Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.27 no.4 Brasília dez. 2018
http://dx.doi.org/10.5123/s1679-49742018000400012
ORIGINAL ARTICLE
The impact of pneumococcal vaccine in reducing pneumonia hospitalizations in children under 5 years old, in Santa Catarina, Brazil, 2006 a 2014*
1Universidade Federal de Santa Catarina, Departamento de Saúde Pública, Florianópolis, SC, Brasil
Objective:
to analyze the impact of pneumococcal conjugate vaccine (PCV10) on pneumonia hospital admissions among children in Santa Catarina, Brazil, 2006-2014.
Methods:
this was an ecological study using data obtained from Brazilian National Health System Information Technology Department (Datasus) for 2006-2009 (pre-vaccination period) and 2010-2014 (post-vaccination period); time trends were evaluated using the Poisson regression coefficient.
Results:
in the comparison between pre- and post-vaccination periods, the percentage difference in the hospitalization rate for children under 1 year old ranged from -44.1% in the Western region to -1.4% in the Serrano Plateau region, and in children between 1-4 years old it ranged from -37.1% in the Northern Plateau region to 16.9% in the Serrano Plateau region (p<0.05); hospitalization rates in the state reduced by 23.3% in children under 1 year old and by 8.4% in those aged 1-4 years.
Conclusion:
a significant reduction in the rate of pneumonia hospitalization in children under 1 year old age was found, suggesting the effectiveness of the vaccine in reducing hospitalizations.
Keywords: Pneumonia; Pneumococcal Vaccines; Hospitalization; Child.
Introduction
Pneumonia and diarrhea are the leading causes of death among children under 5 years old in low income countries globally.1 These deaths could be avoided through prevention measures, such as vaccination, and adequate patient management by family members and health professionals.2
According to the World Health Organization, in 2015 the main cause of death among Brazilian children aged between 1 and 59 months was pneumonia (17.6%), followed by accidents (12.6%) and diarrhea (5.5%).3 Globally, some 156 million new episodes of pediatric pneumonia were recorded in 2000, with more than 95% occurring in middle income countries. Of the total pneumonia cases in these countries, 8.7% to 10.0% are severe and require hospitalization,1,4 while 90.0% (confidence interval: 87%-93%) are cared for in outpatient services.5
In the far south of Brazil, respiratory diseases, including pneumonia, lead to children under 5 years old being hospitalized, especially in winter months.6 Low temperatures, as well as other factors such as malnutrition, low birth weight, non-exclusive breastfeeding in the first four months of life and high family density (seven or more people living in the same household), contribute to increased occurrence of pneumonia in some subtropical regions.7
Studies of these cases have found a reduction in the number of respiratory disease cases and hospitalizations,2 whereby the introduction of 10-valent pneumococcal vaccine (PCV10) is yet another important strategy for reducing cases of pneumonia, acute otitis media and invasive pneumococcal disease (IPD) caused by Streptococcus pneumoniae serotypes contained in the vaccine.8
PCV10 was included on the vaccine calendar and, with effect from March 2010, has been extended to all children under 2 years old. Initially the immunization schedule was comprised of two doses plus a booster dose, but was altered in 2016 to two doses at 2 and 4 months old, plus a booster dose, as well as extending the age group to children up to 4 years old.8 Vaccination has been found to be effective in reducing pneumococcal disease in countries such as Chile,9 Peru,10 Uruguay11 and Mexico;12 however effectiveness depends on the occurrence of the disease and circulating Streptococcus pneumoniae serotypes.
