Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.27 no.4 Brasília dez. 2018 Epub 19-Out-2018
http://dx.doi.org/10.5123/s1679-49742018000400002
ORIGINAL ARTICLE
Hearing Screening in children with Congenital Zika Virus Syndrome in Fortaleza, Ceará, Brazil, 2016
1Secretaria da Saúde do Estado do Ceará, Hospital Infantil Albert Sabin, Fortaleza, CE, Brasil
2Centro Universitário Christus, Faculdade de Medicina, Fortaleza, CE, Brasil
3Pontifícia Universidade Católica de São Paulo, Programa de Pós-Graduação em Fonoaudiologia, São Paulo, Brasil
4Universidade Estadual Paulista Júlio de Mesquita Filho, Departamento de Fonoaudiologia, São Paulo, Brasil
5Universidade Federal do Ceará, Departamento de Saúde Comunitária, Fortaleza, CE, Brasil
Objective:
to describe the results of hearing screening performed in children with Congenital Zika Virus Syndrome (CZS) in Fortaleza, Ceará, Brazil.
Methods:
this was a descriptive cross-sectional study involving children with CZS receiving health care in Fortaleza, 2016; the hearing screening tests performed were immittance audiometry, transient otoacoustic emissions (TOAE), acoustic reflexes, and cochleopalpebral reflex (CPR).
Results:
The study included 45 children with an average age of 10 months. 44 of them underwent tympanometric screening, with 16 of these having the right ear within the normal range and 22 having the left ear within the normal range. Among the 43 children evaluated by TOAE, 30 "passed" in both ears, nine "refered" in both ears and four "refered" just in ear; 13/43 "refered" and needed to repeat screening. 43 children evaluated by CPR, 37 showed responses.
Conclusion:
most of the children evaluated had completed cochlear function and middle ear results refer in compatible with their age range.
Keywords: Epidemiology, Descriptive; Zika Virus Infection; Microcephaly; Arboviruses; Hearing Loss
Introduction
Zika virus was identified in Brazil in April 2015 as the etiologic agent of an acute exanthematic disease spreading throughout Northeast Brazil.1-2 In October of the same year the Brazilian Ministry of Health alerted as to a microcephaly epidemic possibly related to cases of Zika virus infection in pregnant women. In 2016 cases began to be published with radiological alterations suggestive of congenital Zika virus infection.2-6
There were 10,867 reported suspected cases of Congenital Zika Virus Syndrome (CZS) in Brazil between October 2015 and December 2016, of which 2,366 were confirmed.7 The majority of these cases (64.6%) occurred in the Northeast region of the country, in particular in the states of Pernambuco, Bahia, Paraíba and Ceará.7
Although the clinical spectrum of CZS is still not completely elucidated, findings about neurological, ophthalmological and orthopedic harm have already been well described.3,6,8-10 However, few studies have addressed auditory aspects associated with congenital Zika virus infection.11-12 A recent study, involving infants with microcephaly in Pernambuco, reported a proportion of 4/69 for unilateral or bilateral sensorineural hearing loss,13 compatible with hearing losses caused by congenital viral infections. However, cases involving sensorineural hearing loss due to congenital viral infection, usually bilateral, severe and profound, are often undetectable at birth, since such loss can be progressive.14
As the congenital Zika virus syndrome still has characteristics that have not been completely elucidated, periodic and thorough hearing screening tests and assessment of affected children should be encouraged.
The objective of this study was to describe the results of hearing screening in children with CZS in Fortaleza, Ceará, Brazil.
Methods
This was a descriptive cross-sectional study involving children with CZS, accompanied by a pediatric referral hospital located in Fortaleza, capital of Ceará State, in the Northeast region of Brazil.
Children with CZS considered to be eligible for this study were those receiving health care during the 2nd Congenital Zika Follow-up Campaign held on December 2nd and 3rd 2016. Study inclusion criteria were: a) cases with confirmed CZS diagnosis, according to criteria adopted by the Brazilian Ministry of Health;15 and (b) cases reported as confirmed by the Ceará State Department of Health, who had microcephaly at birth and clinical/epidemiological/radiological signs compatible with CZS, with negative results for syphilis, toxoplasmosis, rubella, cytomegalovirus and herpes (TORCH).
