Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.27 no.3 Brasília set. 2018 Epub 05-Out-2018
http://dx.doi.org/10.5123/s1679-49742018000300015
ORIGINAL ARTICLE
Factors associated with cure when treating tuberculosis in the state of Rio de Janeiro, Brazil, 2011-2014
1Fundação Oswaldo Cruz, Escola Nacional de Saúde Pública Sergio Arouca, Rio de Janeiro, RJ, Brasil
2Universidade Federal do Espírito Santo, Programa de Pós-Graduação em Saúde Coletiva, Vitória, ES, Brasil
Objective:
to analyze factors associated with cure when treating tuberculosis (TB) in the state of Rio de Janeiro, Brazil, from 2011 to 2014.
Methods:
this was a cross-sectional study with data from the Notifiable Diseases Information System (SINAN); logistic regression was used for data analysis.
Results:
of the 57,142 cases reported, 49,691 (86.96%) were included in the study; schooling ≥13 years (odds ratio (OR) 3.48; 95%CI 2.74;4.41) and residing in rural areas (OR 2.27; 95%CI 1.38; 3.73) were factors associated with a higher chance of cure; being male (OR 0.71; 95%CI 0.66;0.76) and brown skin color/race (OR 0.73; 95%CI 0.68;0.78) stood out as factors associated with less chance of cure.
Conclusion:
social and economic determinants were identified that influence the treatment outcome, evidencing the need for social support and guaranteed access to health services.
Keywords: Tuberculosis; Public Policy; Social Vulnerability; Cross-Sectional Studies
Introduction
Tuberculosis (TB) is considered one of the most long-standing infectious diseases affecting mankind, with TB deaths having been recorded for at least five thousand years.¹ In the 21st century, TB is still a public health problem worldwide, due to wide geographical dispersion, the existence of multidrug-resistant cases and co-infection with the human immunodeficiency virus (HIV).2 9.6 million cases and 1.5 million deaths from TB were estimated for the year 2014. The estimated incidence of TB-HIV co-infection for the same year was 1.2 million cases, with 400,000 deaths.3
Tuberculosis is related to poverty, social exclusion and deprivation, this being a very common scenario in developing countries where TB continues at considerable levels,4 given the intense social inequality, increasing poverty, uncontrolled urban and population growth which, in turn, are reflected in social inequalities in health.5 In 2016, Brazil was in 18th place among the 22 countries accounting for 80% of TB cases worldwide.6
A year later, in 2015, there were 63,189 new cases of the disease in Brazil, representing an incidence coefficient of 30.9 cases per 100,000 inhabitants.6 In the state of Rio de Janeiro in 2015, the TB incidence rate was 54.5 cases per 100,000 inhabitants, and the TB mortality rate was 5.1 per 100,000 inhabitants.6 However, this distribution is not homogeneous.7 The highest concentration of cases is observed in poorer regions and affects, in particular, some of the most vulnerable groups, characterized by individual and collective susceptibility resulting from the social and economic situation in which they find themselves.4,8
In 2012, a model was proposed capable of linking the social determinants of TB, taking account of the multicausality of disease and three dimensions of this vulnerability: (i) individual; (ii) programmatic or institutional; and (iii) social or contextual.9 Looking at vulnerability from these three dimensions enables different forms of analysis, ranging from proximal to distal determinants, and from the individual level to the institutional and collective level.10
In Brazil, studies 11-13 have demonstrated how these factors affect the outcome of TB treatment, in particular comorbidities (alcoholism and HIV/AIDS), education level, sex, age, income, occupation and family support. The state of Rio de Janeiro is marked by social contrasts: in 2012, it was considered to be the seventh most unequal Federative Unit in Brazil as measured by the Gini coefficient. This reflects the high levels of both income and poverty in the region.8,14 Studies on factors associated with the outcome of TB treatment in Rio de Janeiro can be useful for identifying more vulnerable groups.
The objective of this study was to analyze factors associated with cure when treating tuberculosis (TB) in the state of Rio de Janeiro, Brazil, from 2011 to 2014.
Methods
This was a cross-sectional study using data on reported tuberculosis (TB) cases held on the Notifiable Diseases Information System (SINAN).
Rio de Janeiro state covers an area of 43,781,566km² containing 92 municipalities. In 2015 it had an estimated population of 16,550,024 inhabitants15 distributed between nine health microregions.
The dependent variable was the outcome of treatment, with cure being considered to be a favorable outcome. Unfavorable outcomes were considered to be: abandonment, primary abandonment (non-adherence within the first 30 days), death from TB, drug-resistant tuberculosis (TBDR) and treatment failure.
