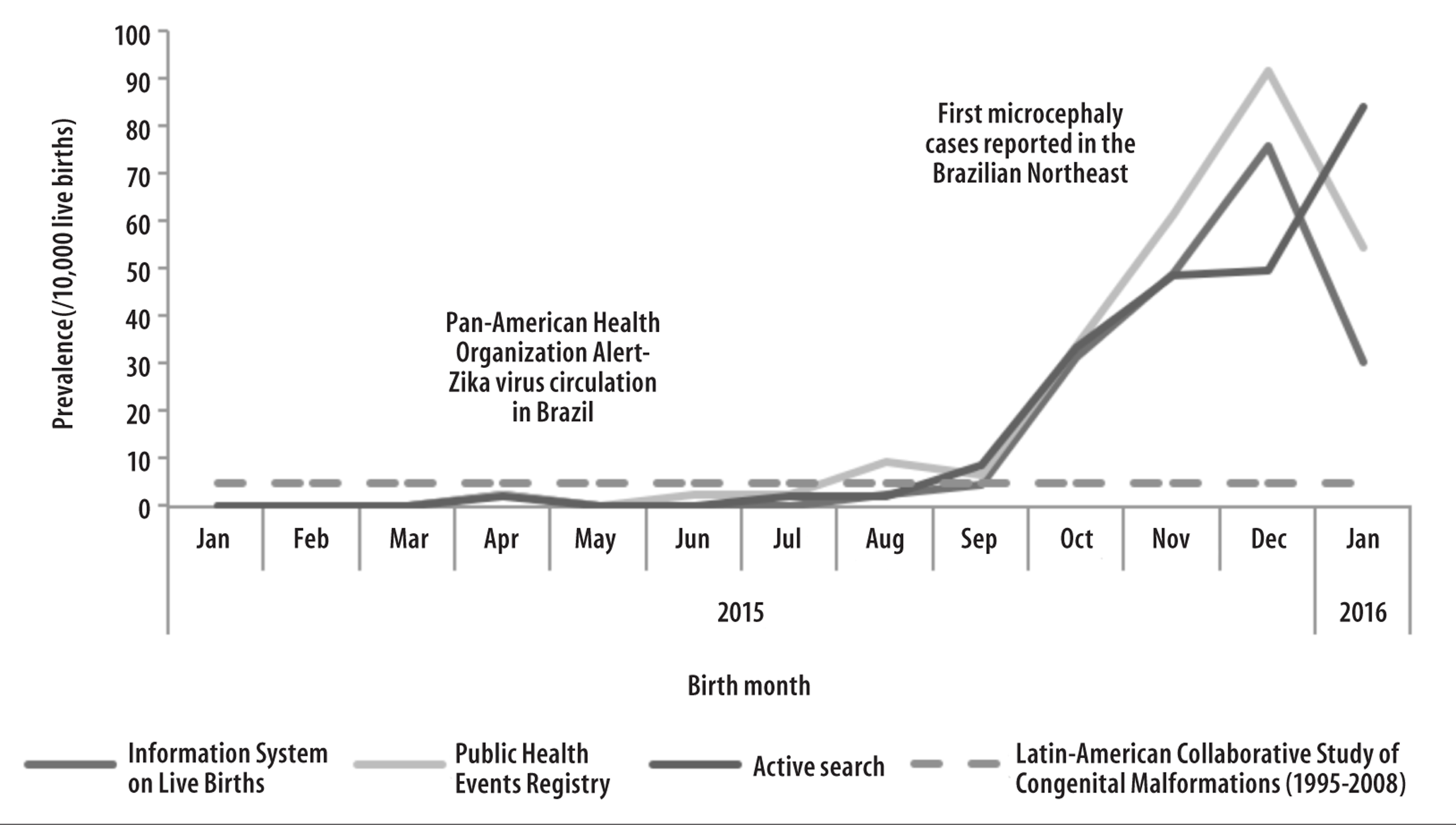
Figure 1 - Estimated prevalence of microcephaly in live births according to birth month in Piauí, January 2015-January 2016
A total of 75 live births met the definition for infection-related microcephaly: 24 were registered both in RESP and Sinasc, 34 only in RESP, 3 only in Sinasc and 14 in neither of the consulted databases. Ultimately, 34 cases were confirmed for infection-related microcephaly, 20 were discarded and 21 were classified as inconclusive due to lack of laboratory tests, imaging exams and medical records that could allow them to be classified as infection-related microcephaly.
