Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.27 no.3 Brasília set. 2018 Epub 18-Jul-2018
http://dx.doi.org/10.5123/s1679-49742018000300003
ORIGINAL ARTICLE
Chikungunya surveillance in Brazil: challenges in the context of Public Health*
1Secretaria de Estado da Saúde de Goiás, Laboratório Estadual de Saúde Pública Dr. Giovanni Cysneiros, Goiânia, GO, Brasil
2Universidade Federal de Goiás, Faculdade de Educação, Goiânia, GO, Brasil
3Universidade Federal de Goiás, Instituto de Ciências Biológicas, Goiânia, GO, Brasil
4Universidade Federal de Goiás, Instituto de Patologia Tropical e Saúde Pública, Goiânia, GO, Brasil
5Ministério da Saúde, Secretaria de Vigilância em Saúde, Brasília, DF, Brasil
Objective:
to describe the challenges in implementing the chikungunya surveillance and prevention system in Brazil.
Methods:
this was a descriptive study of suspected cases of the disease based on records held on the Notifiable Diseases Information System (Sinan) for the period 2014-2016.
Results:
more than 100,000 probable chikungunya cases were notified in Brazil in this period, with the largest concentration in the Northeast states (83.3% between 2014 and 2015; 91.0% in 2016); Sinan provided an excellent opportunity for closing records of cases occurring between 2014 and 2015 (85%) and high completeness of obligatory variables.
Conclusion:
given the imminence of the introduction of chikungunya in Brazil in 2014, advance public health preparation took place in order to minimize its effects on society; implementation of the surveillance system improved collection of information regarding the disease, however many challenges can be seen in practice, in view of increasing case incidence. This requires greater handling capacity in this sector.
Keywords: Chikungunya; Public Health Surveillance; Epidemiology
Introduction
Chikungunya virus (CHIKV) is an RNA genome Alphavirus belonging to the Togaviridae family. It is a vector-borne disease transmitted by Aedes genus mosquito bites. It was isolated for the first time in mid 1953 in an outbreak in Tanzania.1 Since then, the virus has been responsible for outbreaks and epidemics of great magnitude on the Asian and African continents, as observed on Réunion Island in 2004 when a third of the population was infected, resulting in more than 244,000 cases and 203 deaths attributed to the disease it causes.2
In the initial stages of symptomatic disease (acute phase), fever and arthralgia are reported, although these symptoms may persist for up to three months, characterizing the subacute phase. With regard to the chronic phase, which can be disabling for years, chikungunya is a public health problem in countries with tropical climates favorable to the maintenance and widespread dispersion of the Aedes aegypti and Aedes albopictus vectors in their regions.3,4
The magnitude of infection in the Americas was highlighted in December 2013, after the Pan American Health Organization (PAHO) published an epidemiological alert on the evidence of the first autochthonous cases of the disease. By the 52nd epidemiological week (EW) of the following year (2014), 1,071,696 suspected cases of the disease were reported in more than 30 countries on the American continent, such as Mexico, El Salvador, Nicaragua, the Dominican Republic, Puerto Rico, Colombia, Venezuela, Brazil, Suriname, among others, with 169 deaths attributed to chikungunya.5,6
In Brazil, the first autochthonous cases were identified in the Oiapoque, state of Amapá (Northern Brazil), and Feira de Santana, state of Bahia (Northeast Brazil), in September 2014.7 Following genetic analysis of the virus two lineages were detected: the Asian lineage, found in the municipality of Oiapoque; and the Eastern, Central and Southern Africa (ECSA) lineage detected in city of Feira de Santana. No A226V mutation was identified in reported cases. If there had been, CHIKV could also have infected Aedes albopictus mosquitoes.8
Brazilian reality favored the introduction and the spread of the virus. Aedes aegypti can be found in more than 4,000 municipalities, and Aedes albopictus in 3,285.9,10 In addition to this there is high vector dispersion, a large flow of people as well as the population's susceptibility to infection.11
With the introduction of the disease in Brazil, a scenario arose marked by the coexistence of arboviruses. The increase of chikungunya autochthonous cases records of severe cases and deaths, resulted in greater demand for health care services and increasing need of financial and human resources, in order to minimize its effects on society.12
The current panorama of the disease requires updated, reliable and accurate data to be obtained in order to warn about the occurrence of outbreaks and related epidemics. The knowledge of the main actions arising from the implementation of the national chikungunya surveillance and prevention system may indicate if it is working efficiently, in addition to informing, through the results obtained, health planning and decision-making.13
The objective of this study was to describe the challenges in implementing the chikungunya surveillance and prevention system in Brazil and to evaluate the disease in the context of Brazilian Public Health.