The exposure variables analyzed were:
- sex (male; female);
- age (by age groups: under 20; 20 to 39; 40 to 60; over 60);
- ethnicity/skin color (white; black; brown; other [yellow and indigenous]);
- education level (in years of schooling: illiterate; 1 to 4; 5 to 8, 9 to 12; 13 or more);
- area of residence (urban, rural);
- occupational disease (yes; no);
- institutionalized (no; prison; asylum; orphanage; psychiatric hospital; another);
- HIV/AIDS (yes; no);
- alcoholism (yes; no);
- smoking (yes; no);
- diabetes (yes; no);
- other comorbidities (yes; no);
- type of notification (new case; recurrence; return after dropout; unknown, referral);
- supervised treatment (yes; no);
- clinical form (pulmonary, extra-pulmonary, pulmonary + extra-pulmonary);
- X-ray (normal; suspect);
- smear microscopy, 1st sample (negative; positive); and
- sputum culture (negative; positive)
All cases recorded as being finalized were considered to meet the inclusion criterion. All cases that were not conclusively finalized were excluded (information missing, unknown or with change of diagnosis). It is of note that cases with a recorded outcome did not necessarily receive treatment; for example, the outcome ‘death from TB’ includes both treated cases and those not treated initially.
The initial database, formed by the eligible cases, was processed by using filters, with the goal of eliminating duplicates. To this end, was used the Statistical Package for the Social Sciences, version 20.0 (SPSS Inc, Chicago, IL, USA) was used.
The first step taken was to checkthe completeness of two new variables included on the SINAN system: (i) street people (yes; no); and (ii) beneficiary of government income transfer program (yes; no).A descriptive analysis of the variables of interest for the study was then performed (absolute number and frequency). In the crude analysis, we used the Pearson chi-squared test. In the analysis adjusted by non-conditional multiple logistic regression,9 we used a conceptual hierarchical model.
The hierarchical model used was that proposed by Maciel in 2012.9 In this model, the social determinants of TB are classified by level of vulnerability. In the present study, level 1 considered the sex, education level, ethnicity/skin color and age variables; level 2, area of residence, institutionalization, disease related to work; and level 3, smoking, HIV/AIDS, diabetes, and other comorbidities, mental illness and alcoholism. Level 4 was comprised of the analysis of the type of admission, X-ray, sputum culture, clinical form and directly observed treatment.
The Pearson chi-squared test was used to identify the variables significantly associated with the outcome (p<0.05) in the crude analysis, initially input to the non-conditional multiple logistic regression hierarchical model. As the variables lost significance (p>0.05) at each level of regression - from the most distal to the most proximal - they were removed from the model. Odds ratio (OR) and confidence intervals of 95% (95%CI) were calculated, with the help of Stata software version 14. Adjustment of the final model was done using the Hosmer-Lemeshow goodness of fit test.16