Changes in the behavior of the disease over time and alteration in circulating serotypes, as well as the existence of other vaccines such as PCV13, which is already being used in Uruguay11 and Mexico,13,14 demonstrate the importance of circulating serotypes, as well as the importance of in-depth assessments of the distribution of pneumococcal diseases, including pneumonia.15
PCV10 can be seen as one of the important factors in reducing pneumonia hospitalization rates in children under 1 year old in the state of Santa Catarina, given that pneumonia caused by Streptococcus pneumoniae is one of the leading causes of morbidity6 and mortality among children in Southern Brazil. Data from an ecological study of pneumonia hospitalizations in children and adolescents living in the neighboring state of Paraná in relation to the period 2000-2011 describe the occurrence of 2,295,780 hospitalizations of children and adolescents, 59,028 (2.57%) of which were due to bacterial pneumonia.16
In view of the need to integrate different aspects related to measuring the impact of introducing a vaccine in a specific region, the objective of this article is to describe the impact of PCV10 on pneumonia hospitalizations of children under 5 years old in the state of Santa Catarina, Brazil, between 2006 and 2014.
Methods
This study had an ecological observational design, using secondary annual data enabling evaluation over time, i.e. before and after PCV10 implantation.
With regard to the period evaluated, namely 2006-2014, we selected the 4 years prior to PCV10 implantation, the year of its implantation and the 4 following years, exclusively in relation to the state of Santa Catarina.
Data on live births (Live Births Information System - Sinasc), population aged 1 to 4 years old (Brazilian Institute of Geography and Statistics - IBGE), pneumonia hospitalizations (Hospital Information System of the Brazilian National Health System - SIH/SUS) and number of vaccine doses administered (National Immunization Program Information System [SI-PNI]) we selected from Ministry of Health information systems, provided by the Brazilian National Health System Information Technology Department (Datasus)17 and confirmed by the State of Santa Catarina Health Surveillance Directorate database. In turn, the source of data on the number of beds available for hospitalization was the National Registry of Health Establishments (CNES) regarding active beds in the month of July for each year covered by the study. This data was also provided by Datasus.17
All of Santa Catarina’s 295 municipalities were grouped together according to the regions of the state in which they are located. However, as two of the state’s municipalities were emancipated in 2006 and 2010, in order to enable adequate evaluation of each region, all the information for both of these municipalities was added to the municipalities from which they originated, thus totaling 293 municipalities analyzed.
We identified records of pneumonia hospitalizations of children aged under 5 years old and also PCV10 coverage of children aged under 1 year old, the length of time of pneumonia hospitalization per number of inpatient days, the number of beds available for hospitalization, access to primary health care and the human development index (HDI) (Figure 1). Data on estimated coverage of resident population by primary care teams were provided by the State of Santa Catarina Health Surveillance Directorate, based on the ‘estimated population coverage by primary care teams’ indicator, calculated by taking the number of Family Health teams x 3000 x 100, divided by the population in the same place and at the same time, as recommended by the National Primary Care Policy (Health Ministry Ordinance GM/MS No. 2488, dated October 21st 2011); HDI, a concise measurement of the long-term progress of municipalities with regard to three basic dimensions - income, education and health -, was retrieved from material provided by the Brazilian office of the United Nations Development Program (UNDP).

a) Hospitalizations of children aged under 5 years old with pneumonia diagnosed as underlying cause (International Statistical Classification of Diseases and Health-Related Problems - 10th Revision [ICD-10]: chapter X; codes J12-J18; respiratory tract diseases) - in the period between January 2006 and December 2014.
b) Vaccination coverage data - available on the National Immunization Program Information System (SIS-PNI) - for children aged under 1 year old vaccinated in public and private health services with PNI conjugate vaccine against Streptococcus pneumoniae; data distributed by health region between March 2010 and December 2014.
c) PCV10: 10-valent pneumococcal vaccine. Exclude, or conjugate pneumococcal vaccine.
Figure 1 Characterization of data collected on health information systems
Hospital morbidity, i.e. total SUS hospitalizations due to pneumonia, by municipality of residence, meets the criteria of the International Statistical Classification of Diseases and Health-Related Problems - 10th Revision (ICD-10) and takes hospitalization as a recording unit. As such, the same child may have been hospitalized one or more times in a year.17
Vaccination coverage is defined by the National Immunization Program (PNI) as the percentage of the target population covered by a complete immunization schedule17 (total number of 3rd PCV10 doses administered in the year x 100/population aged under 1 year old). PCV10 vaccination coverage for children aged under 1 year old, from 2010 to 2014, was calculated taking the total number of third vaccine doses as the numerator and the number of live births as the denominator. With regard to hospital beds available for hospitalization per 1,000 inhabitants, these were calculated taking the total number of beds available in health establishments registered on the CNES system as the numerator, and the total population, i.e.: total beds available in the region x 1,000/resident population, as the denominator.