Methods
This study was conducted in Brazil, covering all 26 states and the Federal District, starting on 1st January 2014, the same year in which chikungunya was introduced into the country, on 13th October 2016. By means of descriptive case series analysis, the disease was studied according to Health Surveillance aspects within the context of Public Health.13
Information obtained from the online version of the Notifiable Diseases Information System (Sinan-Net) enabled this study to be carried out (<http://portalsinan.saude.gov.br/sinan-net>). The database data in dbf format, was made available to the researcher in reply to an official letter sent to the General Coordination of National Programs for the Prevention and Control of Malaria and Aedes Transmitted Diseases (CGPNCMD), located within the Department of Communicable Diseases Surveillance (DEVIT) of the Health Surveillance Secretariat/Ministry of Health (SVS/MS). Surveillance uses reports containing information about health complaints, drawn up based on notification forms from all over Brazil. Periodically, the data contained on these forms are input to Sinan, the main source of information related diseases in Brazil. Any health establishment, whether public or private, must inform the occurrence of suspected cases to Municipal and State Health Departments which in turn informthe Ministry of Health.14
In order to describe and analyze the quantitative attributes (timeliness and representativeness) and qualitative attributes (data quality) of the health surveillance system, we adopted the guidelines of the Centers for Disease Control and Prevention (CDC) of the United States of America (USA), as per the Updated Guidelines for Evaluating Public Health Surveillance Systems.15
The 'timeliness' attribute indicates the period between the stages of the surveillance system, i.e., the interval of time - in days - between the various stages of the process of notification: date of notification and date of onset of symptoms; date of onset of symptoms and date of notification form input on Sinan; date of notification and date of case closure. According to the Sinan-Net manual, the chikungunya surveillance system will be considered to be operating in a timely manner when 70% of cases are closed on the system in up to 60 days.16 Suspected cases should be included on the system in up to seven days.13
The ‘representativeness’ attribute of the system considers the distribution of the event in the population (per person, place and time), so that all cases are characterized according to their proportion by municipality and by notifying health center.
The quality of the data was represented by the 'completeness' attribute, through the evaluation of key variables (ethnicity/skin color, schooling, district of residence, case confirmation criterion and evolution) and compulsory variables (age, sex, final classification, municipality of residence and date of first symptoms) held on the notification forms. The extent to which the variables were filled in on the forms was rated as valid and not valid (data field unknown, not filled in or blank). The completeness attribute was classified as: excellent (90% or more of records compliant), good (70-89% of the records completed), regular (50-69% of the records completed) or poor (0-49% of the records completed), according to parameters for the evaluation of data completeness and consistency used by a study of dengue in Brazil.17
Analysis of socioeconomic variables was also included in the study (sex, age, ethnicity/skin color and schooling) for all notified cases, given the relevance of this knowledge about the population. However, only confirmed cases of the disease were considered for the purpose of describing the attributes.
The descriptive statistics and the distribution of absolute and relative frequency of chikungunya cases were analyzed using SPSS Statistics for Windows version 23.0® and Microsoft Excel 2013®. QGIS version 2.18 was used to prepare maps. All cases notified on the system having compliant information met the inclusion criterion; however, cases with diverging notified information (date of first symptoms equal to date of birth; and duplicated cases) were excluded.
The research project was submitted to the Ethics Research Committee of the Federal University of Goiás (UFG) and received its approval: Opinion no. 1,186.731 on 17 August 2015, in accordance with National Health Council (CNS) Resolution No 466 of 12 December 2012.