The study project was submitted to the Ethics Committee of the Sergio Arouca National School of Public Health, Oswaldo Cruz Foundation (Fiocruz), and approved on 16 September 2016 by Opinion No. 1727131.
Results
The initial database was comprised of 57,142 notifications. After the use of filters to eliminate duplicates and delete cases with change of diagnosis from the database, 49,691 cases remained (Figure 1). 8,416 cases with no outcome were discarded. The final study sample was comprised of 41,279 cases of tuberculosis, divided between 32,607 having cure as their outcome (79.0%) and 8,672 with an unfavorable outcome (21%).
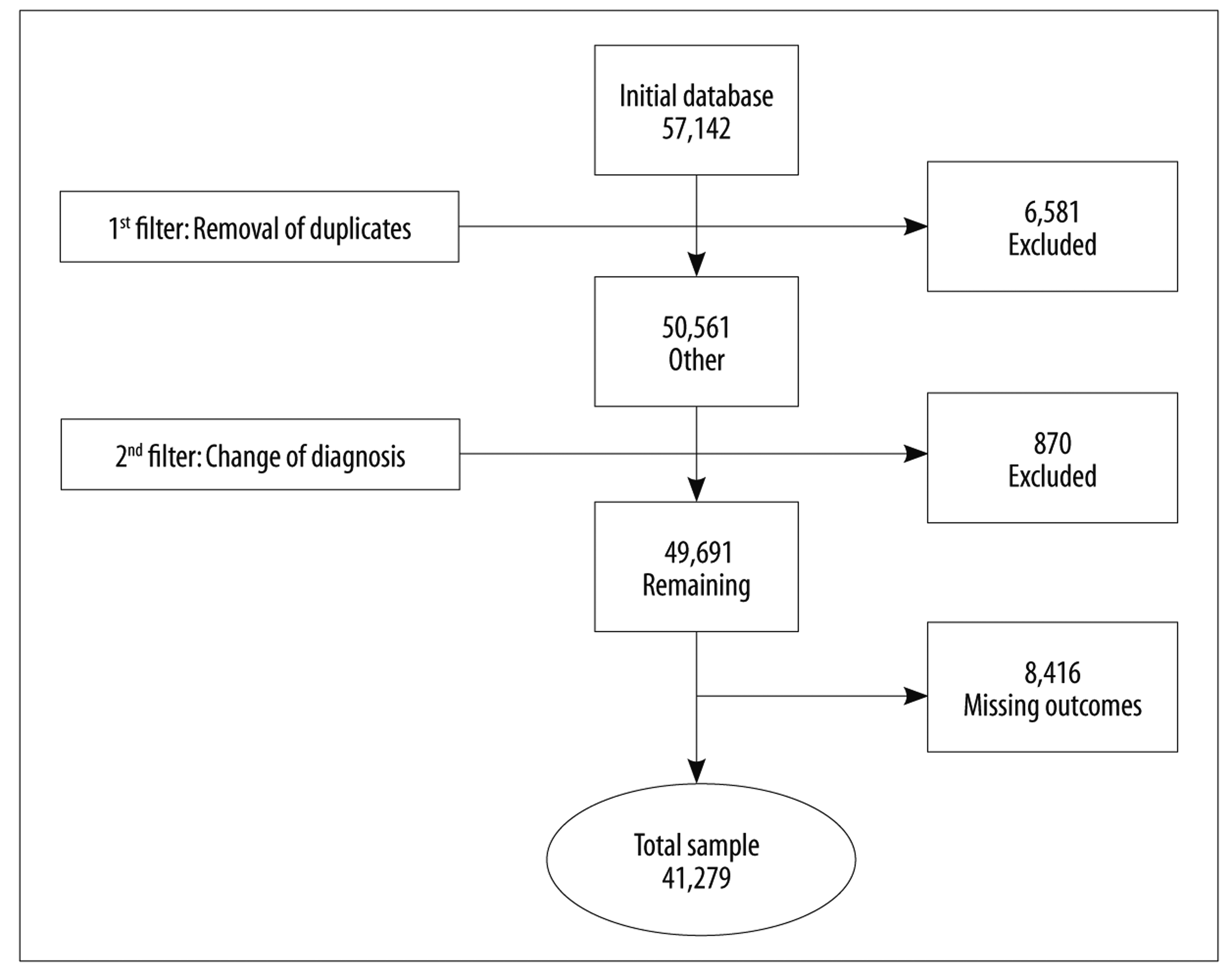
Figure 1 - Flow diagram of the sample of tuberculosis cases taken from the Notifiable Diseases Information System, Rio de Janeiro state, 2011-2014
Regarding the incompleteness of the two new variables included on the SINAN-TB system, regarding street people, 44,302 (89.2%) records were found with no information and 2,353 (4.7%) filled in as unknown; and with regard to the beneficiaries of the government Income Transfer Program, 44,422 (89.4%) records with no information and 3,776 (7.6%) with unknown information were found (data not shown in Tables or Figures).
Table 1 describes the characteristics of the cases: the majority were males(65.0%), aged between 40 and 59 years (34.5%), with 5 to 8 years of schooling (35.1%), diagnosed with the pulmonary form of the disease (85.0%), recorded as new cases (82.8%), with suspected X-ray (95.3%) and positive sputum smear microscopy (69.6%).
Table 1 - Characteristics of notified cases of tuberculosis, Rio de Janeiro state, 2011-2014
| Variables | n | % |
|---|---|---|
| Sex (n=41,275) | ||
| Male | 26,634 | 65.0 |
| Female | 14,641 | 35.0 |
| Age in years (n=41,279) | ||
| <20 | 4,059 | 9.9 |
| 20-39 | 18,180 | 44.0 |
| 40-59 | 14,244 | 34.5 |
| ≥60 | 4,796 | 11.6 |
| Race/skin color (N=37,430) | ||
| White | 14,506 | 39.0 |
| Black | 8,111 | 22.0 |
| Brown | 14,438 | 38.0 |
| Other | 375 | 1.0 |
| Schooling, in years of study (n=28,656) | ||
| Illiterate | 696 | 2.4 |
| 1-4 | 7,366 | 25.7 |
| 5-8 | 10,066 | 35.1 |
| 9-12 | 8,135 | 28.4 |
| ≥13 | 2,396 | 8.4 |
| HIV/AIDS (n=27,901) | ||
| Yes | 3,184 | 13.6 |
| No | 24,717 | 86.4 |
| Alcoholism (n=34,156) | ||
| Yes | 4,597 | 13.5 |
| No | 29,559 | 86.5 |
| Diabetes (n=33,751) | ||
| Yes | 2,745 | 8.1 |
| No | 31,006 | 91.9 |
| Smoker (n=3,541) | ||
| Yes | 332 | 9.4 |
| No | 3,209 | 90.6 |
| Occupational disease (n=27,717) | ||
| Yes | 738 | 2.7 |
| No | 26,979 | 97.3 |
| Clinical form (n=41,279) | ||
| Pulmonary | 35,067 | 85.0 |
| Extra-pulmonary | 4,879 | 11.8 |
| Pulmonary + extrapulmonary | 1,333 | 3.2 |
| Place of residence (n=39,002) | ||
| Rural | 424 | 1.1 |
| Urban | 38,578 | 98.9 |
| Institutionalized (n=36,709) | ||
| No | 34,153 | 93.0 |
| Prison | 1,290 | 3.5 |
| Nursing home | 59 | 0.2 |
| Orphanage | 72 | 0.2 |
| Psychiatric hospital | 89 | 0.2 |
| Other | 1,046 | 2.9 |
| Type of admission (n=41,278) | ||