The confirmed cases of infection-related microcephaly were born from the epidemiological week 39 in 2015; in the following epidemiological weeks, there was a continuous increase in the notification of cases, with a higher concentration between October and November 2015 (Figure 2).
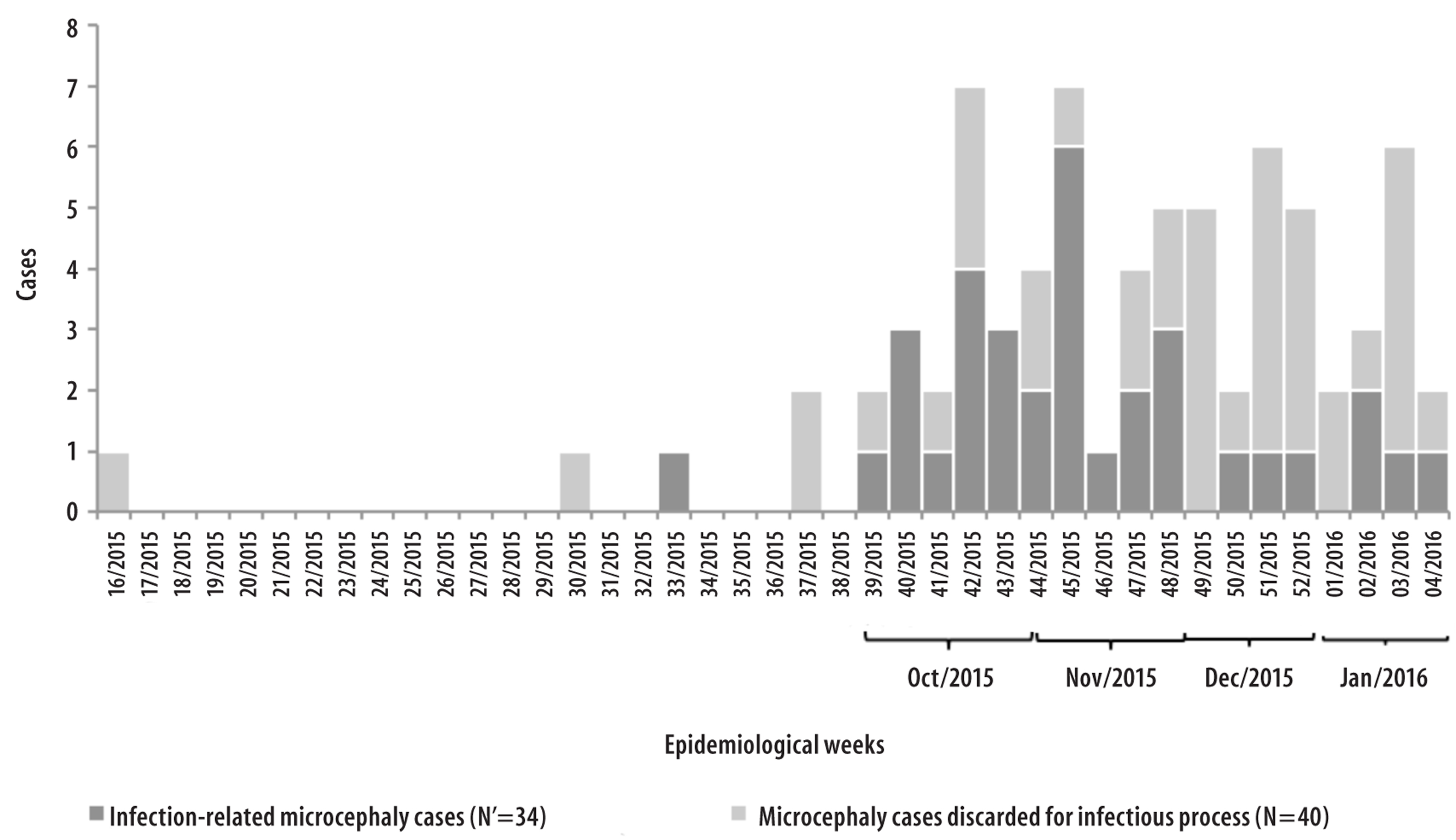
Figure 2 - Number of confirmed and discarded cases of infection-related, according to epidemiological week of birth,Piauí, April 2015 - January 2016
Out of the 34 confirmed cases, 24 were living in Teresina (capital of Piauí State) and 2 in Timon, a municipality in Maranhão State, close to Teresina. The other 8 cases were living in the municipalities of Altos, Barra, Brasileira, Itainópolis, Piripiri, Porto, Ribeiro Gonçalves and União (data not present in the table).
