Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.27 no.2 Brasília jun. 2018 Epub 05-Jun-2018
http://dx.doi.org/10.5123/s1679-49742018000200016
ORIGINAL ARTICLE
Outbreak of Guillain-Barré syndrome possibly related to prior Zika virus infection, Metropolitan Region of Recife, Pernambuco, Brazil, 2015
1Ministério da Saúde, Secretaria de Vigilância em Saúde, Programa de Treinamento em Epidemiologia de Campo Aplicada aos Serviços do Sistema Único de Saúde, Brasília, DF, Brasil
2Ministério da Saúde, Secretaria de Vigilância em Saúde, Coordenação-Geral dos Programas de Controle da Malária e das Doenças Transmitidas pelo Aedes, Brasília, DF, Brasil
3Secretaria Estadual de Saúde de Pernambuco, Secretaria Executiva de Vigilância em Saúde, Diretoria-Geral de Controle de Doenças e Agravos, Recife, PE, Brasil
Objetivo:
to investigate the occurrence of GBS in the Metropolitan Region of Recife, PE, Brazil, 2015.
Methods:
this was a descriptive study using data from the Hospital Information System, National Pharmaceutical Services Management System and interviews; GBS cases were classified according to Brighton criteria and prior infection according to laboratory and clinical criteria.
Results:
in 2015, the number of GBS hospitalizations had a threefold increase in comparison to 2014. We investigated 44 confirmed or probable GBS cases, of which 18 had symptoms of Zika infection up to 35 days before the occurrence of GBS, mainly rash; one case was laboratory-confirmed for Zika virus infection and one death was registered.
Conclusion:
the findings reinforce a possible relationship between GBS and Zika infection, given the absence of increased GBS occurrence in previous dengue epidemic years, absence of chikungunya transmission records, presence of clinical manifestations compatible with infection and a laboratory confirmation.
Keywords: Epidemiology, Descriptive; Disease Outbreaks; Guillain-Barre Syndrome; Zika virus; Brazil
Introduction
Guillain-Barré Syndrome (GBS) is an autoimmune inflammatory monophasic polyradiculoneuropathy, characterized by rapid ascending evolution of limb weakness, almost always symmetrical, hyporeflexia or areflexia and cell-protein dissociation in the cerebrospinal fluid. The disease can reach the maximum severity in up to four weeks, with the development of respiratory failure in approximately 25% of cases. Of these, the majority recover completely; however, there may be serious consequences and deaths in up to 20% and 5% of cases, respectively.1-6
Following the elimination of poliomyelitis, GBS has become the largest cause of flaccid paralysis in the world, representing a serious public health problem whose incidence may vary from 0.4 to 4 cases per 100,000 inhabitants in different regions of the world.4.6-9
GBS is diagnosed according to clinical presentation, electroneuromyography results and cerebrospinal fluid characteristics.1 In addition to clinical support, treatment is based on intravenous immunoglobulin (IVIG) or plasmapheresis immunotherapy.3.6
GBS is frequently preceded by an infection. There are also reports of GBS following vaccination or even after situations of stress induced by trauma, although both situations are considered rare and debatable.5.10 The agent most commonly associated with the disease is the Campylobacter jejuni bacterium, followed by cytomegalovirus, Epstein-Barr virus (EBV), Mycoplasma pneumoniae, Haemophilus influenzae, influenza A virus, hepatitis B and E, and human immunodeficiency virus (HIV) infection.3.6 Currently, there are also records of GBS associated with dengue, chikungunya and Zika virus infections.12-14
In Brazil, even with the occurrence of dengue epidemics in different periods and regions of the country since 1984, and the introduction of chikungunya virus in 2014, no significant increase in GBS hospitalizations had been noticed. However, in the first half of 2015, with the confirmation of Zika virus circulation and the Zika virus epidemic, especially in the Northeast region, the Brazilian Ministry of Health was notified of the increase in hospitalizations due to neurological manifestations in Pernambuco State, including encephalitis, optic neuritis, myelitis, encephalomyelitis and GBS, whereby the latter accounted for more than 80% of cases.
Given the unusual characteristic of this event, the Pernambuco State Health Department (SES/PE) requested the support of the Secretariat of Health Surveillance (SVS) of the Brazilian Ministry of Health in the investigation of GBS cases among residents of the Metropolitan Region of Recife (MRR), the state capital, where the majority of the cases were concentrated.
The study sought to investigate the occurrence of GBS in MRR in 2015, and its possible relation with prior dengue, chikungunya or Zika virus infection.
Methods
This is a descriptive study, including records of GBS hospitalizations and probable cases (confirmed and under investigation) of dengue and/or Zika in Pernambuco between 2010 and 2015, in addition to GBS cases in residents and those hospitalized in the Metropolitan Region of Recife (MRR) between January and June 2015. This study period - January-June/2015 - was selected considering that rumors about the increase of GBS cases in Pernambuco began to occur in May 2015.
MRR is comprised of 14 municipalities and the Fernando de Noronha archipelago, covering an area of 2,703,064 km² and having a population of 3,890,145 inhabitants.