| New case | 34,192 | 82.8 |
| Recurrence | 2,794 | 6.8 |
| Return after abandonment | 2,422 | 5.9 |
| Unknown | 204 | 0.5 |
| Referral | 1,666 | 4.0 |
| X-rays (n=37,685) | ||
| Suspected | 35,901 | 95.3 |
| Normal | 1,784 | 4.7 |
| Sputum smear microscopy, 1st sample (n=31,175) | ||
| Positive | 21,708 | 69.6 |
| Negative | 9,467 | 30.4 |
| Sputum culture (n=6,056) | ||
| Positive | 3,808 | 62.9 |
| Negative | 2,248 | 37.1 |
| Supervised treatment (n=36,902) | ||
| Yes | 16,047 | 44.1 |
| No | 20,345 | 55.9 |
Table 2 presents the results of the crude and adjusted analyses of the hierarchical logistic regression model. On level 1, the male sex was associated with a lower chance of a favorable outcome (OR 0.71; 95%CI 0.66;0.76), when compared to females. We observed a greater chance of a favorable outcome among those with 9-12 years of schooling (OR 2.04; 95%CI 1.68;2.47) and 13 or more years of schooling (OR 3.48; 95%CI 2.74;4.41), compared to the illiterate. Black race/skin color (OR 0.62; 95%CI 0.57;0.67) and brown skin color (OR 0.73; 95%CI 0.68;0.78) were also associated with the lowest chance of cure, compared to those of white race/skin color; the same association was observed in the 20-39 age range (OR 0.65; 95%CI 0.58;0.73).
Table 2 - Distribution and factors associated with tuberculosis outcome according to the hierarchical model of dimensions of vulnerability,a Rio de Janeiro State, 2011-2014
| Characteristics | Unfavorable outcomeb | Favorable outcomec | ORd crude (95%CI)e | P Valuef | ORd adjusted (95%CI)e | P-Value adjustedg |
|---|---|---|---|---|---|---|
| n (%) | n (%) | |||||
| LEVEL 1 | ||||||
| Sex | ||||||
| Female | 2,439 (16.6) | 12,202 (83.4) | 1.00 | <0.001 | 1.00 | <0.001 |
| Male | 6,230 (24.0) | 20,404 (76.0) | 0.65 (0.62;0.69) | 0.71 (0.66;0.76) | ||
| Education level (in years of schooling) | ||||||
| Illiterate | 167 (24.0) | 529 (76.0) | 1.00 | <0.001 | 1.00 | <0.001 |
| 1-4 | 1,729 (23.0) | 5,637 (77.0) | 0.28 (0.22;0.34) | 1.08 (0.90;1.31) | ||
| 5-8 | 2,363 (23.0) | 7,703 (77.0) | 0.28 (0.24;0.33) | 1.11 (0.92;1.33) | ||
| 9-12 | 1,161 (14 .0) | 6,974 (86.0) | 0.28 (0.24;0.33) | 2.04 (1.68;2.47) | ||
| ≥13 | 192 (8.0) | 2,204 (92.0) | 0.52 (0.44;0.61) | 3.48 (2.74;4.41) | ||
| Race/skin color | ||||||
| White | 2,251 (16.0) | 12,255 (84.0) | 1.00 | <0.001 | 1.00 | <0.001 |
| Black | 2,095 (26.0) | 6,016 (74.0) | 1.06 (0.80;1.40) | 0.62 (0.57;0.67) | ||
| Brown | 3,308 (23.0) | 11,130 (77.0) | 0.56 (0.42;0.74) | 0.73 (0.68;0.78) | ||
| Other | 61 (16.0) | 314 (84.0) | 0.65 (0.49;0.86) | 1.03 (0.73;1.46) | ||
| Age (in years) | ||||||
| <20 | 670 (17.0) | 3,389 (83.0) | 1.00 | <0.001 | 1.00 | <0.001 |
| 20-39 | 4,321 (24.0) | 13,859 (76.0) | 1.15 (1.03;1.28) | 0.65 (0.58;0.73) | ||
| 40-59 | 2,792 (20.0) | 11,452 (80) | 0.73 (0.67;0.79) | 0.97 (0.86;1.10) | ||
| ≥60 | 889 (19.0) | 3,907 (81) | 0.93 (0.86;1.01) | 1.08 (0.93;1.25) | ||
| LEVEL 2 | ||||||
| Place of residence | ||||||
| Urban | 8,139 (21.0) | 30,439 (79.0) | 1.00 | <0.001 | 1.00 | <0.001 |
| Rural | 46 (11.0) | 378 (89.0) | 0.45 (0.33;0.62) | 2.27 (1.38;3.73) | ||