Vaccination coverage and the rates of pneumonia hospitalizations of children aged under 5 years old for the period 2006-2014, were initially calculated per municipality of residence, and later recalculated for the regional divisions of Santa Catarina state based on total doses administered and total hospitalizations per population resident in each region.
The outcome of interest was the annual rate of pneumonia ‘hospitalization or hospital morbidity’ (ICD-10: J12-18) in children aged under 1 year old and also aged 1-4 years old, per region. The rate was calculated for two periods, prior to vaccination implantation (2006-2009) and after implantation (2010-2014), dividing total hospitalizations by the mid-year population, for each year analyzed.
The independent variables considered in this study were: PCV10 vaccine coverage in the period 2010-2014; beds available for hospitalization in July of each year in the period 2006-2014, per 1,000 inhabitants; percentage coverage of primary health care teams; and HDI in 2010, by municipality and geographical location. All data were grouped together according to the state’s seven regions: Western, Itajaí, Northern Plateau, Northeast, Serrano Plateau, Greater Florianópolis and Southern.
The descriptive statistics included: (i) tabular representation of the percentage reduction rate and the hospitalization rate time trend, per year, in the period before (2006-2009) and after (2010-2014) PCV10 implantation; and (ii) vaccination coverage in children aged under 1 year old. The time trend of the annual rates, in each period, was presented by region and also for the state as a whole.
Impact of vaccination on the outcome in the state’s regions was defined by Poisson’s regression coefficient for each period, separately, and always adjusted by the population aged under 1 year old and by the population aged 1 to four years old. The difference in the time trend between the periods before and after PCV10 vaccination was calculated by comparing the gradients of the annual rates for each period. The year 2006 was selected as the first of the time series assessed and was, therefore, the reference year.
The data were obtained from the respective Ministry of Health information systems (Figure 1), generated by the TABNET application, developed by DATASUS, and were saved as Excel ® version 2013 electronic spreadsheets. The spreadsheets were merged to form a single database which was exported to Stata® (Statistics/Data Analysis) 11.0 with the aid of the Stat/Transfer application.
Participant privacy and confidentiality were guaranteed by excluding personal information from the databases. The ethical principles applying to research were respected, as recommended by National Health Council/Ministry of Health Resolution CNS No. 412, dated December 12th 2012. The study project was approved by the Federal University of Santa Catarina (UFSC) Research Ethics Committee: Opinion No. 712.012, dated July 8th 2014.
Results
In the period 2006-2014, 75,891 children were admitted to hospital with pneumonia in Santa Catarina: 37,703 hospitalizations between 2006 and 2009; 8,087 in 2010; and 30,101 between 2011 and 2014. The time trend of the reduction in the hospitalization rate ratio over the nine-year period evaluated was significant for the state as a whole (p<0.05). In children aged under 1 year old, a reduction was found in all the regions of the state, with the exception of the Serrano Plateau (Table 1). However, in the 1 to four year age group, in addition to the Serrano Plateau, there was also an increase in the hospitalization rate ratio in the Northeast region following PCV10 implantation in the state.