A retrospective search was conducted in order to identify cases through a review of Hospital Inpatient Authorizations registered on the Brazilian Unified Health System Hospital Information System (SIH/SUS) using code G.61.0 of International Statistical Classification of Diseases and Related Health Problems 10th Revision (ICD-10) for GBS; and by tracking individuals recorded on the National Pharmaceutical Services Management System (Hórus), who used IVIg for treatment of GBS, dispensed by SES/PE Central Pharmacy.
We reviewed the medical records of the identified cases. According to the information recorded, they were included - or not - in this study, according to Brighton criteria, which define five levels of certainty of GBS diagnosis: levels 1 to 3 relate to confirmed cases; level 4 refers to suspected cases (clinical data required for classification missing from medical records); while level 5 refers to cases discarded for presenting an alternative medical diagnosis to the syndrome.1
In this present study, cases were classified according to the following definitions:
Suspected cases - individuals hospitalized and resident in MRR, with presumptive diagnosis of ICD-10 G.61.0 registered on Hospital Inpatient Authorizations and/or who received IVIg for treatment of GBS.
Probable cases - suspected cases with Brighton criteria certainty level 4.
These were considered to be probable cases because, despite the lack of information on the medical records, they were hospitalized in a neurology service and accompanied by neurologists, whereby clinical diagnosis recorded was considered to be sufficient.
Confirmed cases - suspected cases with Brighton criteria level of certainty 1, 2 or 3.
Discarded cases - suspected cases discarded by the physician in charge because the neurological manifestation received alternative diagnosis, corresponding to Brighton criteria level 5.
In order to investigate possible prior infection, based on data registered on medical records or laboratory results and interview reports, confirmed and probable GBS cases were thus classified:
GBS cases with a history of probable prior dengue, chikungunya or Zika (DCZ) infection - confirmed or probable GBS cases with clinical history of fever or rash up to 60 days before the neurological manifestation, or having a positive immunoglobulin M (IgM) serology test result for dengue or chikungunya, or not positive for either of these, or not having had a specific test at the appropriate time;
GBS cases with a confirmed history of DCZ infection - confirmed or probable GBS case with laboratory-confirmed dengue, chikungunya or Zika, through virus isolation or reverse transcriptase reaction followed by polymerase chain reaction (RT-PCR); and
GBS cases with a discarded history of DCZ infection - confirmed or probable GBS cases with no report or record of prior DCZ infection or discarded DCZ infection due to virus isolation and/or RT-PCR or other laboratory confirmed causes, such as Epstein-Barr virus, herpes virus, cytomegalovirus, Campylobacter jejuni, hepatitis and HIV.
Losses were considered to be individuals who received IVIg but for whom no Hospital Inpatient Authorization was found, those whose medical records were not found and those who were not at home or not answered the phone after three attempts to contact them at different times.
In order to diagnose virus infection prior to neurological manifestation, a search was performed for samples of serum, blood, cerebrospinal fluid (CSF), urine and feces of individuals studied, which were still stored at Pernambuco Central Public Health Laboratory (LACEN/PE) or at laboratories of facilities where these patients were hospitalized, this being material intended for etiological research. In addition, those who were still within the appropriate time interval for sample collection at the date of the interview could be submitted to new blood, urine and feces sample collection. The etiological research to be carried out was:
IgM serology for dengue and chikungunya, to be performed at LACEN/PE using blood or serum samples collected between the 7th and 60th day after the date of onset of viral infection symptoms;
DCZ virus isolation or RT-PCR , conducted by the Evandro Chagas Institute (run by the Health Surveillance Secretariat of the Brazilian Ministry of Health - laboratório de referência nacional para o diagnóstico de arboviroses), using samples of blood, cerebrospinal fluid and/or urine collected up until the 5th day after the onset of symptoms of prior infection; and
detection of poliovirus, carried out by the Oswaldo Cruz Institute Foundation, Rio de Janeiro (Fiocruz/RJ) on feces samples collected from children under 5 years of age, up to 14 days after the beginning of neurological manifestation.
At all stages, a structured questionnaire was used as a tool for data collection. It was applied using tablets and included data on individual identification, clinical and epidemiological history, laboratory tests, hospitalization, as well as neurological data and data on prior infections. With regard to vaccination, data were collected from information reported by interviewees and they were not required to provide their vaccination cards.
The variables studied were:
Patient identification: age (in years); sex (male; female); and municipality of residence.
Epidemiological history: history of surgery and pregnancy up until 60 days before the onset of neurological symptoms, vaccination up to 90 days before symptoms, comorbidities.
Clinical data: signs and symptoms of infection (fever, rash, pruritus, headache, myalgia, lumbago, retro-orbital pain, arthralgia, nausea, vomiting, conjunctivitis, joint swelling, other symptoms as reported by participants); signs and symptoms of GBS (motor disabilities, muscular weakness, numbness, weakness, paralysis, loss of sensitivity, spasticity, impairment of the respiratory muscles, impairment of the facial muscles, areflexia, hyporeflexia, hyperreflexia, impairment of sphincters, reduction or loss of consciousness, dysphagia, other symptoms as reported by participants); and period between signs and symptoms of infection and GBS itself (in days).