| Institutionalized | ||||||
| No | 6,895 (20.0) | 27,258 (80.0) | 1.00 | <0.001 | 1.00 | <0.001 |
| Prison | 273 (21.0) | 1,017 (79.0) | 2.36 (2.08;2.68) | 1.04 (0.80;1.34) | ||
| Nursing home | 25 (42.0) | 34 (58.0) | 2.23 (1.86;2.68) | 0.35 (0.15;0.83) | ||
| Orphanage | 19 (26.0) | 53 (74.0) | 0.81 (0.48;1.38) | 0.67 (0.29;1.55) | ||
| Psychiatric hospital | 8 (9.0) | 81 (91.0) | 1.66 (0.97;2.85) | 3.50 (0.82;14.9) | ||
| Other | 391 (37.0) | 655 (63.0) | 6.04 (2.89;12.63) | 0.40 (0.32;0.49) | ||
| Occupational Disease | ||||||
| No | 5,536 (21.0) | 21,443 (79.0) | 1.00 | 0.001 | 1.00 | 0.116 |
| Yes | 116 (16.0) | 622 (84.0) | 1.38 (1.13;1.69) | 1.24 (0.95;1.63) | ||
| LEVEL 3 | ||||||
| HIV/AIDS | ||||||
| No | 4,547 (18.0) | 20,170 (82.0) | 1.00 | <0.001 | 1.00 | 0.260 |
| Yes | 1,290 (41.0) | 1,894 (59.0) | 0.33 (0.30;0.36) | 0.73 (0.42;1.27) | ||
| Alcoholism | ||||||
| No | 5,339 (18.0) | 24,220 (82.0) | 1.00 | <0.001 | 1.00 | 0.123 |
| Yes | 1,539 (33.0) | 3,058 (67.0) | 0.44 (0.41;0.50) | 0.65 (0.38;1.12) | ||
| Smoking | ||||||
| No | 684 (21.0) | 2,525 (79.0) | 1.00 | <0.001 | 1.00 | 0.081 |
| Yes | 134 (40.0) | 198 (60.0) | 0.40 (0.32;0.51) | 0.50 (0.23;1.09) | ||
| Diabetes | ||||||
| No | 6,324 (20.0) | 24,682 (80.0) | 1.00 | <0.001 | 1.00 | 0.111 |
| Yes | 473 (17.0) | 2,272 (83.0) | 1.23 (1.11;1.36) | 0.56 (0.28;1.14) | ||
| Other comorbidities | ||||||
| No | 3,960 (18.0) | 17,532 (82.0) | 1.00 | <0.001 | 1.00 | 0.895 |
| Yes | 1,511 (30.0) | 3,447 (70.0) | 0.51 (0.48; 0.55) | 1.04 (0.56;1.96) | ||
| LEVEL 4 | ||||||
| Notification Type | ||||||
| New case | 5,939 (17.0) | 28,253 (83.0) | 1.00 | <0.001 | 1.00 | <0.001 |
| Recurrence | 733 (26.0) | 2,061 (74.0) | 1.87 (1.67;2.09) | 0.55 (0.43;0.72) | ||
| Return after abandonment | 1,390 (57.0) | 1,032 (43.0) | 1.10 (0.96;1.27) | 0.21 (0.16;0.28) | ||
| Unknown | 139 (68.0) | 65 (32.0) | 0.29 (0.25;0.33) | 0.69 (0.03;13.9) | ||
| Referral | 470 (28.0) | 1,196 (72.0) | 0.18 (0.13;0.25) | 0.41 (0.28;0.60) | ||
| Supervised Treatment | ||||||
| No | 4,227 (21.0) | 16,118 (79.0) | 1.00 | 0.001 | 1.00 | 0.307 |
| Yes | 3,110 (19.0) | 12,937 (81.0) | 1.09 (1.04;1.15) | 0.91 (0.75;1.10) | ||
| Clinical form | ||||||
| Pulmonary | 7,650 (22.0) | 27,417 (78.0) | 1.00 | <0.001 | 1.00 | 0.624 |
| Extra-pulmonary | 642 (13.0) | 4,237 (87.0) | 1.43 (1.26;1.61) | 1.22 (0.67;2.23) | ||
| Pulmonary + extrapulmonary | 380 (29.0) | 953 (71.0) | 2.63 (2.28;3.04) | 0.84 (0.51;1.39) | ||
| X-rays | ||||||
| Normal | 257 (14.0) | 1,527 (86.0) | 1.00 | <0.001 | 1.00 | 0.207 |
| Suspected | 7,454 (21.0) | 28,447 (79.0) | 0.64 (0.56;0.73) | 1.51 (0.80;2.84) | ||
| Sputum smear microscopy (1st) | ||||||
| Negative | 1,762 (19.0) | 7,705 (81.0) | 1.00 | <0.001 | 1.00 | 0.005 |
| Positive | 4,731 (22.0) | 16,977 (78.0) | 0.82 (0.77;0.87) | 0.73 (0.59;0.91) | ||
| Sputum culture | ||||||
| Negative | 367 (16.0) | 1,881 (84.0) | 1.00 | <0.001 | 1.00 | <0.001 |
| Positive | 1,073 (28.0) | 2,735 (72.0) | 0.50 (0.44;0.57) | 0.58 (0.47;0.71) | ||
a) Level 1: Sex + race/color + age + schooling.