Results
A total of 47,830 chikungunya cases was reported in Brazil in 2014 and 2015 (EW 1 of 2014 [29/12/2013 to 04/01/2014], EW 52 of 2015 [27/12/2015 to 02/01/2016]). There was a significantly greater number of notifications in 2015 (43,253), when compared to the previous period. However, a significant increase in reported cases was found with effect from 2016 in the period covered by our study (EW 1 [03/01/2016 to 09/01/2016] to EW 41 [09/10/2016 to 10/13/2016]), namely: 133,404 probable cases recorded on Sinan-Net, with 63,810 cases confirmed either by laboratory criteria (based on laboratory diagnosis) or by clinical-epidemiological criteria. In 2014 and 2015 there was a greater proportion of notified cases in the states of the Northeastern region (39,851 notified cases), accounting for 83.3% of the Sinan records of chikungunya.14,033 of these cases (29.3%) were confirmed according to the two criteria mentioned above.
This higher prevalence was attributed to the high number of cases reported in the state of Bahia, where the highest proportions occurred in the microregions of Feira de Santana (70.1%), Serrinha (74.2%) and Salvador (84.7%). However, with effect from 2016, most suspected cases of chikungunya reported on the surveillance system occurred in the states of Ceará and Pernambuco (Figure 1). The period covering 2016 also stands because there was the largest proportion of confirmed cases - 63,810 cases (47.8%), in relation to previous years - 14,033 cases (29.3%), taking all the states of the Federation, including the Fernando de Noronha Archipelago (Figure 2).

a) Sinan-Net: Notifiable Diseases Information System.
Figure 1 - Probable chikungunya cases reported on Sinan-Neta, Brasil, 2016
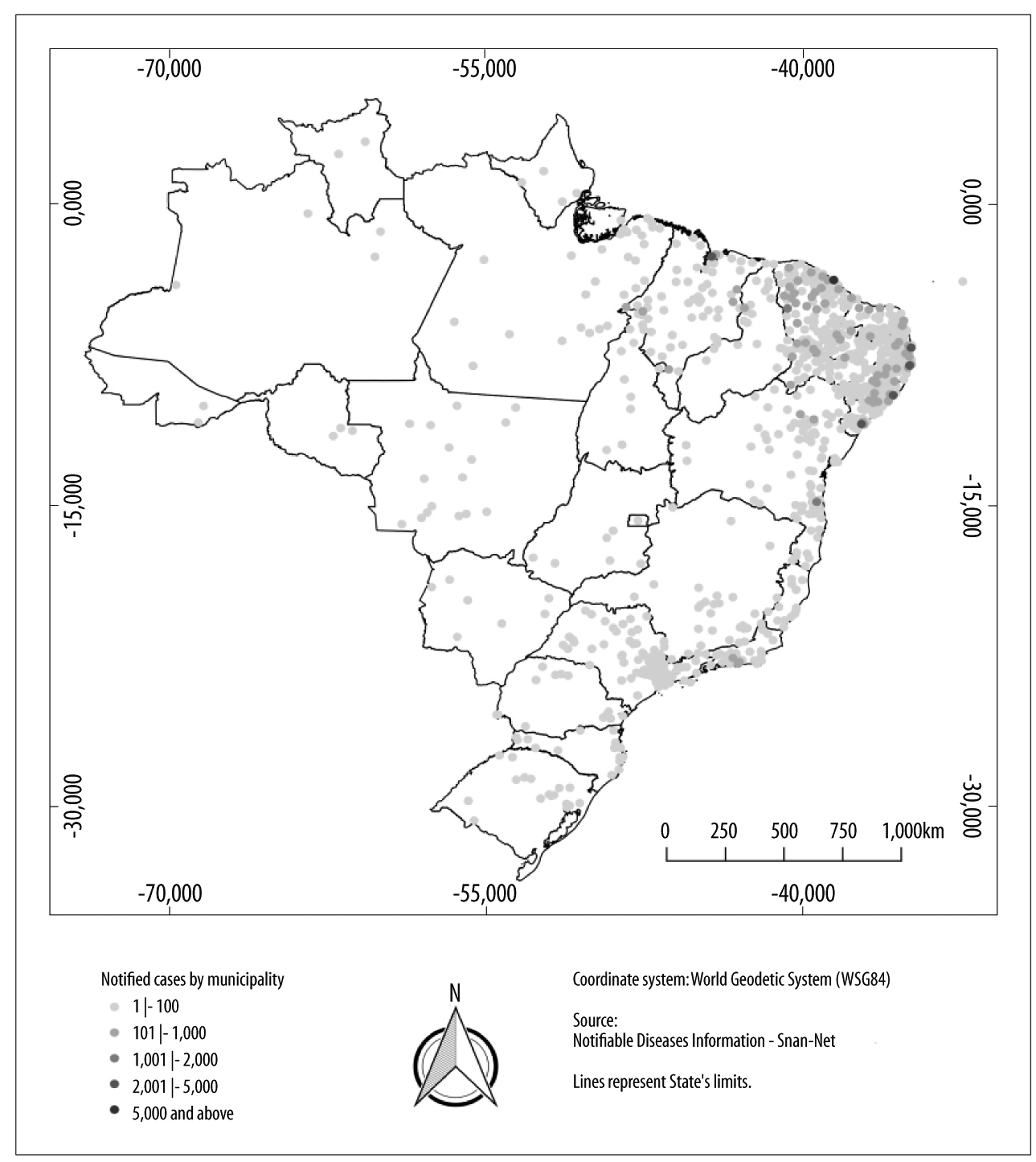
a) Sinan-Net online Notifiable Diseases Information System.