Table 1 - Time trend of annual pneumonia hospitalization rates in children under 5 years old by period of 10-valent pneumococcal vaccine (PCV10) implantation, Santa Catarina, 2006-2014
| Regions | PCV10 implantation period | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Age (in years) | Pre | Implantation | Post | |||||||
| 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | |||
| Western | <1 | HRRa (95%CIc) | 1.1 | 1.0 | 1.0 | 0.8b | 0.7b | 0.7b | 0.6b | 0.6b |
| (1.0;1.2) | (0.9;1.1) | (0.9;1.0) | (0.7;0.8) | (0.7;0.8) | (0.7;0.8) | (0.6;0.7) | (0.6;0.7) | |||
| 1 a 4 | HRRa (95%CIc) | 1.2b | 1.2b | 1.3b | 1.2b | 1.0 | 1.0 | 0.9b | 0.9b | |
| (1.1;1.3) | (1.1;1.3) | (1.2;1.4) | (1.1;1.2) | (0.9;1.0) | (0.9;1.0) | (0.8;0.9) | ( 0.8;0.9) | |||
| Itajaí | <1 | HRRa (95%CIc) | 0.9 | 0.9b | 1.0 | 0.8b | 0. 8b | 0.9 | 0.7b | 0.6b |
| (0.9;1.0) | (0.8;0.9) | (0.9;1.1) | (0.8;0.9) | (0.8;0.9) | (0.9;1.0) | (0.6;0.8) | (0.5;0.6) | |||
| 1 a 4 | HRRa (95%CIc) | 1.0 | 1.2b | 1.4b | 1.2b | 1.2b | 1.2b | 1.0 | 0.8b | |
| (0.9;1.1) | (1.1;1.3) | (1.3;1.5) | (1.1;1.3) | (1.1;1.3) | (1.1;1.3) | (0.9;1.0) | (0.7;0.8) | |||
| Greater Florianópolis | <1 | HRRa (95%CIc) | 1.4b | 1.0 | 1.4b | 1.0 | 0.9 | 0.9 | 0.8b | 0.8b |
| (1.1;1.6) | (0.8;1.2) | (1.1;1.6) | (0.8;1.2) | (0.7;1.1) | (0.8;1.1) | (0.7;0.9) | (0.6;0.9) | |||
| 1 a 4 | HRRa (95%CIc) | 1.2b | 1.2b | 2.1b | 1.5b | 1.5b | 1.2 | 1.3b | 1.2 | |
| (1.1;1.4) | (1.1;1.5) | (1.8;2.4) | (1.3;1.7) | (1.3;1.7) | (1.0;1.4) | (1.1;1.5) | (1.00;1.4) | |||
| Serrano Plateau | <1 | HRRa (95%CIc) | 1.0 | 0.8b | 0.9 | 0.9 | 0.9 | 1.1 | 1.0 | 0.8b |
| (0.8;1.2) | (0.7;0.9) | (0.8;1.1) | (0.7;1.0) | (0.7;1.0) | (0.9;1.3) | (0.9;1.2) | (0.7;0.9) | |||
| 1 a 4 | HRRa (95%CIc) | 1.0 | 0.8b | 1.0 | 1.1 | 1.2 | 1.2b | 1.3b | 1.0 | |
| (0.9;1.2) | (0.7;0.9) | (0.9;1.2) | (0.9;1.2) | (1.0;1.3) | (1.1;1.4) | (1.1;1.5) | (0.9;1.2) | |||
| Northeast | <1 | HRRa (95%CIc) | 0.8b | 0.5b | 0.7b | 0.7b | 0.8b | 0.7b | 0.7b | 0.5b |
| (0.7;0.9) | ( 0.4;0.6) | (0.6;0.8) | (0.6;0.8) | (0.7;0.9) | (0.6;0.8) | (0.6;0.8) | (0.4;0.6) | |||
| 1 a 4 | HRRa (95%CIc) | 0.9 | 0.6b | 1.2b | 1.2b | 1.0 | 1.0 | 1.1 | 0.8b | |
| (0.8;1.0) | (0.5;0.7) | (1.1;1.4) | (1.1;1.4) | (0.9;1.2) | (0.9;1.1) | (0.9;1.2) | (0.7;0.9) | |||
| Southern | <1 | HRRa (95%CIc) | 1.1 | 0.9 | 0.7b | 0.6b | 0.8b | 0.8b | 0.7b | 0.7b |
| (0.9;1.2) | (0.8;1.0) | (0.6;0.8) | (0.6;0.9) | (0.7;0.9) | (0.7;0.9) | (0.6;0.8) | (0.6;0.8) | |||
| 1 a 4 | HRRa (95%CIc) | 1.3b | 1.2b | 1.3b | 1.1 | 1.1 | 1.3b | 1.2b | 1.2b | |
| (1.1;1.4) | (1.1;1.3) | (1.1;1.4) | (1.0;1.2) | (1.0;1.3) | (1.1;1.4) | (1.1;1.3) | (1.1;1.3) | |||