Level 2: Level 1 (significant) + place of residence + institutionalized + occupational disease.
Level 3: Level 2 (significant) + HIV/AIDS + alcoholism + smoking + diabetes + mental illness + comorbities.
Level 4: Level 3 (significant) + admission type + sputum smear (1st sample) + sputum culture + x-ray + supervised treatment.
b) Unfavorable outcomes were considered to be: abandonment, primary abandonment, death from TB, drug-resistant tuberculosis and failure.
c) Favorable outcome: Cure.
d) OR: odds ratio.
e) 95%CI: 95% confidence interval.
f) p-value in the non-adjusted analysis.
g) P-value in the adjusted analysis.
On level 2, living in the rural zone was associated with a greater chance of an unfavorable outcome (OR 2.27; 95%CI 1.38;3.73). However, being institutionalized in an asylum (OR 0.35; 95%CI 0.15;0.83) and in other establishments except prisons, asylums, orphanages and psychiatric hospitals (OR 0.40; 95%CI 0.32;0.49) had lower chances of a favorable outcome, when compared to not being institutionalized.
None of the variables considered on level 3 (smoking, alcoholism and comorbidities) showed a statistically significant association with the outcome.
On level 4, all types of admission, when compared to new cases, showed a lower chance of cure: recurrence (OR 0.55; 95%CI 0.43;0.72), return after dropout (OR 0.21; 95%CI 0.16;0.28) and referral (OR 0.41; 95%CI 0.28;0.60). Similarly, there was an association with positive sputum smear microscopy (OR 0.73; 95%CI 0.59;0.91) and with positive sputum culture (OR 0.58; 95%CI 0.47;0.71), both when compared to the negative results (Table 2). The final model showed a good fit, as indicated by the model adjustment test: chi-squared test value of 7.03 and p-value equal to 0.533.
Discussion
The results found in this study enabled identification of factors associated with the outcome of tuberculosis treatment in the state of Rio de Janeiro between 2011 and 2014. Being of the male sex, having low schooling, having black or brown skin color, being a young adult, living in an urban area, being institutionalized, having a suspect chest X-ray result, having positive 1st sample smear and positive sputum culture results, were factors associated with lower chance of cure. These data demonstrate the relationship between tuberculosis and precarious living conditions.17
Being of the male sex was associated with a lower chance of curing tuberculosis. This corroborates the findings of a retrospective cohort study conducted in Cuiabá, MT, between 1998 and 2000, when predictive factors for the abandonment of TB treatment were analyzed.18 A similar result was found in a case-control study performed in Manaus, AM, and Fortaleza, CE, between 2006 and 2008 as to factors associated with abandonment of TB treatment in Primary Health Care services, according to which individuals of the male sex had a 1.78 greater chance of abandoning treatment, when compared to those of the female sex.19 This difference according to sex may be explained by the fact of lower demand for health services by men:20 women are believed to be more concerned about their health than men and as such they tend to use these services with greater frequency.20 Furthermore, the priorities of health service programmatic activities are directed towards maternal and child health, such as family planning, clinical gynecological and obstetric care, the post delivery period and the menopause, among other health care issues.
Low schooling was associated with a lower chance of cure. This reflects an adverse economic and social situation, which contributes to making individuals susceptible to illness and an unfavorable treatment outcome.19 Low schooling was also associated with treatment dropout in a study conducted in the municipality of Porto Alegre, RS.21
In the present study, the 20-39 age range also showed a lower chance of a favorable outcome. This finding is in keeping with a study on factors associated with TB treatment abandonment in Londrina, PR, in 2006, which found that being a young adult was a risk factor for treatment dropout.22 Individuals in this age group comprise the largest portion of alcohol users, smokers and illicit drug users, especially in large urban centers.23-25 The contribution of these factors to TB treatment failure has already been demonstrated in other studies. The same reasoning can be applied to the condition of residing in urban areas, where the incidence of TB is greater in poor outskirts which are known for their social vulnerability,17 as is the case of the metropolitan region of Rio de Janeiro.