The exclusion criteria were: a) cerumen obstruction of the external auditory meatus; (b) influenza or cold symptoms that could compromise the examination; and (c) behaviors preventing the proper performance of the hearing screening tests, such as crying, screaming and excessive movement of the head and body.
Hearing ability was investigated by means of screening, with fast and simple procedures to identify those children needing subsequent diagnostic investigation for confirmation of the type and degree of hearing loss.
The hearing screening tests were administered to all children, by a speech therapist specialized in audiology and experienced in child hearing assessment. The following tests were performed: immittance audiometry, transient otoacoustic emissions (TOAE), acoustic reflexes and cochleopalpebral reflex (CPR).
These tests were selected considering that babies with CZS can be very irritable, which makes it very difficult to carry out tests that require a long amount of time during natural sleep.
The following equipment was used: Heine mini otoscope for otoscopy, agogo bells for CPR tests, Interacoustics OtoRead evoked otoacoustic emission recording equipment and Interacoustics AD250H immittance audiometer. All the equipment was calibrated beforehand according to internationally recommended technical standards, and the tests were performed in a quiet air-conditioned room, with the child either asleep or in a calm behavioral state.
When performing immitance audiometry we used the tympanometric curve classification suggested by Jerger:16
‘A' - normal mobility of the tympanic-ossicular system, where compliance values are between 0.3 and 1.6 and pressure is between +100 and -100daPa;
‘Ar' - low mobility of the tympanic-ossicular system, where compliance values are below 0.3ml and pressure is between +100 and -100daPa;
‘Ad' - hypermobility of the tympanic-ossicular system, where compliance values are greater than 1.6Ml and pressure is between +100 and -100daPa;
‘B' - lack of mobility of the tympanic-ossicular system, when compliance and pressure values are not measurable; and
‘C' - negative air pressure in the middle ear, where compliance values are between 0.3 and 1.6ml and pressure is between -200 and -100daPa.
Following classification of the tympanometric curve, the results were divided into two groups: normal (tympanometric curve type 'A') and altered (tympanometric curve type 'Ar', 'Ad', 'C' or 'B'). The 226Hz probe was used as the criterion for reflex analysis. In cases with alteration of tympanometric curve type 'B', the TOAE test was performed, since this type of test characterizes the presence of liquid, making it impossible to capture emissions. These children were referred to an ear, nose and throat (ENT) specialist.
After the assessment of the type of tympanometric curve, the ipsilateral acoustic reflexes were measured and classified as 'present' or 'absent'. ‘Present’ was taken to be when the reflex was triggered between 70 and 100dB above the threshold in least two frequencies, including the 1,000Hz and 2,000Hz frequency.17
Following the assessment of the conditions of the middle ear, cochlear (inner ear) function was assessed specifically in relation to outer hair cells (OHC), by examining Transient Otoacoustic Emissions. This result was classified as "pass" or "refer". Pass when when there were responses in at least three frequency bands, including the 4kHz frequency; and with regard to the signal, noise equal or greater than 6dB.18
The cochleopalpebral reflex (CPR) was then examined. The classification used was 'present' or 'absent'. ‘Present’ was when the agogo bell was struck quickly and loudly and the child opened and closed its eyelids rapidly.19
Children who showed signs of changes or failure in any one of the screening procedures were given a written referral to an ENT specialist requesting subsequent hearing screening retest or diagnostic audiological assessment for confirmation of hearing loss.
The additional data were collected by means of interviews with the mothers or guardians of the children present at the moment of care, using a semi-structured questionnaire developed by the speech therapy team at the Ceará pediatric referral hospital. The interviews were conducted by a team of speech therapists present during the Zika Follow-up Campaign. Some variables were taken from medical records, including gestational age, head circumference, tests performed (TORCH), presence of symptoms during pregnancy and the trimester in which the onset of these symptoms occurred.