Figure 2 - Confirmed chikungunya case distribution reported on Sinan-Neta, Brazil, 2016
In relation to the epidemiological profile of the population studied, the predominant age range found in the case notifications was 20-39 years (35.8%), while 29.7% were aged 40-59 years and 21.7% of cases reported were aged under 19 years old. There was a higher proportion of females (65.2%), as well as a higher proportion of people referring brown skin /color (47.9%) among the notified cases as a whole.
In relation to symptomatic cases, the following were identified as the main acute symptoms of chikungunya infection: fever (90.2%), arthralgia (76.3%), headache (66.1%) and myalgia (65.1%) (Table 1).
Table 1 - Frequently reported clinical symptoms of acute chikungunya infection
| Symptoms | Frequency | Percentage (%) |
|---|---|---|
| Fever | 57. 569 | 90.2 |
| Arthralgia | 48,661 | 76.3 |
| Headache | 42,176 | 66.1 |
| Myalgia | 41,556 | 65.1 |
| Back pain | 17,297 | 27.1 |
| Rash | 17,026 | 26.7 |
| Nausea | 14,426 | 22.6 |
| Arthritis | 12,093 | 18.9 |
| Vomiting | 10,933 | 17.1 |
| Retro-orbital pain | 6,247 | 9.8 |
| Petechiae | 4,057 | 6.4 |
| Conjunctivitis | 3,595 | 5.6 |
| Leukopenia | 1,062 | 1.7 |
| Proof of the positive loop | 530 | 0.8 |
| Total cases | 63,810 |
Source: Notifiable Diseases Information System - Net (Sinan-Net) - epidemiological weeks 1-41/2016 (data subject to change).
With regard to the 'timeliness' attribute, it was found that more than 85% of cases were closed in up to 60 days in 2014 and 2015, and 72% in 2016, thus surpassing the national goal (70%). However, only 68.7% of cases were reported in up to seven days in 2014 and 2015, and 76.4% in 2016. In the first two years of study there was a lower proportion of notifications made in up to five days (60.4%), although this increased in the last year of the study (69.6%). With regard to the input of notification forms on the system, 54.8% of them were found to have been input up to 15 days in 2014 and -2015; in 2016, however, only 26.6% were input in a timely manner. When calculating this attribute (timeliness) and the data completeness attribute, only 13,995 confirmed cases of chikungunya for the years 2014-2015 were taken into consideration.
There was representativeness of records of suspected cases in all units of the Federation, covering 18% of Brazilian municipalities. It was also found that more than 3,000 health centers notified chikungunya on Sinan. The system showed excellent completeness (Table 2). The completeness of the compulsory variables was considered excellent (90% or more compliant records), in contrast to greater variability in the completion of the key variables.