Hospitalization: place of hospitalization; hospitalization period (in days); and mechanical ventilation (yes; no).
Analysis of CSF: characteristics of CSF (number of cells and protein measurement); period between GBS and collection of CSF; results of serology for dengue or chikungunya; result of RT-PCR for DCZ.
Electroneuromyography: result compatible or not.
Received treatment for GBS; if yes, what treatment.
Databases containing aggregated data (with no identification of the subjects) from the Notifiable Diseases Information System (Sinan Net and Online), provided by SES/PE, were used as a source of data for probable cases of dengue and Zika. It should be noted that at that time SES/PE opted to issue guidance that suspected cases of Zika were to be notified on dengue notification forms.
The analysis were performed with the aid of Epi Info™, version 7.1.5.2, and Microsoft Office Excel 2010®, by means of descriptive statistics and with results presented in the form of absolute and relative frequencies, measures of central tendency and dispersion, rates of incidence and prevalence.
The source used to calculate GBS incidence rates was the population estimate for July 1, 2015, whilst the source used to calculate incidence rates by age and sex was the 2012 intercensitary projection. Both sources were provided by the SUS IT Department (Datasus).15
GBS incidence rates were calculated based on the ratio between the number of confirmed and probable cases and the resident population in MRR (per 100,000 inhabitants). The estimated prevalence of GBS cases with prior viral infection was calculated by the ratio between the cases of GBS with signs of infection and the number of confirmed and probable cases of GBS in the period studied, in percentages.
Before the interviews, participants were informed, verbally, about the purpose and methods of investigation, that their participation was voluntary and that confidentiality of information was guaranteed. The interview was conducted after verbal consent from each participant.
This study was conducted in the context of health surveillance actions, seeking to clarify an unusual situation which was considered to be a potential public health emergency, thus exempting it from submission to an Ethics Research Committee, as per National Health Council Resolution No. 510 of 7 April 2016.
Results
Pernambuco registered 31,395 probable cases of dengue in 2012, an epidemic year for dengue fever in that state, and 96 hospitalizations for Guillain-Barre syndrome. Between January and June of 2015, there were 66,488 registered probable cases of dengue and Zika and 128 GBS hospitalizations, three times more in relation to 2014 (N=43). When this investigation ended, no autochthonous cases of chikungunya had been recorded, thus indicating the absence of circulation of this virus in the Metropolitan Region of Recife (MRR) (Figure 1).
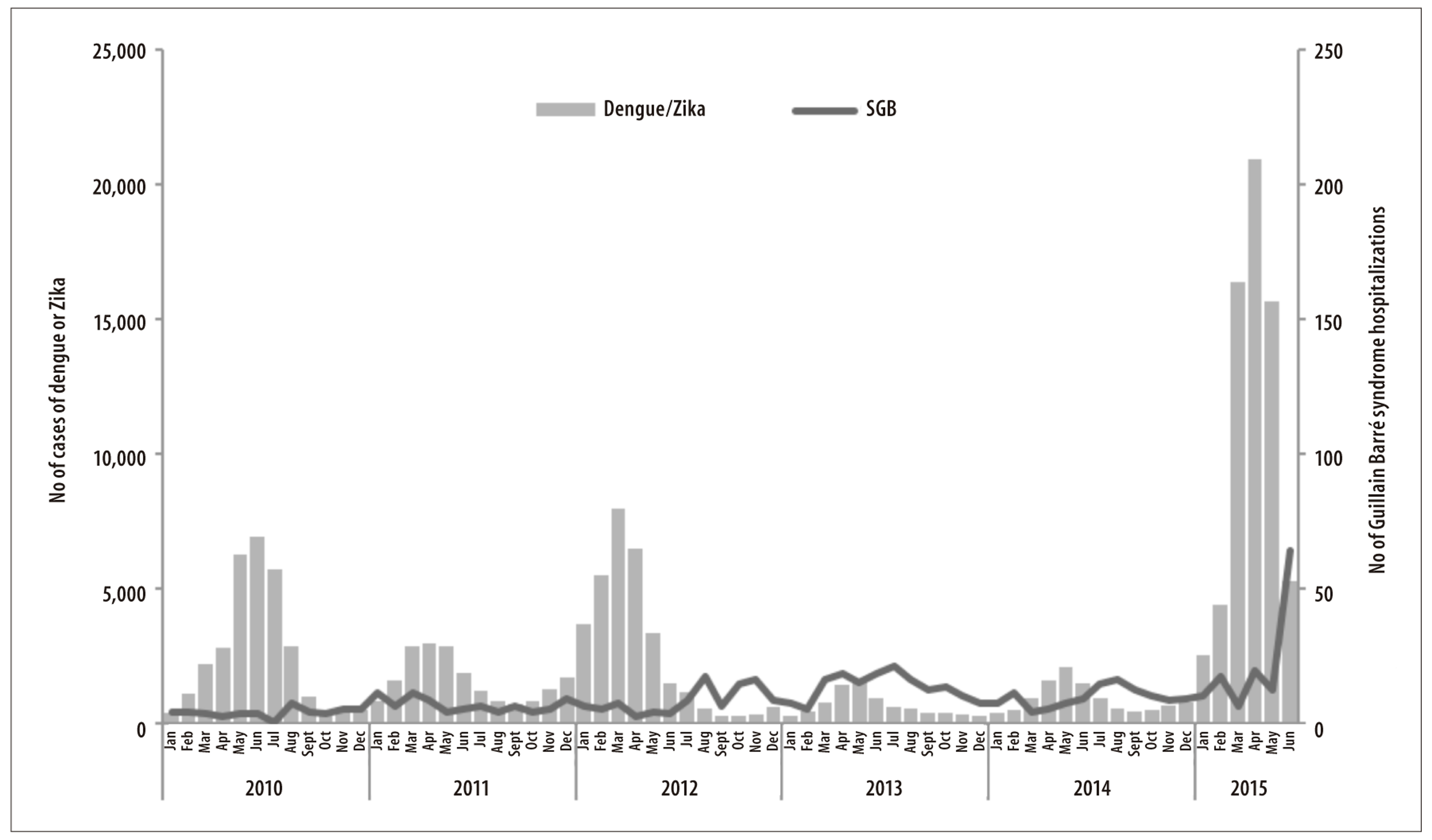
Source: Dengue/Zika -Sinan Net and Sinan Online; and SGB - SIH/SUS, SES/PE (Data updated on 29/07/2015).