Mothers of the confirmed cases had a median age of 25 years with interquartile range (IQR) of eight years, 34 presented single pregnancy and 22 had a cesarean section. Ten mothers reported having had rash during pregnancy, six of them in the first trimester of pregnancy. Eight mothers presented fever during pregnancy, five of them during the first trimester of pregnancy. Seven mothers presented concomitant rash and fever with no known cause, five of them in the first trimester of pregnancy (data not present in the table).
Out of the 34 live births confirmed for infection-related microcephaly, 21 were female, with a median gestational age of 38 weeks (IQR=3 weeks). The HC at birth had a median of 29 cm (IQR=2 cm), and HC measured after 48 hours had a median of 33 cm (IQR=3 cm). The thoracic circumference had a median of 24 cm (IQR=3 cm). The medians of birth weight and length at birth were, respectively, 2,372 g (IQR=712 g) and 43 cm (IQR=2 cm) (data not presented in the table).
Among the mothers who underwent laboratory tests for the detection of dengue, chikungunya and Zika virus, two presented IgM positive results: one for dengue and one for chikungunya. No positive results were obtained for Zika virus. Regarding the infants, laboratory tests for dengue, chikungunya and Zika virus were conducted in 11 investigated cases, and one of them presented IgM positive for Zika virus (Table 1).
Table 1 - Results of the tests for dengue, chikungunya and Zika virus in mothers and live births (N=34) confirmed for infection-related microcephaly,Piauí, January 2015-January 2016
| Reagent/performed | Mothers’ tests | Live births’ tests | ||||
|---|---|---|---|---|---|---|
| Dengue | Chikungunya | Zika | Dengue | Chikungunya | Zika | |
| n | n | n | n | n | n | |
| IgMa | 1/17 | 1/15 | 0/1 | 0/11 | 0/11 | 1/1 |
| PCRb | - | - | 0/8 | - | - | 0/6 |
| NS1 Antigen | 0/7 | - | - | 0/5 | - | - |
| Virus isolation | - | - | - | - | - | - |
| PRNTc | - | - | - | - | - | - |
a) IgM: immunoglobulin M.
b) PCR: Polymerase Chain Reaction test.
c) PRNT: plaque-reduction neutralization test.
Among the mothers who underwent the recommended tests for Torch infections, one was confirmed for toxoplasmosis and one for syphilis. Eleven live births underwent such tests, resulting in a positive result for syphilis in the treponemal test (FTA-ABS) (Table 2).
Table 2 - Test results for the detection of syphilis, toxoplasmosis, rubella, cytomegalovirus and herpes in mothers and live births (n=34) confirmed for infection-related microcephaly, Piauí, January 2015-January 2016
| Reagent/performed | Syphilis | Toxoplasmosis | Rubella | Cytomegalovirus | Herpes | |
|---|---|---|---|---|---|---|
| n | n | n | n | n | ||
| Mothers | IgMa | - | 1/19 | 0/16 | 0/16 | 0/14 |
| IgGb | - | 9/16 | 15/18 | 16/17 | 15/16 | |
| VDRLc | 3/17 | - | - | - | - | |
| FTA-absd | 1/3 | - | - | - | - | |
| PCRe | - | - | - | - | - | |
| Live births | IgMa | - | 0/11 | 0/11 | 0/11 | 0/11 |
| IgGb | - | 4/10 | 5/10 | 10/11 | 9/11 | |
| VDRLc | 1/11 | - | - | - | - | |
| FTA-absd | 1/1 | - | - | - | - | |
| PCRe | - | - | - | - | - | |
a) IgM: immunoglobulin M.