Table 2 - Percentage of selected variables on chikungunya containing valid information on the Notifiable Diseases Information System, Brazil, 2014-2015
| Variables | Completeness (%) | Classification |
|---|---|---|
| Ethnicity/skin color | 10,063 (71.9) | Good |
| Age Group | 13,991 (99.9) | Excellent |
| Sex | 13,992 (99.9) | Excellent |
| Schooling | 6,837 (48.8) | Poor |
| District of residence | 7,977 (56.9) | Regular |
| Final classification | 10,019 (71.5) | Good |
| Confirmation criteria | 13,954 (99.7) | Excellent |
| Case evolution | 12,831 (91.6) | Excellent |
| Municipality of residence | 13,891 (99.2) | Excellent |
| First symptoms date | 13,988 (99.9) | Excellent |
| Total | 13,995 (100.0) |
Source: Notifiable Diseases Information System - Net (Sinan epidemiological weeks 01/2014-52/2015 (data subject to change).
Note:
Range of completeness used for classification:
Excellent (90% or more compliant records);
Good (70-89% of the records completed);
Regular (50-69% of the records completed); and
Poor (0-49% of the records completed).
Discussion
In Brazil, the first cases of chikungunya were reported in the states of Bahia and Amapá; however, within a short period cases of the disease were reported in all states of the Federation, with a high number of suspected cases between 2014 and 2016. The spread of the disease, although it did happen, was much lower than expected, when compared to other countries, especially in Central America and the Caribbean.9
In spite of being recently deployed, it was found that the chikungunya surveillance and prevention system was representative (in the study period) when compared to the records on the dengue surveillance and prevention system. This is because the system already used for dengue was quickly adapted to receive notifications of chikungunya, thus contributing to the quality of the ‘representativeness’ attribute.17
The important role played by Health Surveillance in controlling chikungunya was reflected in the excellent timeliness of case closure in the first two years (2014 and 2015). However, timeliness of notification is the main component from the point of view of adopting control measures at the right time. This attribute (notification timeliness) did not demonstrate the level of quality expected: more than 30% of notifications held on the system were made seven days after the onset of symptoms. The shortage of trained professionals to perform surveillance activities, coupled with the lack of financial resources, deserves attention because of the resulting difficulty in performing the diagnosis of the disease and its adequate clinical management, which may lead, indirectly, to increased cases among the population.18,19 As highlighted by Cerroni & Carmo (2015), the loss of timeliness in relation to case notification and closure is prejudicial to ensuring knowledge of the real epidemiological status of diseases needed for Public Health decision-making.20
With effect from 2016, with the release of Sinan online version 3.0, it became possible to included chikungunya data. Because of this, reported and investigated dengue and chikungunya cases began to be recorded on a single form, thus integrating and improving at the same time data collection for both of these diseases (<portalsinan.saude.gov.br/images/documentos/Agravos/Dengue/Ficha_DENGCHIK_FINAL.pdf; http://sinan.saude.gov.br/sinan>).
As regards data quality from the perspective of completeness, in spite of it being compulsory to fill in the variables, nevertheless a small percentage of fields were incomplete. Although filling in the 'schooling' variable is not directly related to system efficiency in detecting outbreaks and epidemics, nevertheless it is relevant because it is a socioeconomic variable.21 As also demonstrated by Almeida et al. (2012), it is outstanding in this study that epidemiological data are more complete than data on socioeconomic context. In particular, the ethnicity/skin color variable was found to be less complete than all the other variables.22
In contrast to the low frequency of severe cases of chikungunya reported at the beginning of the transmission period, there was a greater proportion of deaths from the disease with effect from 2016, with 201 confirmed cases.23 This reinforces the need for timely recognition of severe cases with complications and, consequently, the possibility of interfering in the determinants in order to limit the occurrence of deaths. Studies have identified important complications (cardiovascular and respiratory alterations, meningoencephalitis, nervous system alterations) related to serious cases in infants (>9 days of age).24,25
Although our study found that cases were notified in all age groups, the highest proportion of suspected cases (35.8%) was identified in individuals aged 20-39 years, thus coinciding with the economically active age range. Cases were predominant among females (65.2%), and this distribution has also been found in other countries with records of chikungunya outbreaks and epidemics.26,27
The limitations identified in this study are the same as those inherent to secondary data. The high proportion of notification form fields left blank and/or incomplete, as well as lack of relevant information, may have influenced the data on the characterization of the clinical and epidemiological profile of chikungunya in Brazil. This limitation extends to professionals who analyse the situation of the disease directly, hindering the knowledge needed for the management of patients’ epidemiological and clinical profile and, consequently, the adoption of more effective public policies on chikungunya control and prevention, as well as more effective care of cases.