Figure 1 - Distribution of cases of dengue or Zika and hospitalizations for Guillain-Barre syndrome, according to month and year of occurrence, Pernambuco, 2010-2015
For the descriptive study in the MRR, the investigation started from 56 GBS cases hospitalized between 1st January and 30 June 2015. Based on Brighton criteria, 12 cases were discarded, nine taken as probable and 35 were confirmed. Taking the 44 confirmed or probable cases, an incidence rate of 1.2/100,000 inhabitants in MRR was found. The rate was equal between the sexes. Incidence rates were higher in individuals aged 60 to 69 years (3.2/100,000 inhab.) and those living in Cabo de Santo Agostinho (2,1/100,000 inhab.), a municipality located 14 km from the capital, Recife (Table 1).
Table 1 - Confirmed and probable cases and incidence of Guillain-Barré syndrome, according to sex, age range and municipality of residence, Pernambuco, 2015
| Variables | N | Incidence (Per 100,000 inhabitants). |
|---|---|---|
| Sex | ||
| Female | 23 | 1.2 |
| Male | 21 | 1.2 |
| Age group (in years) | ||
| 10-19 | 3 | 0.5 |
| 20-29 | 6 | 0.9 |
| 30-39 | 9 | 1.3 |
| 40-49 | 11 | 2.1 |
| 50-59 | 6 | 1.6 |
| 60-69 | 7 | 3.2 |
| 70-79 | 2 | 1.7 |
| ≥80 | 1 | 1.8 |
| Municipality of residencea | ||
| Cabo de Santo Agostinho | 4 | 2.1 |
| Jaboatão dos Guararapes | 11 | 1.7 |
| Ipojuca | 1 | 1.2 |
| Recife | 19 | 1.2 |
| Abreu e Lima | 1 | 1.0 |
| Igarassu | 1 | 1.0 |
| Paulista | 3 | 1.0 |
| Olinda | 3 | 0.8 |
| Camaragibe | 1 | 0.7 |
a) The municipalities of the Metropolitan Region of Recife not listed in this table did not record cases of Guillain-Barre syndrome in the period studied.
Four cases were lost at the interview stage, resulting in 40 investigated cases, notified from 23/12/2014 to 19/06/2015, with a peak in epidemiological week 14(April) (Figure 2). Seventeen of them were classified as GBS cases with probable prior DCZ infection and one was confirmed, which corresponded to an estimated prevalence of 45%. Of these 18 cases, 10 were in the 40-59 year age range, with a median age of 44 years (range from 14 to 62 years) and equal distribution between the sexes (Table 2).