In Recife, PE, between 2005 and 2010, when analyzing factors associated with the outcome of tuberculosis treatment, the crude analysis found association between type of admission and outcome;26 return after abandonment showed 7.2 times more chance of abandoning treatment, while recurrence showed 1.9 times more chance. The study conducted in Recife also found that return after abandonment was associated with a 79% higher chance of an unfavorable outcome, and a 45% higher chance of recurrence. Positive sputum smear microscopy and positive sputum culture were also associated with an unfavorable outcome. It is possible that this finding is associated with more severe clinical TB, related to cavitary disease, which hinders treatment of the disease.27
A limitation of the present study includes issues related to the incompleteness of the variables, including individuals without information on TB treatment outcome and individuals with unknown or inconsistent information for some variables analyzed. These are limitations found in other studies based on SINAN system data and are to be expected when using secondary databases.28,29 However, due to sample size, this study has sufficient statistical power to detect differences between the groups.
Conducted in the state of Rio de Janeiro, this study allowed us to identify factors determining the outcome of TB treatment in a state with the highest burden of TB in the country. The outcome of tuberculosis treatment has proven to be associated with the clinical characteristics of TB and sociodemographic factors, corroborating the theoretical model proposed here of dimensions of vulnerability.9
Rio de Janeiro state is marked by social inequalities, especially in large urban centers. In terms of public policies, measures are needed to ensure universal access to services and, especially, social protection, in order to modify these social and economic determinants that influence the outcome of treating tuberculosis. As to surveillance actions, the importance of the complete filling in of the notification form is emphasized, considering the considerable incompleteness we found, especially in relation to the new SINAN-TB system variable: being a beneficiary of the government income transfer program.
REFERENCES
1. Wilbur AK, Buikstra JE. Patterns of tuberculosis in the Americas - How can modern biomedicine inform the ancient past. Mem Inst Oswaldo Cruz. 2006 Dec;101(2):59-66. [ Links ]
2. San Pedro A, Oliveira RM. Tuberculose e indicadores socioeconômicos: revisão sistemática da literatura. Rev Panam Salud Publica. 2013;33(4):294-301. [ Links ]
3. World Health Organization. Global tuberculosis report 2014 [Internet]. Geneve: World Health Organization; 2014 [citado 2018 Jun 26]. 171 p. Available from: Available from: http://apps.who.int/medicinedocs/documents/s21634en/s21634en.pdf [ Links ]
4. Reis DC, Almeida TAC, Quites HFO, Sampaio MM. Perfil epidemiológico da tuberculose no município de Belo Horizonte (MG), no período de 2002 a 2008. Rev Bras Epidemiol. 2013 set;16(3):592-602. [ Links ]
5. Santos HLN. Fatores associados ao óbito por tuberculose nos pacientes acompanhados no Hospital São Luís. Maranhão [monografia]. São Luís (MA): Universidade Federal do Maranhão; 2014. [ Links ]
6. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Perspectivas brasileiras para o fim da tuberculose como problema de saúde pública. Bol Epidemiológico [Internet]. 2016 [citado 2018 jun 26];47(13):1-15. Disponível em: Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2016/marco/24/2016-009-Tuberculose-001.pdf [ Links ]
7. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. O controle da tuberculose no Brasil: avanços, inovações e desafios. Bol Epidemiológico [Internet]. 2014 [citado 2018 jun 26];44(2):1-13. Disponível em: Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2014/abril/10/Boletim-Tuberculose-2014.pdf [ Links ]
8. Niachiata LYI, Bertolozzi MR, Takahashi RF, Fracolli LA. A utilização do conceito de “vulnerabilidade” pela enfermagem. Rev Latino-Am Enfermagem. 2008 set-out;16(5):923-8. [ Links ]
9. Maciel ELN. Determinantes sociais da tuberculose: elementos para a ação. In: Landim FLP, Catribi AMF, Collares PMC. Promoção da saúde na diversidade humana e nos itinerários terapêuticos. Campinas: Saberes; 2012. [ Links ]
10. Oliveira DLLC, Rossetto M, Hahn G, Maffaccioli R, Almeida CP, Manica S, et al. A utilização da noção de vulnerabilidade na produção do conhecimento sobre tuberculose: revisão integrativa. Rev Gaúcha Enferm. 2015;36(esp):247-53. [ Links ]