Despite these limitations, the study analyses the surveillance system right at the beginning of chikungunya casey detection in Brazil. Initially, active surveillance actions were predominant (Decree No 205 of 17 February 2017). However, as more cases were identified, there was a migration to passive surveillance strategies.11,28 As a consequence, throughout the national territory, Public Health authorities being notified of suspected cases with transmission stood out as the main form of chikungunya surveillance.11
Due to chikungunya being in the spotlight, including on the international scenario, and its relevance for Public Health, its investigation was extended to negative dengue cases.19 Undoubtedly, deploying pecific serological diagnosis for chikungunya seems to be a challenge commonly faced by the sector, either because of the unavailability of specific tests, or because of it being insufficient to cover the entire population. This situation of absence of diagnostic methods for the purposes of investigation using the medical records of patients with suspected acute febrile illness was found in an outbreak in Yemen in 2010 and 2011.29 On this issue, Gibney et al (2011)26 reported that proper diagnosis is critical for minimizing the risk of introduction of CHIKV in the USA, despite having noted its low availability in that country’s laboratories.
Based on the experience gained in the management of dengue cases and the challenges for the care of patients, the Brazilian Ministry of Health provided guidelines for health professionals about the diagnosis and clinical management of chikungunya. These were published in the guide 'Chikungunya: clinical management'.4 It is worth noting that this is an updated version of the document, which not only contributes to the best solution of cases but is also useful for avoiding the occurrence of severe cases and deaths.
While many countries have experienced chikungunya outbreaks and epidemics,18,19,25 Brazil, even with high incidence of dengue with effect from 2010, had low CHIKV transmission scenarios. Moreover, with effect from 2015, with the identification of autochthonous Zika virus circulation, Brazil has had to reckon with the possibility of the occurrence of simultaneous outbreaks and epidemics caused by all three viruses.30 This new scenario made it necessary to make progress with the surveillance system for these diseases and to adapt Sinan, the objectives of which are to input and disseminate notifiable disease data at all three levels of government, in real time, and thus provide quick and complete information for health analysis and decision-making in health.20 In this context, ongoing evaluations of strategies are crucial for a better understanding of trends and adequacy of the health care system, with the aim of reducing the magnitude of the epidemics and, above all, the deaths caused by these diseases.
REFERENCES
1. Robinson MC. An epidemic of virus disease in Southern Province, Tanganyika Territory, in 1952-53. I. Clinical features. Trans R Soc Trop Med Hyg. 1955 Jan;49(1): 28-32 [ Links ]
2. Lo Presti A, Cella E, Angeletti S, Ciccozzi M. Molecular epidemiology, evolution and phylogeny of Chikungunya virus: an updating review. Infect Genet Evol. 2016 Jul;41:270-8. [ Links ]
3. Campbell LP, Luther C, Moo-Llanes D, Ramsey JM, Danis-Lozano R, Peterson AT. Climate change influences on global distributions of dengue and chikungunya virus vectors. Philos Trans R Soc B Biol Sci. 2015 Apr;370(1665):20140135. [ Links ]
4. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Chikungunya: manejo clínico [Internet]. Brasília: Ministério da Saúde; 2017 [citado 2017 jul 10]. 65 p. Disponível em: Disponível em: http://bvsms.saude.gov.br/publicacoes/chikungunya_manejo_clinico_1ed.pdf . [ Links ]