b) IgG: immunoglobulin G.
c) VDRL: Venereal Disease Research Laboratory nontreponemal test (VDRL).
d) FTA-abs: fluorescent treponemal antibody absorption (FTA-ABS) test.
e) PCR: Polymerase Chain Reaction test.
A total of 34 live births with microcephaly underwent imaging exams: 7 were subjected to transfontanellar ultrasound, 34 to CT scans, and 2 to MRI. Of those, 34 infants had calcifications, 23 had cerebral atrophies, 14 had lissencephaly, 12 had ventriculomegaly, and 6 had dysgenesis. Four live births presented more than one CT scan, performed in different periods, totaling 38 exams (Table 3).
Table 3 - Signs identified in imaging exams of live births confirmed for infection-related microcephaly (n=34), Piauí, January 2015-January 2016
| Signs | USG TF a | TC b | MRI c |
|---|---|---|---|
| n=7 | n=38 d | n=2 | |
| Calcification | 5 | 28 | 1 |
| Ventriculomegaly | 4 | 7 | 1 |
| Lissencephaly | 1 | 12 | 1 |
| Cerebral atrophy | 5 | 16 | 2 |
| Dysgenesis of the corpus callosum | - | 6 | - |
a) USG TF: transfontanellar ultrasound.
b) CT: computed tomography scan.
c) MRI: magnetic resonance imaging.
d) 4/34 of confirmed live births underwent two CT scans, totaling 38 results.
Discussion
The results show a microcephaly outbreak among live births in Piauí from September 2015 onwards. Prevalence during the outbreak was 2.6-fold the prevalence in Brazil and 3.7-fold the prevalence in Latin America, when compared to the one presented in the ECLAMC study for the period from 1995 to 2008.9 This increase happened after PAHO statement on the confirmation of Zika virus circulation in Brazil and the emergence of cases in neighboring states.16
The increase in microcephaly cases in Piauí is consistent with outbreaks in neighboring states, such as Pernambuco, Paraíba and Bahia.17,18 In addition, the introduction of Zika virus happened before the increase of microcephaly cases in Sinasc, as the literature indicates that the virus has been circulating in Brazil since October 2014.12,13 Laboratorial confirmation of infection in the first trimester of pregnancy strengthened the hypothesis of a possible relation between Zika virus and microcephaly cases.19
In 2015, the highest incidence of probable dengue cases in Piauí occurred in April20 and the highest prevalence of microcephaly cases in December, a period of nine months between the two events. As the clinical conditions are similar, and there was no surveillance of Zika virus in Brazil at that time, it is possible that in 2015 the cases of Zika infection were reported as probable cases of dengue, reinforcing the hypothesis that the mothers of microcephaly-borne infants presented Zika virus infection during the first trimester of pregnancy.19,21 The fact that the occurrence of Zika virus infection was not substantial in Piauí, at its outset, may be related to the relatively low infestation of Aedes aegypti mosquito in the state's municipalities, especially in the capital, Teresina, where the house infestation rate was below 1.0. In contrast, the capitals of the neighboring states were on alert, with rates of house infestation between 1.0 and 3.9.22
Another important factor to consider was the identification of laboratory evidence of recent dengue, chikungunya and TORCH infections among the mothers of the investigated live births, although there was no expression of these events in their children.