The following variables were analyzed:
a) variables related to mother and child:
- gestational age at birth (in months);
- tests performed (serology for TORCH: positive; negative);
- type of delivery (normal; cesarean);
- sex of the child (male; female);
- presence of symptoms during pregnancy (rash and/or fever);
- trimester of appearance of rash in pregnancy (first, second, third); and
- head circumference (cm);
b) immitance audiometry results for each ear (pressure between -200 and +200daPa) and compliance (between 0 and 3ml) (normal; altered);
c) the stapedial reflex (SR) (ipsilateral) of each ear (at the following frequencies: 500; 1,000; 2,000; 4,000 kHz) (present; absent);
d) transient otoacoustic emissions (TOAE) test result for each ear (at the following frequencies: 1; 1.5; 2; 3; 4kHz) (present; absent);
e) cochleopalpebral reflex (agogo bell) (present; absent); and
f) case management defined by the speech therapist (referral to ENT specialist; or referral for diagnosis of the brainstem auditory evoked potential and auditory steady state potential; or retesting - TOAE).
The data collection instrument underwent a pilot study to test understanding of the questions and applicability to the specific audience. The professionals were trained and also supervised by an experienced speech pathologist as patients were examined. The questionnaires were reviewed prior to data input. Epi Info was used for data input and consistency checking.
The variables relating to childbirth (type of delivery, presence of symptoms during pregnancy and trimester in which symptoms occurred), variables relating to the child (sex, gestational age, and head circumference) and variables relating to audiological aspects (type of tympanometric curve, stapedial reflex result, TOAE result and cochleopalpebral reflex) were compared between children with laboratory-confirmed CZS and those with CZS confirmed by clinical/epidemiological/radiological criteria. In order to enable better understanding of hearing test results, the data were separated by ear, and correlated with the tests performed.
Mean head circumference at birth (HC), taken from medical records and checked with the parents, was calculated by dividing the number of HC measurements by the number of children for whom this measurement was available. Both the mean and standard deviation were calculated using Epi Info.
The data were input to Epi Info and analyzed using this program. The association between the results of hearing screening, the confirmation criterion and the clinical-epidemiological aspects were analyzed in contingency tables, estimating prevalence ratios and respective 95% confidence intervals (95%CI). Associations were verified using the Fisher exact test, adopting a significance level of 5%.