| Northern Plateau | <1 | HRRa (95%CIc) | 0.8b | 0.6b | 0.8b | 0.5b | 0.5b | 0.7b | 0.7b | 0.5b |
| (0.7;0.9) | (0.5;0.7) | (0.7;0.9) | (0.4;0.6) | (0.4;0.6) | (0.6;0.8) | (0.6;0.8) | (0.4;0.6) | |||
| 1 a 4 | HRRa (95%CIc) | 1.0 | 0.8b | 1.0 | 0.6b | 0.6b | 0.8b | 0.7b | 0.7b | |
| (0.8;1.1) | (0.7;0.9) | (0.8;1.1) | (0.5;0.8) | (0.5;0.7) | (0.7;0.9) | (0.6;0.9) | (0.6;0.8) | |||
| Santa Catarina | <1 | HRRa (95%CIc) | 1.0 | 0.8b | 0.9b | 0.8b | 0.8b | 0.8b | 0.7b | 0.6b |
| (0.9;1.0) | (0.8;0.9) | (0.9;0.9) | (0.7;0.8) | (0.7;0.8) | (0.7;0.8) | (0.6;0.7) | (0.5;0.6) | |||
| 1 a 4 | HRRa (95%CIc) | 1.1b | 1.1b | 1.3b | 1.1b | 1.0 | 1.1b | 1.0 | 0.9b | |
| (1.0;1.2) | (1.0;1.1) | (1.2;1.3) | (1.1;1.2) | (1.0;1.1) | (1.0;1.1) | (0.9;1.0) | (0.8;0.9) | |||
a) Hospitalization rate ratio in relation to the year 2006 (indicates reference category)
b) p<0.05.
c) 95%CI: 95% confidence interval.
When the mean hospitalization rates for children aged under 1 year old were examined, the variation was greater than expected, with a reduction of 23.3% in those under 1 year old and 8.4% in those aged one to 4 years old (Table 2).
Table 2 -Annual mean pneumonia hospitalization rate (per 1000 live births) in children under 5 years old, by region and by 10-valent pneumococcal vaccine (PCV10) implantation period, Santa Catarina, 2006-2014
| Regions | Pneumonia hospitalization rate in children under 1 year olda | Pneumonia hospitalization rate in children aged 1-4 years olda | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre-vaccination period | 95%CIb | Post-vaccination period | 95%CIb | Reduction % | Pre-vaccination period | 95%CIb | Post-vaccination period | 95%CIb | Reduction % | |
| Western | 84.1 | 81.9;86.2 | 58.3 | 56.8;59.9 | -44.1 | 22.8 | 22.3;23.3 | 19.0 | 18.6;19.5 | -20.0 |
| Itajaí | 53.8 | 52.2;55.4 | 43.6 | 42.4;44.9 | -23.3 | 15.4 | 14.9;15.8 | 14.3 | 13.9;14.7 | -7.3 |
| Greater Florianópolis | 19.4 | 18.2;20.6 | 14.4 | 13.5;15.3 | -34.6 | 6.4 | 6.0;6.7 | 6.2 | 5.8;6.5 | -3.3 |
| Serrano Plateau | 61.7 | 58.0;65.5 | 60.9 | 57.5;64.3 | -1.4 | 14.7 | 13.9;15.6 | 17.7 | 16.8;18.6 | 16.9 |
| Northeast | 31.3 | 29.7;32.9 | 27.3 | 26.1;28.6 | -14.6 | 9.0 | 8.6;9.4 | 9.8 | 9.4;10.2 | 8.2 |
| Southern | 42.5 | 40.6;44.4 | 33.9 | 32.4;35.4 | -25.5 | 12.2 | 11.6;12.5 | 12.1 | 11.6;12.5 | -0.0 |
| Northern Plateau | 40.6 | 37.9;43.2 | 28.9 | 26.9;31.0 | -40.2 | 12.5 | 11.8;13.2 | 9.2 | 8.6;9.7 | -37.0 |
| Total | 49,8 | 49,0;50,5 | 38,2 | 37,6;38,8 | -23,3 | 14,1 | 13,9;14,3 | 13,0 | 12,8;13,1 | -8,4 |
a) p<0.005.
b) 95%CI: 95% confidence interval.