5. Pan American Health Organization. World Health Organization. Number of reported cases of chikungunya fever in the Americas, by country or territory 2013-2014 (to week noted) [Internet]. 2014 [cited 2017 Nov 2]. Available in: Available in: http://www.paho.org/hq/index.php?option=com_docman&task=doc_download&Itemid=270&gid=28697&lang=en . [ Links ]
6. Pan American Health Organization. World Health Organization. Epidemiological alert: chikungunya fever [Internet]. 2013 [cited 2017 Nov 2]. Available in: Available in: http://www.paho.org/hq/index.php?option=com_docman&task=doc_view&Itemid=270&gid=23806&lang=en . [ Links ]
7. Nunes MR, Faria NR, Vasconcelos JM, Golding N, Kraemer MU, Oliveira LF, et al. Emergence and potential for spread of Chikungunya virus in Brazil. BMC Med. 2015 Apr;13:102. [ Links ]
8. Madariaga M, Ticona E, Resurrecion C. Chikungunya: bending over the Americas and the rest of the word. Braz J Infect Dis. 2016 Jan-Feb;20(01):91-8. [ Links ]
9. Carvalho RG, Oliveira RL, Braga IA. Updating the geographical distribution and frequency of Aedes albopictus in Brazil with remarks regarding its range in the Americas. Mem Inst Oswaldo Cruz. 2014 Sep;109(6);787-96. [ Links ]
10. Chaves TSS, Pellini ACG, Mascheretti M, Jahnel MT, Ribeiro AF, Rodrigues SG, et al. Travelers as sentinels for Chikungunya fever, Brazil. Emerg Infect Dis. 2012 Mar;18(3):529-30. [ Links ]
11. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento das Doenças Transmissíveis. Plano de contingência para a febre Chikungunya [Internet]. Brasília: Ministério da Saúde; 2014 [citado 2018 mar 5]. 48 p. Disponível em: Disponível em: http://bvsms.saude.gov.br/bvs/publicacoes/plano_contingencia_nacional_febre_chikungunya.pdf [ Links ]
12. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 19, 2017. Bol Epidemiológico [Internet]. 2017 [citado 2017 jul 11]; 48(16):1-10. Disponível em: Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2017/maio/25/Monitoramento-dos-casos-de-dengue-febre-de-chikungunya-e-febre-pelo-virus-Zika-ate-a-Semana-Epidemiologica.pdf . [ Links ]
13. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Coordenação-Geral de Desenvolvimento da Epidemiologia em Serviços. Guia de Vigilância em Saúde [Internet]. Brasília: Ministério da Saúde; 2016 [citado 2017 jul 10]. 773 p. Disponível em: Disponível em: http://bvsms.saude.gov.br/bvs/publicacoes/guia_vigilancia_saude_1ed_atual.pdf . [ Links ]
14. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Sistemas de informação de agravos de notificação - Sinan: normas e rotinas [Internet]. 2. ed. Brasília: Ministério da Saúde; 2007 [citado 2018 mar 5]. 68p. Disponível em: Disponível em: http://bvsms.saude.gov.br/bvs/publicacoes/07_0098_M.pdf [ Links ]
15. Centers for Disease Control and Prevention. Updated guidelines for evaluating public health surveillance systems: recommendations from the guidelines working group. MMWR. 2001 Jul;50(RR13):1-35. [ Links ]
16. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Roteiro para uso do Sinan-Net, análise da qualidade da base de dados e cálculo de indicadores epidemiológicos e operacionais [Internet]. Brasília: Ministério da Saúde; 2008 [citado 2016 jan 27]. Disponível em: Disponível em: http://www1.saude.ba.gov.br/dis/arquivos_pdf/Cadernos%20de%20Analise%20do%20SINAN/Caderno%20de%20analise%20GERAL.pdf . [ Links ]
17. Barbosa JR, Barrado JCS, Zara ALSA, Siqueira Júnior JB. Avaliação da qualidade dos dados, valor preditivo positivo, oportunidade e representatividade do sistema de vigilância epidemiológica da dengue no Brasil, 2005 a 2009. Epidemiol Serv Saúde. 2015 jan-mar;24(1):49-58. [ Links ]
18. Randrianasolo L, Raoelina Y, Ratsitorahina M, Ravolomanana L, Andriamandimby S, Heraud JM, et al. Sentinel surveillance system for early outbreak detection in Madagascar. BMC Public Health. 2010 Jan;10:31. [ Links ]