This study is part of a research initiative on analysis of biopsychosocial changes occurring in Zika virus embryopathy cases in Ceará, approved on 24 September 2016 by means of Certification of Submission for Ethical Appraisal (CAAE) 57109816.7.0000.5042: Protocol 1.743.023. The children’s parents or guardians were informed about the research and signed a Free and Informed Consent form.
After screening, the children participating in the study were referred for brainstem auditory evoked potential tests, referred to ENT specialists, referred for transient otoacoustic emissions retesting and/or steady state auditory evoked potential testing, as indicated.
Results
Seventy-three children were brought to the Zika Follow-up Campaign. Of these, 23 were excluded because they had cerumen obstruction of the external auditory meatus, and a further five because they had colds and behaviors that prevented the examinations from being carried out. 45 children were evaluated, 27 of whom were of the female sex (Figure 1).
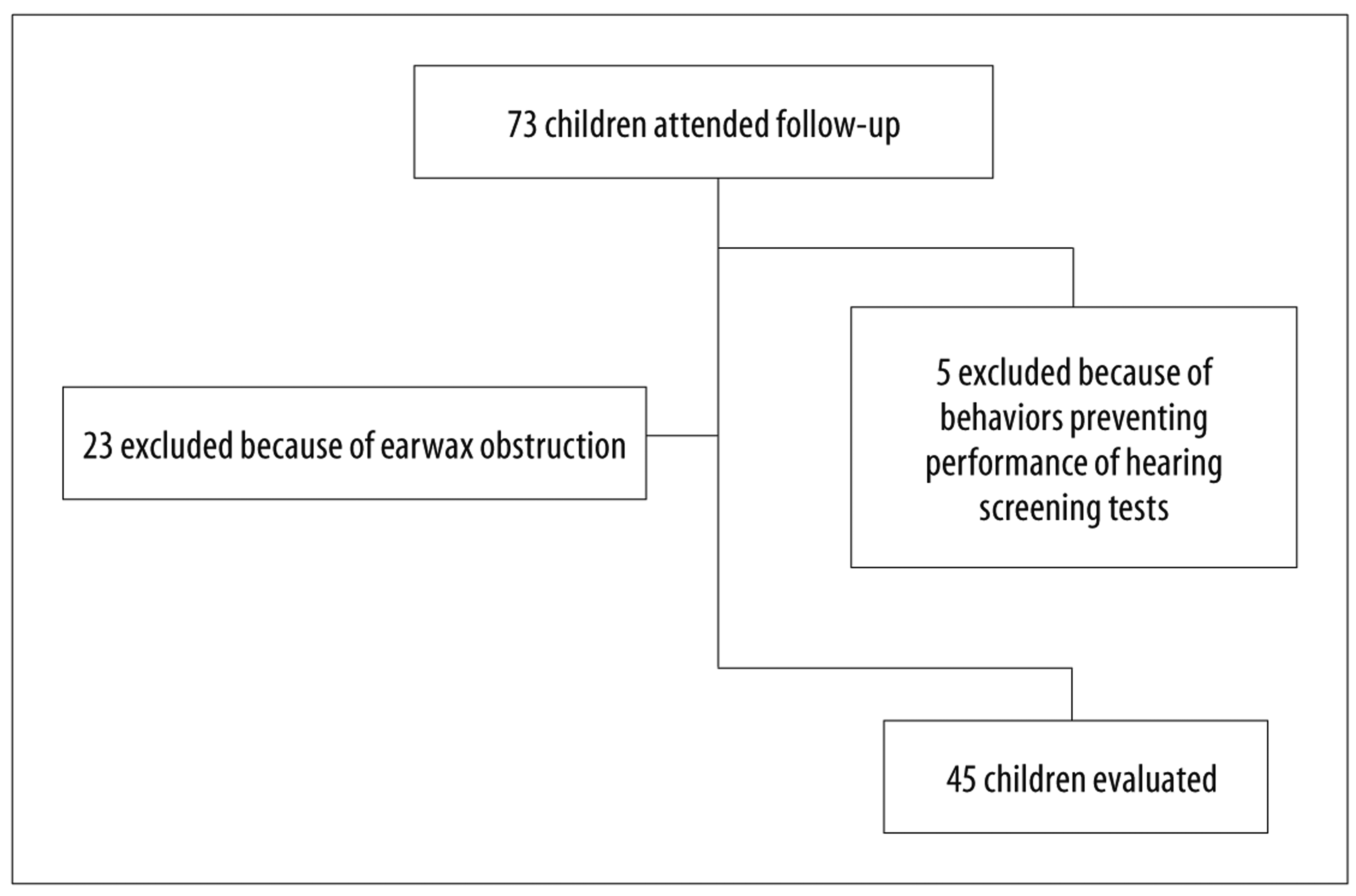
Figure 1 - Flowchart for the assessment of children with Congenital Zika Virus Syndrome at the 2nd Zika Follow-up Campaign in Fortaleza, Ceará, 2017
Mean head circumference at birth was 29.4cm (standard deviation: 2.34cm) and average age at the time of assessment was 10 months (range: 1.20). In relation to pregnancy, 32 children were born at term and 26 were delivered by cesarean section. Of the 30 mothers who reported the presence of rash during pregnancy, 20 reported this symptom in the first trimester.
Eleven cases had laboratory confirmation of the presence of CZS and the remainder were confirmed by the State Department of Health using clinical/epidemiological/radiological criteria. Even though there was higher prevalence of reported symptoms of Zika virus infection during pregnancy in women with case confirmation using the clinical-epidemiological criterion, as well as in other aspects related to childbirth or to the child, no variable analyzed presented a statistically significant difference in relation to the confirmation criterion (Table 1).
Table 1 - Characteristics of mothers and children with Congenital Zika Virus Syndrome (n=45), by case confirmation criteria, among children seen during the 2nd Zika Follow-up Campaign in Fortaleza, Ceará, 2017
| Variables | Laboratory confirmation | Clinical/ Epidemiological/ radiological confirmation | PR | 95% CIb | P -value |
|---|---|---|---|---|---|
| n | n | ||||
| Mother reported virus symptoms during pregnancy | |||||
| Yes | 6 | 26 | |||
| No | 5 | 8 | 0.49 | 0.18;1.32 | 0.163 |
| Trimester of gestation with onset of virus infection symptoms | |||||
| First | 3 | 18 | |||
| Other | 3 | 8 | 0.52 | 0.13;2.18 | 0,390c |
| Gestational age at birth | |||||
| Preterm | 1 | 8 | |||
| Term | 10 | 26 | 0.40 | 0.06;2.73 | 0,416c |
| Type of delivery | |||||
| Cesarean | 6 | 23 | |||
| Normal | 5 | 11 | 0.66 | 0.24;1.83 | 0.483 |
| Child's sex | |||||
| Female | 5 | 22 | |||
| Male | 6 | 12 | 0.56 | 0.20;1.55 | 0.304 |
a) PR: prevalence ratio.