Variation in the mean hospitalization rate among children aged under 1 year old was negative in all regions of the state, with the largest reductions being found in the Western and Northern Plateau regions (reductions of more than 40.0%). The only region with insignificant reduction in hospitalizations in this age range was the Serrano Plateau: -1.4%. Still in relation to the under 1 year old group, the lowest hospitalization rates were found in Greater Florianópolis: 19.4% in the pre-vaccination period and 14.4% in the post-vaccination period, representing a reduction of 34.6% (Table 2).
In the 1 to four year old age range, mean hospitalization rates were lower: 22.8% in the Western region, 15.4% in Itajaí and 14.7% in the Serrano Plateau region in the pre-vaccination period; while in the post-vaccination period the rates were 19.0% in the Western region, 14.3% in Itajaí and 17.7% in the Serrano Plateau region. The data indicate reductions of 20.0% and 7.3% in the Western and Itajaí regions, respectively, and a 16.9% increase in the hospitalization rate in the Serrano Plateau region (Table 2).
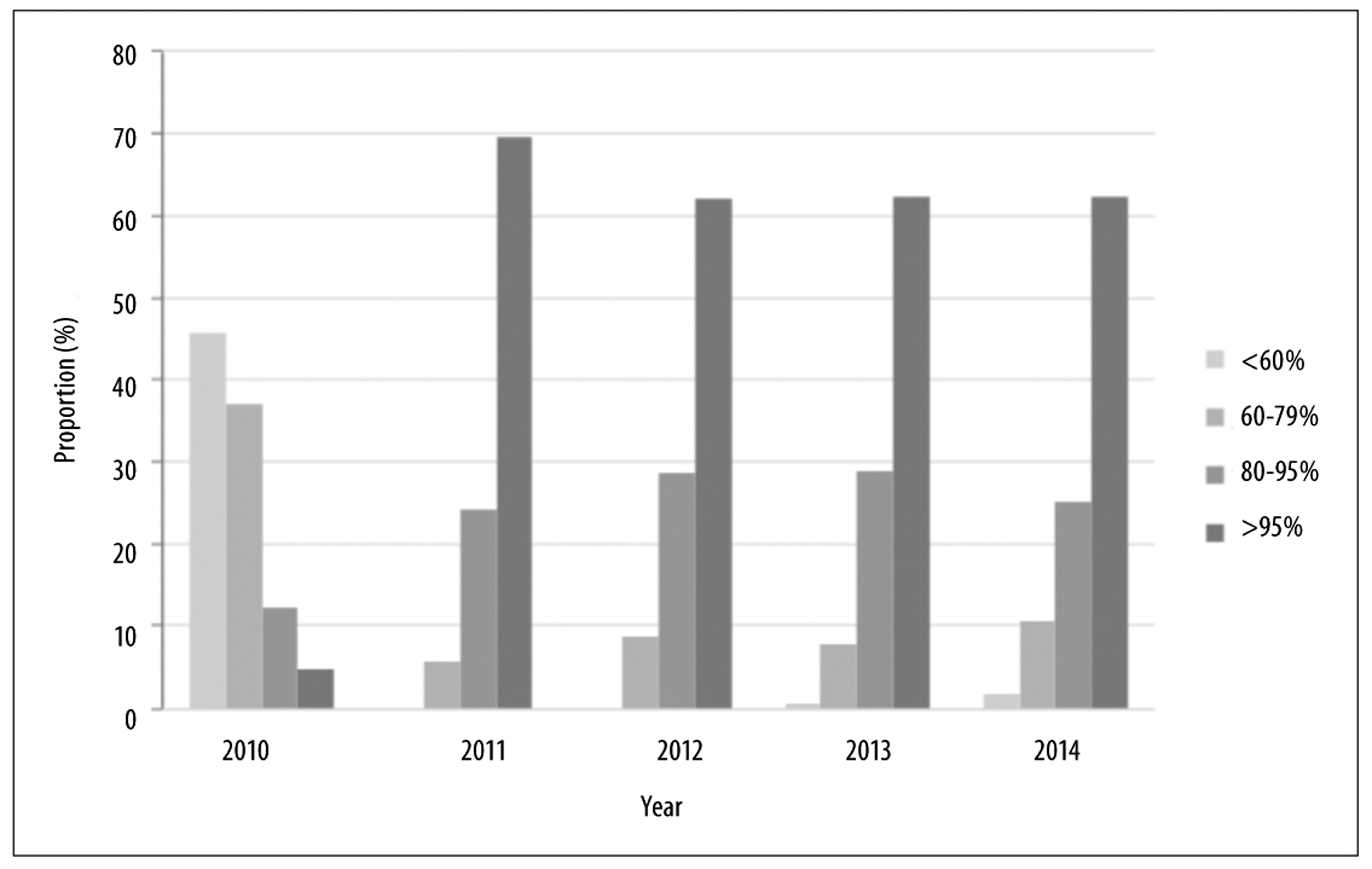
Santa Catarina is a state that has high vaccination coverage, although it is heterogeneously distributed between its municipalities. With regard to vaccination coverage over the PCV10 implantation period, we found that in 2010 only 3.4% of municipalities achieved coverage of 95% or more; while in subsequent years PCV10 coverage increased and became more homogenous (Figure 2).
Discussion
The study enabled the impact of PCV10 on reducing pneumonia hospitalization rates in Santa Catarina to be identified, with a 23.3% reduction in children under 1 year old and an 8.4% reduction in children aged 1 to 4 years old.
The greatest impact of vaccination on reducing hospitalization was found in children aged under 1 year old and in the Western region of the state, where the high hospitalization rates in 2006 affected the state’s overall rates. A study conducted in the state of Minas Gerais in 2016 also found regional variations and a 19% reduction in acquired pneumonia among children aged under 1 year old resident in the community of Alfenas, between 2007 and 2009, when compared to the period comprising 2011-2013 - these being periods before and after PCV10 implantation by the PNI.18