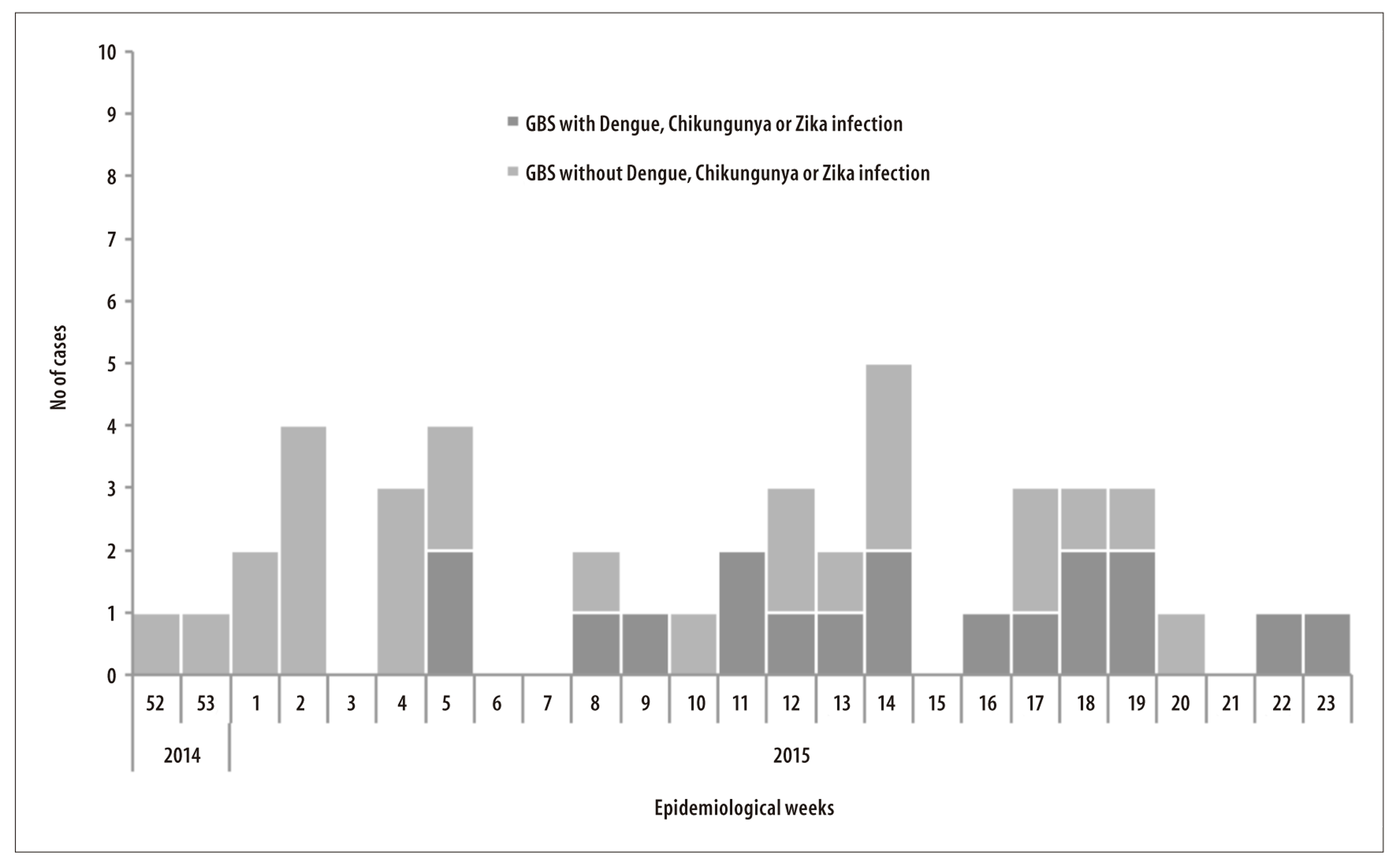
Figure 2 - Cases of Guillain-Barre Syndrome (GBS) and GBS with prior infection with dengue, chikungunya or Zika, according to the epidemiological week of initial neurological symptoms, Metropolitan region of Recife, 2015
Table 2 - Confirmed and probable cases of Guillain-Barré syndrome with prior Zika virus infection (N=18), according to sociodemographic characteristics and possible etiologies, Metropolitan region of Recife, 2015
| Variables | N |
|---|---|
| Sex | |
| Male | 9 |
| Female | 9 |
| Age group (in years) | |
| 10-19 | 2 |
| 20-29 | 3 |
| 30-39 | 2 |
| 40-49 | 5 |
| 50-59 | 5 |
| 60-69 | 1 |
| Municipality of residence | |
| Recife | 10 |
| Jaboatão dos Guararapes | 4 |
| Abreu e Lima | 1 |
| Ipojuca | 1 |
| Olinda | 1 |
| Paulista | 1 |
| Vaccine | |
| Influenza (Campaign) | 1 |
| Human papilloma virus (Campaign) | 1 |
| Diphtheria and tetanus, MMR and hepatitis B | 1 |
| Surgery | - |
| Negative results to other etiologies | |
| Cytomegalovirus | 2 |
| Epstein-Barr virus | 2 |
| Results for dengue, chikungunya or Zika | |
| Dengue: no-reagent IgM serology | 1 |
| Zika: RT-PCRa detectable | 1 |
a) RT-PCR: Reverse transcription polymerase chain reaction, or reverse transcriptase reaction, followed by polymerase chain reaction.
The symptom of infectious disease prior to the GBS most reported by individuals was rash (15/18). None of them reported an episode of diarrhea in the period studied. The median time between prior viral infection and the onset of neurological symptoms was 8 days (range of 0 to 35 days). Among the most frequently reported neurological signs and symptoms, muscle weakness was highlighted (17/18), followed by paresthesia (16/18), hyporeflexia and/or areflexia (15/18), progressing upward (Figure 3).

Figure 3 - Confirmed and probable cases of Guillain-Barré Syndrome with Zika virus infection (N=18), according to signs and symptoms of infection and neurological signs, Metropolitan region of Recife, 2015
As to diagnosis, 16/18 cases had a record of the result of CSF analysis and showed a median of 127 mg protein/dL (range 35 to 245 mg/dL) and two cells/mm³ (range 0 to 17 cells/mm3), with a median time of collection of eight days (range of 3 to 39 days) after the onset of neurological symptoms.