b) 95%CI: 95% confidence interval.
c) Fisher's exact test.
The immitance audiometry test was performed on 44 of the 45 children. It was not possible to perform the examination in one child because it kept crying and was irritated. Thus, in relation to the right ear, 16 children were diagnosed as being within normality (type 'A' curve) and 28 refered tympanometric screening (12 type 'Ar' and 16 type 'C'). With regard to the left ear, 22 children were diagnosed as being within normality (type 'A' curve) and 22 refered (12 type 'Ar'; 10 type 'C'). A greater number of failures was found in the right ear (Table 2).
Table 2 - Results of hearing screening of children with Congenital Zika Virus Syndrome among those tested at the 2nd Zika Follow-up Campaign in Fortaleza, Ceará, 2017
| Audiologic Tests | n | % |
|---|---|---|
| Immitance Audiometry (right ear) | ||
| Altered | 28 | 63.6 |
| Normal | 16 | 36.4 |
| Immitance Audiometry (left ear) | ||
| Altered | 22 | 50.0 |
| Normal | 22 | 50.0 |
| Stapedial Reflex (right ear) | ||
| Absent | 12 | 27.3 |
| Present | 32 | 72.7 |
| Stapedial Reflex (left ear)¥ | ||
| Absent | 11 | 25.0 |
| Present | 33 | 75.0 |
| TOAE (right ear) | ||
| Refered | 11 | 25.6 |
| Passed | 32 | 74.4 |
| TOAE (left ear)& | ||
| Refered | 12 | 27.9 |
| Passed | 31 | 72.1 |
| RCP& | ||
| Absent | 6 | 14.0 |
| Present | 37 | 86.0 |
TOAE: Transient Otoacoustic Emissions.
CPR: Cochleapalpebral Reflex.
a) Fisher's exact test.
& 43 evaluated children.
¥ 44 evaluated children.
The ipsilateral acoustic reflex test was performed on 44 of the 45 children. The examination was not performed in one of them because the child kept crying and was irritated. With regard to the right ear, 32 children showed presence of acoustic reflex and other 12 showed absence of this reflex. Among these 12 children who showed no acoustic reflex in the right ear, ten had alterations in the middle ear, being three type 'Ar' and seven type 'C'. As for the left ear, 33 children showed presence of reflex and only 11 presented absence, eight of whom had an altered tympanometric curve: five type 'Ar' and three type 'C' (Table 2).
Among the 43 out of 45 children transient otoacoustic emissions (TOAE) test was performed, 30 "passed" with both ears, nine "refered" in both ears and four "refered" in only one ear (one in the right ear and three in the left ear) (Table 2 and Table 3).
Table 3 - Results of hearing screening of children with Congenital Zika Virus Syndrome, by ear examined, among those seen at the 2nd Zika Follow-up Campaign in Fortaleza, Ceará, 2017
| Audiologic Tests | Tympanometry | |||
|---|---|---|---|---|
| Right | Left | |||
| Altered n | Normal n | Altered n | Normal n | |
| TOAEa | ||||
| Refered | 10 | 1 | 9 | 3 |
| Passed | 17 | 15 | 12 | 19 |
| Stapedial Reflex | ||||
| Absent | 10 | 2 | 8 | 3 |
| Present | 18 | 14 | 14 | 19 |
a) TOAE: Transient Otoacoustic Emissions.
The TOAE and tympanometry tests were performed on 43 of the 45 children: 13/43 refered screening so that the tests needed to be repeated. Considering only the right ear, 32 "passed" the TOAE test. Among those who "refered", one had a normal tympanometric curve and ten had alterations. As for the left ear, 31 children "passed"; of the 12 that "refered," nine had tympanometric curve alterations and three had a normal tympanometric curve (Table 2 and Table 3).