One newborn was IgM positive for Zika, which suggests connection between the microcephaly and previous virus infection. The blood sample was collected from the newborn 21 days after birth and the IgG data for Zika, for both mother and infant, were absent, besides the fact that the mother did not present fever or rash during pregnancy; however, it does not discard the possibility of recent infection in the newborn. According to the literature, fetal infections by dengue or chikungunya are rare, and usually non-teratogenic. The mothers presented a high frequency of IgG antibodies for TORCH (approximately 94% of those who underwent laboratory exams), which, possibly, contributed for not having mother-to-child transmission.17,19,23-25 Although only 10 out of the 34 investigated mothers had presented symptoms of rash and fever during pregnancy, the possible asymptomatic infection by Zika virus is a hypothesis for the presence of microcephaly in their children.12,17
This study contributes to the validation of imaging findings to identify cases of infection-related microcephaly, since 33/34 cases of microcephaly of infectious origin were identified by imaging, and only one was confirmed by laboratory tests. Studies have already shown the possibility of imaging tests being an indicator prior to confirmation by laboratory findings of the infectious process of microcephaly cases, evidence confirmed in this investigation.6
The calculated prevalence - with due caution - can be considered accurate, given the use of databases for detecting cases through the information systems; for proper monitoring of the cases, it is necessary to adequately fill in the forms and analyze the databases, in the places of medical assistance to pregnant women, which require trained personnel for the analysis based on other records (e.g., Mortality Information System (SIM) versus Sinasc, or even RESP versus Sinasc). A study carried out in Teresina in 2002 indicated that the reliability of the records of live births in Sinasc was good,26 and can be a parameter for capturing cases. However, another national study showed that 40% of the cases of congenital anomalies identified were not reported in Sinasc, recommending caution in the use that information on live births in Brazil.26,27
This study shows the low quality of prenatal care in Brazil, especially in the Northeast: half of the mothers investigated did not undergo the laboratory tests recommended during pregnancy, consistent with studies describing prenatal care in the country as partially adequate, due to the failure to hold the minimum number of visits and the required exams, which reflects on maternal and infant morbidity and mortality.28-30
As a first limitation of the study, there was difficulty in collecting information due to the difficult in reading the medical records or lack of data, impairing the classification of cases (mainly regarding the opportunity of maternal samples collection during gestation and postpartum) and reducing the number of cases that could be analyzed: 21 newborns with inconclusive data, of an amount of 75 investigated. To minimize this limitation, we prioritized the review of medical records by health professionals, in addition to consulting the clinicians in case of doubt.
Another limitation was the difficulty in diagnosing the infectious processes, given the different opportunity windows for collecting exams, both in the live births and in the mothers, which did not allow a precise understanding whether the mother’s infection reached the fetus. We tried to minimize this difficulty with a careful review of the clinical signs registered. In order to minimize the limitation resulting from cross-reactions between arboviruses in the diagnostic tests, we considered the tests performed at the Central Public Health Laboratory of Piauí, a state reference.
In conclusion, our findings show that there was a microcephaly outbreak, possibly related to the introduction of the Zika virus in Piauí. This conclusion is based on the temporal relationship between the Zika and microcephaly epidemics and the detection of the Zika virus infection in a newborn. Microcephaly cases were detected through clinical and imaging diagnoses, recorded in the official health information systems - Sinasc and RESP -, despite the difficulties in closing a diagnosis through laboratory exams.
Piauí State Health Department received the following recommendations: (I) monitor microcephaly and other congenital anomalies trends through comparative analyses of the Public Health Event Registry - RESP - and the Information System on Live Births - Sinasc -; (II) qualification of services for registration in those systems; (III) investigation of Zika virus infection in pregnant women with rash and in live microcephaly-borne infants; and (IV) inclusion of the Zika virus infection survey in prenatal care. In addition, it is recommended to conduct eco-epidemiological studies on the prevalence and competence of arboviruses in human infection, concomitant with the development of cohort studies to measure the risk of Zika virus infection, or assessing factors associated with microcephaly or other congenital malformations.