Most of the cases (13/18) had an imaging exam and/or electro neurophysiological exam, in which 12 of these cases were submitted to computed tomography and 9/13 were referred to magnetic resonance imaging. They had normal results and neurological injury was discarded. Only 4/13 cases underwent electroneuromyography. All presented results compatible with GBS, according to clinical data registered by the physician in charge in their medical records.
The median length of hospital stay was 15 days (range of 4 to 39 days), 3/18 cases needed mechanical ventilation, and 14/18 were treated with IVIg with a median of 30 vials (range 15 to 40) per case.
Regarding the progression of the 18 GBS cases with probable prior DCZ infection, 1 of them died, 9 showed clinical improvement with reversible or irreversible sequels, and 8 progressed to overall improvement at the time of the hospital discharge.
In the 90-day period prior to neurological symptoms, only 3 of the 18 cases reported were vaccinated for influenza (1/3); HPV (1/3), and diphtheria and tetanus, MMR and hepatitis B (1/3). As to other possible etiologies, data was found in two medical records, with negative results for cytomegalovirus, Epstein-Barr virus and HIV. A search carried out for diagnoses made in the acute phase found that only 2/18 cases had collected samples for viral examination at an appropriate time: (i) a negative IgM serology test result for dengue, (ii) and a positive RT-PCR result for Zika virus, according to analysis performed by the Pernambuco Oswaldo Cruz Institute Foundation (Fiocruz/PE) on a sample of CSF taken four days after the onset of muscle weakness and 23 days after the onset of infection symptoms (Table 2).
Discussion
The study confirmed the occurrence of an outbreak of GBS in Pernambuco in the first half of 2015, concomitant with the introduction of Zika virus in the Northeastern Brazilian states. The majority of cases interviewed in MRR presented evidence of probable dengue or Zika infection. As at the end of this study, there were no records of autochthonous chikungunya transmission in Pernambuco state.
GBS incidence of 1.2 cases per 100,000 inhabitants was within the global recorded range of incidence, according to studies mainly conducted in Europe and in North America,6,7,16 and also studies conducted in Arab countries,8 Asia,17 Caribbean countries and India,3 since the 1960s, although incidence was higher than that found by a study conducted in São Paulo between 1995 and 2002.9
There was no difference in GBS incidence according to sex, despite being slightly more common in men.7 As to age, incidence increased in more advanced age groups, which is to be expected due to the risk of exposure to the infectious agent being greater as age advances.1,4,7 Incidence varied greatly among the different municipalities, what may possibly be related to higher degrees of exposure to infectious agents.3.6
About 2/3 of GBS cases are associated with prior infection; however, the specific agent of infection is unknown in approximately 60% of these cases, showing the difficulty in identifying the organism possibly involved.4,8,18 The prevalence of prior viral infection found in this study was 45%, corroborating the present report, especially when considering the oligosymptomatic and asymptomatic nature of dengue and Zika infections.19
From January to June 2015, GBS with probable or confirmed prior DCZ infection affected men and women equally; and older age groups, similarly to the incidence rates found among the GBS hospitalizations in MRR mentioned earlier. The cases presented the characteristic GBS symptoms, which may be related to the case definitions used.
In the same way as the study conducted in a hospital in São Paulo between 1995 and 2002 we found that 20% of cases required mechanical ventilation while, other studies report a percentage of around 25% of individuals with GBS developing respiratory failure.1,9,18 Deaths accounted for 6% of cases. This proportion is compatible with expected mortality: between 5 to 25% of cases. In our study, the proportion of deaths found remained close to the lower limit of the expected range, possibly due to the fact that most cases were admitted to a hospital specialized in neurology and received treatment with IVIg.6.20
The status of clinical infection found coincided with the signs and symptoms most frequently found in cases of DCZ infection, reinforcing the hypothesis of the participation of these diseases as triggering factors of GBS.21
As for the other etiologies investigated, all studied individuals who received vaccines showed signs and symptoms of a possible prior infection with dengue or Zika. Causal associations between vaccine and GBS may be suggested in individual cases, by temporal relation, but in the absence of previous infections.6,7,22
The strongest association found so far is between GBS and prior C. jejuni infection. This bacterium has been found in between 25 and 50% of adult individuals and with greater frequency in Asian countries, followed by cytomegalovirus, Epstein-Barr virus and M. pneumoniae, according to studies conducted in Europe, Taiwan and Japan between the years 1990 and 2000. The fact that both individuals tested for cytomegalovirus, Epstein-Barr virus and HIV had negative results discarded the hypothesis of prior infection with these viruses. No records were found for C. jejuni in any of the individuals. Moreover, none of them made reference to episodes of diarrhea prior to neurological manifestation.17,23,24
There have been records of neurological manifestations secondary to dengue infection since the 1960s in epidemic regions, such as Asia, India and Latin America, with presentation of several neurological manifestations, including GBS.14,25,26 However, no increase in GBS hospitalizations in Pernambuco was detected in epidemic years of dengue preceding the period studied.