11. Silva PF, Moura GS, Caldas AJM. Factors associated with pulmonary TB treatment dropout in Maranhão State, Brazil, from 2001 to 2010. Cad Saúde Pública. 2014 Aug;30(8):1745-54. [ Links ]
12. Albuquerque MFPM, Ximenes RAA, Lucena-Silva N, Souza WV, Dantas AT, Dantas OMS, et al. Factors associated with treatment failure, dropout, and death in a cohort of tuberculosis patients in Recife, Pernambuco State, Brazil. Cad Saúde Pública. 2007 Jul;23(7):1573-82. [ Links ]
13. Paixão LM, Gontijo ED. Profile of notified tuberculosis cases and factors associated with treatment dropout. Rev Saúde Pública. 2007 Apr;41(2):205-13. [ Links ]
14. Serviço Brasileiro de Apoio à Micro e Pequanas Empresas - Sebrae. Renda, pobreza e desigualdade social no estado do Rio de Janeiro. Nota Conjuntural. Observat Sebrae/RJ. 2014 jan;28:1-14. [ Links ]
15. Instituto Brasileiro de Geografia e Estatística. Série histórica e estatística [Internet]. 2016 [citado 2016 set 14]. Disponível em: Disponível em: http://seriesestatisticas.ibge.gov.br/lista_tema.aspx?op=0&de=16&no=6 [ Links ]
16. Lemeshow S, Hosmer DW Jr. A review of goodness of fit statistics for use in the development of logistic regression models. Am J Epidemiol. 1982 Jan;115(1):92-106. [ Links ]
17. Santos TMMG, Nogueira LT, Santos LNM, Costa CM. Caracterização dos casos de tuberculose notificados em um município prioritário do Brasil. Revi Enferm UFPI. 2012 jan-abr;1(1):8-13. [ Links ]
18. Ferreira SMB, Silva AMC, Botelho C. Abandono do tratamento da tuberculose pulmonar em Cuiabá (MT) - Brasil. J Bras Pneumol. 2005 out;31(5):427-35. [ Links ]
19. Braga JU, Pinheiro JS, Matsuda JS, Barreto JAP, Feijão AMM. Fatores associados ao abandono do tratamento nos serviços de atenção básica em dois municípios brasileiros, Manaus e Fortaleza, de 2006 a 2008. Cad Saúde Colet. 2012;20(2):225-33. [ Links ]
20. Pinheiro RS, Viacava F, Travassos C, Britto AS. Gênero, morbidade, acesso e utilização do serviço de saúde no Brasil. Ciênc Saúde Coletiva. 2002;7(4):687-707. [ Links ]
21. Campani STA, Moreira JS, Tietbohel CN. Fatores preditivos para o abandono do tratamento da tuberculose pulmonar preconizado pelo Ministério da Saúde do Brasil na cidade de Porto Alegre (RS). J Bras Pneumol. 2011 nov-dez;37(6):776-82. [ Links ]
22. Giroti SKO, Belei RA, Moreno FN, Silva FS. Perfil dos pacientes com tuberculose e os fatores associados ao abandono do tratamento. Cogitar Enferm. 2010 abr-jun;15(2):271-7. [ Links ]
23. Silva PF, Moura GS, Caldas AJM. Factors associated with pulmonary TB treatment dropout in Maranhão State, Brazil, from 2001 to 2010. Cad Saúde Pública. 2014 Aug;30(8):1745-54. [ Links ]
24. Paixão LMM, Gontijo ED. Perfil de casos de tuberculose notificados e fatores associados ao abandono, Belo Horizonte, MG. Rev Saúde Pública [Internet]. 2007 abr;41(2):205-13. [ Links ]
25. Mascarenhas MDM, Araújo LM, Gomes KRO. Perfil epidemiológico da tuberculose entre casos notificados no Município de Piripiri, Estado do Piauí, Brasil. Epidemiol Serv Saúde. 2005 jan-mar;14:7-14. [ Links ]
26. Silva CCAV, Andrade MS, Cardoso MD. Fatores associados ao abandono do tratamento da tuberculose em indivíduos acompanhados em unidades de saúde de referência na cidade do Recife, Estado de Pernambuco, Brasil, entre 2005 e 2010. Epidemiol Serv Saúde. 2013 jan-mar;22(1):77-85. [ Links ]
27. Palaci M, Dietze R, Hadad DJ, Ribeiro FKC, Peres RL, Vinhas AS, et al. Cavitary disease and quantitative sputum bacillary load in cases of pulmonary tuberculosis. J Clin Microbiol. 2007 Dec;45(12):4064-6. [ Links ]
28. Gomes T, Vinhas SA, Reis-Santos B, Palaci M, Peres RL, Aguiar PP, Ribeiro FKC, et al. Extrapulmonary tuberculosis: Mycobacterium tuberculosis strains and host risk factors in a large urban setting in Brazil. PLoS One. 2013 Oct;8(10):e74517. [ Links ]
29. Reis-Santos B, Locatelli R, Horta BL, Faerstein E, Sanchez MN, Riley LW, et al. Socio-demographic and clinical differences in subjects with tuberculosis with and without diabetes mellitus in Brazil - a multivariate analysis. PLoS One. 2013 Apr;8(4):e62604. [ Links ]
*Article originated from the Master's dissertation entitled 'Vulnerabilities and their influence on the outcome of the tuberculosis treatment, Rio de Janeiro state, 2011 to 2014”, written by Janine Nascimento dos Santos at the Postgraduate Program in Epidemiology and Control of Tuberculosis of the Sergio Arouca National School of Public Health (ENSP/Fiocruz), in 2016.
Received: November 26, 2017; Accepted: March 05, 2018











 texto em
texto em 