Of the 43 children evaluated regarding the cochleopalpebral reflex (CPR), 37 showed responses. Among the six with no response, five had tympanometric curve alterations - four bilateral; and one on the right side -; only one child showed absence of CPR and a normal tympanometric curve (Table 2 and Table 3).
Discussion
The majority of children with CZS evaluated had full cochlear function with regard to outer hair cells, and alterations of the middle ear compatible with children with normal development for their age and without CZS.
The findings showed that ‘Ar’ and ‘C’ type curves were the most frequent. It is known that the sensory deprivation resulting from changes in the middle ear, aggravated by the number and duration of disease episodes, can affect speech perception and make it difficult to understand, especially in noisy environments, and may even harm the child’s development of language.20 A study performed with children under two years of age, with normal development, also found high prevalence (68.4%) of tympanometry tests compatible with alteration of the middle ear (type 'B' and 'C'). The same study highlights the first year of life as the period of greatest incidence of middle ear liquid.20
Tympanometry consists of checking the mobility of the tympanic-ossicular system through the variation introduced into the external auditory meatus, being effective in the early identification of middle ear disorders.20,21 Recurrent otitis and diseases that affect the middle ear are very common in children and infants and this is why tympanometry was used in the hearing screenig.
Associated with the tympanometric curve analysis, the ipsilateral acoustic reflex test was also conducted with stimulation in loud intensity, allowing the contraction of the stapedius muscle and therefore enabling assessment of the integrity of the tympanic-ossicular system.19 This reflex was found in most of the ears, thus suggesting integrity of the auditory pathway.
Over half of the children from the sample "passed" the transient otoacoustic emissions (TOAE) screenig in both ears, suggesting that the cochlear function with regard to outer hair cells was intact. However, more than a third "refered" in at least one ear. This outcome was different from a study conducted in another municipality in the Brazilian Northeast, where, when assessing 23 cases, only two of them presented auditory deficit.22 This difference is probably due to the tympanometric curve, because the majority of children who "refered" the TOAE screenig also showed a change in the tympanometric curve, thus justifying the absence of response and indicating changes in the conduction of sound. In addition, both studies were carried out with small samples, thus requiring careful consideration of the results. Therefore, we suggest that this assessment can be expanded to include a larger number of children with CZS.
Based on the assumption that changes to middle ear function may interfer in TOAE acquisition - and that middle ear effusion is found frequently in children under two years old, it is possible that the absence of transient otoacoustic emissions responses in this study sample is associated with discrete tympanometric alterations. Nevertheless, recent findings in children with CZS pointed possibility that conductive hearing loss may not be related to Zika virus infection, since the hearing loss caused by congenital viral infections would be sensorineural loss.12 These issues need to be reviewed and assessed in greater depth in future studies.
A study comparing TOAE and tympanometry results with infants with no hearing loss risk indicators showed a greater proportion of tympanometric alteration in the group of infants with absence of TOAE,21 corroborating the findings of our study. Otitis media with effusion is often asymptomatic and can go unnoticed owing to its symptoms being less evident than those of acute otitis media. It can occur spontaneously due to Eustachian tube dysfunction or as a result of a previous infectious process, among other factors.23
The use of automatic brainstem auditory evoked potential (AABR) should be included in the hearing screening protocol for children affected by CZS. It is a more precise procedure for identifying children with possible sensorineural hearing losses, as advocated by the Joint Committee on Infant Hearing (JCIH)24 and the Multidisciplinary Committee on Hearing Health (COMUSA).25 However, it is known that these protocols are still not properly implemented in Brazil, nor is universal neonatal hearing screening (UNHS). Their implementation would contribute greatly to hearing loss identification, diagnosis and intervention among children, especially those with CZS.
All the children in the sample who had middle ear alterations showed absence of ipsilateral stapedial reflex. Measurement of acoustic reflex in the neonatal and infant population proved itself once more to be an adequate and possible assessment method, capable of contributing with information about the integrity of the auditory pathway, even though this test is not frequently performed in this population.