In the literature, the first GBS cases associated with Zika infection were recorded in French Polynesia in 2013, and in Puerto Rico in 2016. In both regions, prior Zika virus infection was laboratory-confirmed, with rash being the symptom most reported by 81% and 53% of patients, respectively; the median time between infection and neurological symptoms in both events referred to was six days (range: 4-10 days) and 5 days (range: 0-17 days), respectively.13.27 These main results are very similar to the clinical characteristics found in the MRR individuals described, e.g. rash reported by 83% of patients and the median of eight days between infection and GBS symptoms.
Among the limitations of our study, we highlight case underestimation. SIH/SUS is a health information system used only by public health services or hospitals linked to Brazilian Unified Health System, others GBS patients may have been hospitalized using another ICD-10 code and diagnosed later. The Horus system keeps no record of hospital, residential address or telephone number of case recorded on it, thus hindering their being located. The incompleteness of medical records may have hindered the more accurate description of the neurological syndrome and infections prior to GBS.
In terms of etiologic diagnosis, there was no opportunity to collect samples and send them for the laboratory tests as planned, nor did we find samples collected in advance and stored that could be used for the diagnosis of prior infection. In most cases, no results from earlier diagnoses were found.
After this, and other research in Brazil,28 a national protocol was established for the investigation of GBS cases following DCZ infection. An analytical study was alsos developed with the objective of investigating the possible factors associated with the occurrence of GBS in Pernambuco.
A recommendation was made to SES/PE to include DCZ serologies in etiological examination of GBS cases, as per the protocol defined by the Brazilian Ministry of Health, and to establish a state-wide referral flow for electroneuromyography for GBS case confirmation.29
This study stems from a series of investigations carried out by the Brazilian Ministry of Health in support of State and Municipal Health Departments, with the aim of clarifying the consequences of Zika virus infection. After the confirmation of occurrence of the GBS outbreak in Pernambuco, between January and June of 2015, the description of the cases investigated in MRR has proved to be a plausible hypothesis that these cases described, as well as the increase in GBS incidence, were associated with the prior Zika infection: (I) there were no records of increased incidence of GBS in other epidemic years of dengue observed in Pernambuco; (ii) as at the end of this study, there had been no record of chikungunya transmission in MRR; (iii) rash was the most frequently reported sign among cases with evidence of DCZ infection, and is compatible with Zika virus disease and similar to the findings in the Dominican Republic and Puerto Rico;13.27 and (iv) the laboratory confirmation of Zika in a case of Guillain-Barré syndrome in MRR.
Referências
1. Sejvar JJ, Kohl KS, Gidudu J, Amato A, Bakshi N, Baxter R, et al. Guillain-Barré syndrome and Fisher syndrome: Case definitions and guidelines for collection, analysis, and presentation of immunization safety data. Vaccine. 2011 Jan;29(3):599-612. [ Links ]
2. Dimachkie MM, Barohn RJ. Guillain-Barré syndrome and variants. Neurol Clin. 2013 May;31(2):491-510. [ Links ]
3. Van den Berg B, Walgaard C, Drenthen J, Fokke C, Jacobs BC, van Doorn PA. Guillain-Barré syndrome: pathogenesis, diagnosis, treatment and prognosis. Nat Rev Neurol. 2014 Aug;10(8):469-82. [ Links ]
4. Vellozzi C, Iqbal S, Broder K. Guillain-Barré syndrome, influenza, and influenza vaccination: the epidemiologic evidence. Clin Infect Dis. 2014 Apr;58(8):1149-55. [ Links ]
5. Eldar AH, Chapman J. Guillain Barré syndrome and other immune mediated neuropathies: diagnosis and classification. Autoimmun Rev. 2014 Apr-May;13(4-5):525-30. [ Links ]
6. Willison HJ, Jacobs BC, van Doorn PA. Guillain-Barré syndrome. Lancet. 2016 Aug;388(10045):717-27. [ Links ]
7. Sejvar JJ, Baughman AL, Wise M, Morgan OW. Population incidence of Guillain-Barré syndrome: a systematic review and meta-analysis. Neuroepidemiology. 2011;36(2):123-33. [ Links ]
8. Benamer HT, Bredan A. Guillain-Barré syndrome in Arab countries: a systematic review. J Neurol Sci. 2014 Aug;343(1-2):221-3. [ Links ]