Referências
1. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Protocolo de vigilância e resposta à ocorrência de microcefalia relacionada à infecção pelo vírus Zika [Internet]. Brasília: Ministério da Saúde; 2015 [citado 2017 set 11]. 55 p. Disponível em: Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2015/dezembro/09/Microcefalia---Protocolo-de-vigil--ncia-e-resposta---vers--o-1----09dez2015-8h.pdf [ Links ]
2. World Health Organization. Centers for Disease Control and Prevention (US). International Clearinghouse for Birth Defects Surveillance and Research. Birth defects surveillance: a manual for programme managers [Internet]. Genebra: World Health Organization; 2014 [cited 2017 Sep 11]. 116 p. Available in: Available in: http://apps.who.int/iris/bitstream/10665/110223/1/9789241548724_eng.pdf?ua=1&ua=1 [ Links ]
3. Fenton TR, Kim JH. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 2013 Apr;13:59. [ Links ]
4. Ferreira H, Barbosa C. Microcefalia primária grave - revisão de 10 casos. Acta Pediatr Port. 2000;31(1):11-5. [ Links ]
5. Sá LMSMP. Intervenção precoce e microcefalia estratégias de intervenção eficazes [dissertação]. Lisboa (Portugal): Escola Superior de Educação João de Deus; 2013. [ Links ]
6. Aragão MFV, Linden VVD, Brainer-Lima AM, Coeli RR, Rocha MA, Silva PS, et al. Clinical features and neuroimaging (CT and MRI) findings in presumed Zika virus related congenital infection and microcephaly: retrospective case series study. BMJ. 2016 Apr;353:i1901. [ Links ]
7. Herrero MM, López JCC. Macro-microcefalia: trastornos del crecimiento craneal. In: García JN, Fernández CC. Protocolos diagnósticos y terapéuticos en pediatría - Tomo 1: genética, dismorfologia e neurología. 2. ed. Madrid: Asociación Española de Pediatría; 2008. p.185-93. [ Links ]
8. Society for Maternal-Fetal Medicine Publications Committee. Ultrasound screening for fetal microcephaly following Zika virus exposure. Am J Obstet Gynecol. 2016 Jun;214(6):B2-4. [ Links ]
9. Nazer HJ, Cifuentes OL. Congenital malformations in Latin America in the period 1995-2008. Rev Med Chil. 2011 Jan;139:72-8. [ Links ]
10. Castilla EE, Orioli IM. ECLAMC: the Latin-American collaborative study of congenital malformations. Community Genet. 2004;7(2-3):76-94. [ Links ]
11. Marinho F, Araújo VEM, Porto DL, Ferreira HL, Coelho MRS, Lecca RCR, et al. Microcefalia no Brasil : prevalência e caracterização dos casos a partir do Sistema de Informações sobre Nascidos Vivos (Sinasc), 2000-2015. Epidemiol Serv Saúde. 2016 out-dez;25(4):701-12. [ Links ]
12. Fantinato FFST, Araújo ELL, Ribeiro IG, Andrade MR, Dantas ALM, Rios JMT, et al. Descrição dos primeiros casos de febre pelo vírus Zika investigados em municípios da região Nordeste do Brasil, 2015. Epidemiol Serv Saúde. 2016 out-dez;25(4):683-90. [ Links ]
13. Vargas A, Saad E, Dimech GS, Santos RH, Sivini MAVC, Albuquerque LC, et al. Características dos primeiros casos de microcefalia possivelmente relacionados ao vírus Zika notificados na Região Metropolitana de Recife, Pernambuco. Epidemiol Serv Saúde. 2016 out-dez;25(4):691-700. [ Links ]
14. Secretaria Estadual de Saúde (PE). Secretaria Executiva de Vigilância em Saúde. Protocolo clínico e epidemiológico: microcefalia [Internet]. 2. ed. Pernambuco: Secretaria Estadual de Saúde; 2015 [citado 2017 set 11]. 42 p. Disponível em: Disponível em: http://portal.saude.pe.gov.br/sites/portal.saude.pe.gov.br/files/protocolo_microcefalia_versao02.pdf [ Links ]