19. Ho K, Ang LW, Tan BH, Tang CS, Ooi PL, James L, et al. Epidemiology and control of chikungunya fever in Singapore. J Infect. 2011 Apr;62(4):263-70. [ Links ]
20. Cerroni MP, Carmo EH. Magnitude das doenças de notificação compulsória e avaliação dos indicadores de vigilância epidemiológica em municípios da linha de fronteira do Brasil, 2007 a 2009. Epidemiol Serv Saúde. 2015 out-dez;24(4):617-28. [ Links ]
21. Felix JD, Zandonade E, Amorim MHC, Castro DS. Avaliação da completude das variáveis epidemiológicas do Sistema de Informação sobre Mortalidade em mulheres com óbitos por câncer de mama na Região Sudeste - Brasil (1998 a 2007). Ciênc Saúde Coletiva. 2012 Apr;17(4):945-53. [ Links ]
22. Almeida MVS, Amorim MHC, Thule LCS, Zandonade E. Avaliação da qualidade dos dados do sistema de informação do câncer do colo do útero em Vitória-ES, Brasil. Rev Bras Cancerol. 2012 jul-set;58(3):427-33. [ Links ]
23. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 33, 2017. Bol Epidemiológico: [Internet]. 2017 [citado 2017 set 01];48(27): 1-13. Disponível em: Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2017/agosto/29/2017-026-Monitoramento-dos-casos-de-dengue-febre-de-chikungunya-e-febre-pelo-virus-Zika-ate-a-Semana-Epidemiologica-33-de-2017.pdf . [ Links ]
24. Renault P, Solet JL, Sissoko D, Balleydier E, Larrieu S, Filleul L, et al. A major epidemic of Chikungunya virus infection on Réunion Island, France, 2005-2006. Am J Trop Med Hyg. 2007 Oct;77(4):727-31. [ Links ]
25. Sissoko D, Malvy D, Giry C, Delmas G, Paquet C, Gabrie P, et al. Outbreak of Chikungunya fever in Mayotte, Comoros archipelago, 2005-2006. Trans R Soc Trop Med Hyg. 2008 Aug;102(8):780-6. [ Links ]
26. Gibney KB, Fischer M, Prince HE, Kramer LD, St George K, Kosoy OL, et al. Chikungunya fever in the United States: a fifteen year review of cases. Clin Infect Dis. 2011 Mar;52(5):e121-26. [ Links ]
27. Napoli C, Salcuni P, Pompa MG, Declich S, Rizzo C. Estimated imported infections of Chikungunya and dengue in Italy, 2008 to 2011. J Travel Med. 2012 Sep-Oct;19(5):294-7. [ Links ]
28. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento das Doenças Transmissíveis. Preparação e resposta à introdução do vírus Chikungunya no Brasil: baseado no livro Preparación y respuesta ante la eventual introducción del virus Chikungunya en las Américas [Internet]. Brasília: Ministério da Saúde; 2014 [citado 2017 jul 6]. 100 p. Disponível em: Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2014/setembro/09/preparacao-e-resposta-virus-chikungunya-web.pdf . [ Links ]
29. Malik MR, Mnzava A, Mohareb E, Zayed A, AlKohlani A, Thabet AA, et al. Chikungunya outbreak in Al-Hudaydah, Yemen, 2011: epidemiological characterization and key lessons learned for early detection and control. J Epidemiol Glob Health. 2014 Sep;4(3):203-11. [ Links ]
30. Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária. Recomendações técnicas ao Sistema Nacional de Vigilância Sanitária para colaborar no combate ao Aedes aegypti e prevenção e controle da Dengue, Chikungunya e infecção pelo vírus Zika [Internet]. Brasília: Agência Nacional de Vigilância Sanitária; 2016 [citado 2018 mar 5]. 32 p. Disponível em: Disponível em: http://aprav.com.br/wp-content/uploads/2016/04/Cartilha-ANVISA-aedes-1.pdf [ Links ]
*The authors received financial support from the Foundation for the Support of Research of Goiás State, Process No. 201510267000931. The financial support had no influence on the study design, data collection and analysis, decision to publish or preparation of the manuscript. The article is derived from the Master's thesis entitled 'Health Surveillance of fever Chikungunya in Brazil in the context of the Brazilian Unified Health System', presented by Nayara Messias da Silva to the Postgraduate Collective Health Program of Federal University of Goiás, in 2016.
Received: April 04, 2017; Accepted: February 18, 2018











 texto em
texto em 