9. Rocha MSG, Brucki SMD, Carvalho AAS, Lima UWP. Epidemiologic features of Guillain-Barré syndrome in São Paulo, Brazil. Arq Neuro-Psiquiatr. 2004 Mar;62(1):33-7. [ Links ]
10. Goodfellow JA, Willison HJ. Guillain-Barré syndrome: a century of progress. Nat Rev Neurol. 2016 Dec;12(12):723-31. [ Links ]
11. Lim JY, Lim Y-H, Choi E-H. Acute-onset chronic inflammatory demyelinating polyneuropathy in hantavirus and hepatitis B virus coinfection. Medicine (Baltimore). 2016 Dec;95(49):e5580. [ Links ]
12. Oehler E, Fournier E, Leparc-Goffart I, Larre P, Cubizolle S, Sookhareea C, et al. Increase in cases of Guillain-Barré syndrome during a Chikungunya outbreak, French Polynesia, 2014 to 2015. Euro Surveill. 2015 Dec;20(48):30079. [ Links ]
13. Cao-Lormeau V-M, Blake A, Mons S, Lastère S, Roche C, Vanhomwegen J, et al. Guillain-Barré Syndrome outbreak associated with Zika virus infection in French Polynesia: a case-control study. Lancet. 2016 Apr;387(10027):1531-9. [ Links ]
14. Carod-Artal FJ, Wichmann O, Farrar J, Gascón J. Neurological complications of dengue virus infection. Lancet Neurol. 2013 Sep;12(9):906-19. [ Links ]
15. Ministério da Saúde (BR). Departamento de Informática do Sistema Único de Saúde - Datasus. Informações de Saúde - TABNET. Projeção populacional intercensitária, 2012 [Internet]. 2015 [ citado 2016 fev 15]. Disponível em: Disponível em: http://www2.datasus.gov.br/DATASUS/index.php?area=0206 [ Links ]
16. Webb AJS, Brain SAE, Wood R, Rinaldi S, Turner MR. Seasonal variation in Guillain-Barré syndrome: a systematic review, meta-analysis and Oxfordshire cohort study. J Neurol Neurosurg Psychiatry. 2015 Nov;86(11):1196-201. [ Links ]
17. Huang WC, Lu CL, Chen SC. A 15-Year nationwide epidemiological analysis of Guillain-Barré Syndrome in Taiwan. Neuroepidemiology. 2015 Jun:44(4):249-54. [ Links ]
18. Hardy TA, Blum S, McCombe PA, Reddel SW. Guillain-Barré Syndrome: Modern Theories of Etiology. Curr Allergy Asthma Rep. 2011 Jun;11(3):197-204. [ Links ]
19. Petersen EE, Staples JE, Meaney-Delman D, Fischer M, Ellington SR, Callaghan WM, et al. Interim Guidelines for Pregnant Women During a Zika Virus Outbreak - United States, 2016. MMWR Morb Mortal Wkly Rep. 2016 Jan;65(2):30-3. [ Links ]
20. Vucic S, Kiernan MC, Cornblath DR. Guillain-Barré syndrome: an update. J Clin Neurosci. 2009 Jun;16(6):733-41. [ Links ]
21. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Coordenação-Geral de Desenvolvimento da Epidemiologia em Serviços. Guia de Vigilânia em Saúde [Internet]. Brasília: Ministério da Saúde; 2016 [citado 2018 mar 12]. 773 p. Disponível em: Disponível em: http://www.rio.rj.gov.br/dlstatic/10112/6385405/4170293/GUIADEVS2016.pdf [ Links ]
22. Israeli E, Agmon-Levin N, Blank M, Chapman J, Shoenfeld Y. Guillain-Barré Syndrome - a classical autoimmune disease triggered by infection or vaccination. Clin Rev Allergy Immunol. 2012 Apr;42(2):121-30. [ Links ]
23. Jacobs BC, Rothbarth PH, van der Meche FGA, Herbrink P, Schmitz PI, de Klerk MA, et al. The spectrum of antecedent infections in Guillain-Barré syndrome: a case-control study. Neurology. 1998 Oct;51(4):1110-5. [ Links ]
24. Islam Z, Jacobs BC, van Belkum A, Mohammad QD, Islam MB, Herbrink P, et al. Axonal variant of Guillain-Barré syndrome associated with Campylobacter infection in Bangladesh. Neurology. 2010 Feb;74(7):581-7. [ Links ]
25. Gonçalves E. Acute inflammatory demyelinating polyradiculoneuropathy (Guillain-Barré syndrome) following dengue fever. Rev Inst Med Trop Sao Paulo. 2011 Jul-Aug;53(4):223-5. [ Links ]
26. Chaudhary SC, Mohanty D, Sonkar SK, Gupta DK, Gupta A. Unusual manifestation of dengue fever. BMJ Case Rep. 2011 Jun;2011. [ Links ]
27. Dirlikov E, Major CG, Mayshack M, Medina N, Matos D, Ryff KR, et al. Guillain-Barré Syndrome During Ongoing Zika Virus Transmission - Puerto Rico, January 1 - july 31, 2016. MMWR Morb Mortal Wkly Rep. 2016 Sep;65(34):910-4. [ Links ]
28. Malta JMAS, Vargas A, Leite PL, Percio J, Coelho GE, Ferraro AHA, et al. Síndrome de Guillain-Barré e outras manifestações neurológicas possivelmente relacionadas à infecção pelo vírus Zika em municípios da Bahia, 2015. Epidemiol Serv Saúde. 2017 jan-mar;26(1):9-18. [ Links ]
29. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Protocolo de vigilância dos casos de manifestações neurológicas com histórico de infecção viral prévia [Internet]. Brasília: Ministério da Saúde; 2015 [citado 2017 set 14]. 9 p. Disponível em: Disponível em: http://production.latec.ufms.br/modulos/zika/res/u4/protocolo-vigilancia-manifestacoes-neurologicas-zika-virus.pdf [ Links ]
Received: April 25, 2017; Accepted: February 19, 2018











 texto em
texto em 