This study expands knowledge about possible auditory alterations in children with CZS. This knowledge is needed to establish goals for interdisciplinary intervention, despite the limitations of this study. Among its limitations, we highlight the lack of information on auditory thresholds and lack of complete audiological diagnosis. However, the results relate only to the hearing screening stage, with a strong indication of the need for complementation of medical and audiological evaluation to confirm temporary hearing losses, such as conductive, and sensorineural hearing losses. There are also limitations related to diagnostic conclusion, in addition to the difficulties in being able to assess children with CZS while they are in a state of calm and deep sleep in order to carry out electrophysiological tests commonly used in the pediatric population, the objective of which is to determine the degree and type of hearing loss. The use of sedation and/or anesthesia may prove to be necessary in this sense.
Other limitations are the small sample size and the fact that only a quarter of the cases were laboratory-confirmed. All the other cases were confirmed by the Ceará State Department of Health using the clinical/epidemiological/radiological criterion, after excluding of those with negative TORCH results. Even so, it was evidenced - statistically - that these children had the same profile, differing only in the absence of the confirmatory test, to which many mothers did not have access during the beginning of the epidemic.
It is suggested that tympanometry should be included in hearing screening, before referring for ABO since this is a test that is hard to access through the Brazilian Unified Health System in some regions of Brazil.
The majority of children with CZS screened showed full cochlear function in relation to outer hair cells, and alterations of the middle ear compatible with children with normal development for their age. We suggest that hearing screening should be carried out in all children with CZS soon after birth, and that only children with test failures should be referred for medical and audiological diagnosis, at an early age, which would also facilitate the performance of these procedures.
In addition, hearing screening using TOAE and impedance audiometry, as these are easy to perform, proved capable of identifying children to be referred for medical and audiological diagnosis. The findings also suggest the need to scale up access to hearing health referral services, capable of providing continuity to the hearing screening process in order to gain knowledge of the epidemiology of hearing loss in children with congenital Zika virus syndrome.
Referências
1. Campos GS, Bandeira AC, Sardi SI. Zika virus outbreak, Bahia, Brazil. Emerg Infect Dis. 2015 Oct;21(10):1885-6. [ Links ]
2. Heukelbach J, Alencar CH, Kelvin AA, Oliveira WK, Cavalcanti LPG. Zika virus outbreak in Brazil. J Infect Dev Ctries. 2016 Feb;10(2):116-20. [ Links ]
3. Schuler-Faccini L, Ribeiro EM, Feitosa IML, Horovitz DDG, Cavalcanti DP, Pessoa A, et al. Possible association between Zika virus infection and microcephaly - Brazil, 2015. MMWR Morb Mortal Wkly Rep. 2016 Jan;65(3):59-62. [ Links ]
4. Ministério da Saúde (BR). Ministério da Saúde confirma relação entre Zika vírus e microcefalia [Internet]. 2015 [citado 2017 jan 30]. Disponível em: Disponível em: http://portalsaude.saude.gov.br/index.php/o-ministerio/principal/secretarias/svs/noticias-svs/21016-ministerio-da-saude-confirma-relacao-entre-virus-zika-e-microcefalia [ Links ]
5. Eickmann SH, Carvalho MDCG, Ramos RCF, Rocha MAW, van der Linden V, Silva PFS. Zika virus congenital syndrome. Cad Saúde Pública. 2016 Jul;32(7):e00047716. [ Links ]
6. Rasmussen SA, Jamieson DJ, Honein MA, Petersen LR. Zika virus and birth defects--reviewing the evidence for causality. N Engl J Med. 2016 May;374(20):1981-7. [ Links ]
7. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre de chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 52, 2016. Bol Epidemiológico [Internet]. 2017 [citado jun 2018 28];48(3):1-11. Disponível em: Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2017/abril/06/2017-002-Monitoramento-dos-casos-de-dengue--febre-de-chikungunya-e-febre-pelo-v--rus-Zika-ate-a-Semana-Epidemiologica-52--2016.pdf [ Links ]
8. Moore CA, Staples JE, Dobyns WB, Pessoa A, Ventura CV, Fonseca EB, et al. Characterizing the pattern of anomalies in congenital zika syndrome for pediatric clinicians. JAMA Pediatr. 2017 Mar;171(3):288-95. [ Links ]
9. Aragão MFV, van der Linden V, Brainer-Lima AM, Coeli RR, Rocha MA, Sobral da Silva P, et al. Clinical features and neuroimaging (CT and MRI) findings in presumed Zika virus related congenital infection and microcephaly: retrospective case series study. BMJ. 2016 Jun;353:i1901. [ Links ]
10. Ventura CV, Maia M, Bravo Filho V, Góis AL, Belfort Júnior R. Zika virus in Brazil and macular atrophy in a child with microcephaly. Lancet. 2016 Jan;387(10015):228. [ Links ]
11. Miranda FilhoDB, Martelli CMT, Ximenes RAA, Araújo TVB, Rocha MAW , Ramos RCF , et al. Initial description of the presumed congenital Zika syndrome. Am J Public Health. 2016 Apr;106(4):598-600. [ Links ]
12. Leal MC, Muniz LF, Caldas NetoSS, van der Linden V, Ramos RC. Sensorineural hearing loss in a case of congenital Zika virus. Braz J Otorhinolaryngol. 2016 Jun;S1808-8694(16):30127-6. [ Links ]
13. Leal MC, Muniz LF, Ferreira TSA, Santos CM, Almeida LC, van der Linden V, et al. Hearing loss in infants with microcephaly and evidence of congenital Zika virus infection - Brazil, November 2015-May 2016. MMWR Morb Mortal Wkly Rep. 2016 Sep;65(34):917-9. [ Links ]
14. Cohen BE, Durstenfeld A, Roehm PC. Viral causes of hearing loss: a review for hearing health professionals. Trends Hear. 2014 Jul;18:233121651454136. [ Links ]
15. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Protocolo de vigilância e resposta à ocorrência de microcefalia relacionada à infecção pelo vírus Zika [Internet]. Brasília: Ministério da Saúde; 2015 [citado 2017 set 11]. 55 p. Disponível em: Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2015/dezembro/09/Microcefalia-Protocolo-de-vigilância-e-resposta-versão-1-09dez2015-8h.pdf [ Links ]
16. Jerger J. Clinical experience with impedance audiometry. Arch Otolaryngol. 1970 Oct;92(4):311-24. [ Links ]
17. Moller AR. Acoustic reflex in man. J Acous Soc Am. 1962 Sep;34(9B):1524. [ Links ]
18. Garcia MV, Azevedo MF, Testa JR. Accoustic immitance measures in infants with 226 and 1.000hz probes: correlation with otoacoustic emissions and otoscopy examination. Braz J Otorhinolaryngol. 2009 Jan-Feb;75(1):80-9. [ Links ]
19. Redondo MC, Bunner AP. Avaliação clínica da audição da criança: procedimentos e interpretação de resultados. In: Levy CCAC. Manual de audiologia pediátrica. Barueri (SP): Manole; 2015. p. 58-82. [ Links ]
20. Saes SO, Goldberg TB, Montovani JC. Secretion of middle ear in infants: occurrence, recurrence and related factors. J Pediatr. 2005 Mar-Apr;81(2):133-8. [ Links ]
21. Linares AE, Carvallo RMM. Acoustic immittance in children without otoacoustic emissions. Braz J Otorhinolaryngol. 2008 May-Jun;74(3):410-6. [ Links ]
22. Microcephaly Epidemic Research Group. Microcephaly in infants, Pernambuco State, Brazil, 2015. Emerg Infect Dis. 2016 Jun;22(6):1090-3. [ Links ]
23. Di Francesco RC, Barros VB, Ramos R. Otitis media with effusion in children younger than 1 year. Rev Paul Pediatr. 2016 Jun;34(2):148-53. [ Links ]
24. American Academy of Pediatrics, Joint Committee on Infant Hearing. Year 2007 position statement: principles and guidelines for early hearing detection and intervention programs. Pediatrics. 2007 Oct;120(4):898-921. [ Links ]
25. Lewis DR, Marone SAM, Mendes BCA, Cruz OLM, Nóbrega M. Multiprofessional committee on auditory health: COMUSA. Braz J Otorhinolaryngol. 2010 Jan-Feb;76(1):121-8. [ Links ]
Received: January 18, 2018; Accepted: May 07, 2018











 texto em
texto em 