15. Henriques CMP, Duarte E, Garcia LP. Desafios para o enfrentamento da epidemia de microcefalia. Epidemiol Serv Saúde. 2016 jan-mar;25(1):7-10. [ Links ]
16. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Situação epidemiológica de ocorrência de microcefalias no Brasil, 2015. Bol Epidemiológico. 2015;46(34):1-3. [ Links ]
17. Oliveira WK, Cortez-Escalante J, Oliveira WTGH, Carmo GMI, Henriques CMP, Coelho GE, et al. Increase in reported prevalence of microcephaly in infants born to women living in areas with confirmed Zika virus transmission during the first trimester of pregnancy - Brazil, 2015. Morb Mortal Wkly Rep. 2016 Mar;65(9):242-7. [ Links ]
18. Martines RB, Bhatnagar J, Keating MK, Silva-Flannery L, Muehlenbachs A, Gary J, et al. Evidence of Zika virus infection in brain and placental tissues from two congenitally infected newborns and two fetal losses - Brazil, 2015. Morb Mortal Wkly Rep. 2016 Feb;65(6):159-60. [ Links ]
19. Nunes ML, Carlini CR, Marinowic D, Kalil Neto F, Fiori HH, Scotta MC, et al. Microcephaly and Zika virus: A clinical and epidemiological analysis of the current outbreak in Brazil. J Pediatr. 2016 Mai-Jun;92(3):230-40. [ Links ]
20. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre de chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 52, 2015. Bol Epidemiológico. 2016;47(3):1-10. [ Links ]
21. Pessôa R, Patriota JV, Souza ML, Felix AC, Mamede N, Sanabani SS. Investigation into an outbreak of dengue-like illness in Pernambuco, Brazil, revealed a cocirculation of Zika, Chikungunya, and Dengue virus type 1. Medicine (Baltimore). 2016 Mar;95(12):e3201. [ Links ]
22. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Resultado do LIRAa: janeiro a fevereiro de 2015 [Internet]. Brasília: Ministério da Saúde; 2015 [citado 2017 set 11]. p. 29. Disponível em: Disponível em: http://lproweb.procempa.com.br/pmpa/prefpoa/ondeestaoaedes/usu_doc/liraa-jan-fev-2015msaude.pdf [ Links ]
23. Miranda MMS, Souza LMG, Aguiar RALP, Corrêa Júnior MD, Maia MMM, Borges RS, et al. Rastreamento das infecções perinatais na gravidez: realizar ou não? Femina. 2012 jan-fev;40(1):13-22. [ Links ]
24. Figueiredo LT, Carlucci RH, Duarte G. Prospective study with infants whose mothers had dengue during pregnancy. Rev Inst Med Trop. 1994 Sep-Oct;36(5):417-21. [ Links ]
25. Mussi-Pinhata MM, Yamamoto AY. Infecções congênitas e perinatais. J Pediatr . 1999;75(Supl1):S15-30. [ Links ]
26. Mascarenhas MDM, Gomes KRO. Confiabilidade dos dados do Sistema de Informações sobre Nascidos Vivos em Teresina, Estado do Piauí, Brasil - 2002. Ciên Saúde Colet. 2011;16(Supl 1):1233-9. [ Links ]
27. Luquetti DV, Koifman RJ. Qualidade da notificação de anomalias congênitas pelo Sistema de Informações sobre Nascidos Vivos (Sinasc): estudo comparativo nos anos 2004 e 2007. Cad Saúde Pública. 2010 set;26(9):1756-65. [ Links ]
28. Leal MC, Theme-Filha MM, Moura EC, Cecatti JG, Santos LMP. Atenção ao pré-natal e parto em mulheres usuárias do sistema público de saúde residentes na Amazônia Legal e no Nordeste, Brasil 2010. Rev Bras Saúde Matern Infant. 2015 jan-mar;15(1):91-104. [ Links ]
29. Tomasi E, Fernandes PAA, Fischer T, Siqueira FCV, Silveira DS, Thumé E, et al. Qualidade da atenção pré-natal na rede básica de saúde do Brasil: indicadores e desigualdades sociais. Cad Saúde Pública. 2017;33(3):e00195815. [ Links ]
30. Viellas EF, Domingues RMSM, Dias MAB, Gama SGN, Theme Filha MM, Costa JV, et al. Assistência pré-natal no Brasil. Cad Saúde Pública. 2014;30(Supl 1):85-100. [ Links ]
Received: February 10, 2017; Accepted: August 21, 2017











 texto em
texto em 