Notwithstanding, when assessing in detail all the regions of the state of Santa Catarina, the Serrano Plateau region did not have such a reduction. In addition to not having a significant reduction in the pneumonia hospitalization rate, this region also had lower vaccination coverage in the year the vaccine was implanted, as well as having the lowest number of hospital beds per 1,000 inhabitants and the state’s lowest HDI, in the period analyzed.
Primary Health Care, with its child care practices, such as monitoring, immunization and care for prevalent diseases, has priority actions for avoiding hospitalization due to pneumonia. However, when hospitalization is necessary, the number of hospital beds available is another important aspect to be verified.
The predominance of higher pneumonia hospitalization rates among children aged under 1 year old was described in the First Brazilian Consensus on Pneumonia,19 being particularly higher in those aged under 6 months old. The main prevention measures, including vaccination, should be concentrated on this age group.19
Since 2010, the PNI has sought to control diseases caused by Streptococcus pneumoniae and, in order to achieve this objective, vaccine coverage should be greater than 95%.8 The expectation that introducing the vaccine would lead to a significant reduction in pneumonia hospitalization rates in children aged under 5 years old was confirmed, albeit with distinct characteristics for each region. The fact is that a 10% increase in vaccination coverage enabled a 3% reduction in pneumonia hospitalization rates in children aged under 1 year old.
PCV10 was found to be effective in reducing cases of acute otitis media in Finland20 and also in reducing pneumococcal meningitis incidence rates, with 87.7% effectiveness and 81.3% effectiveness for pneumonia, thus also promoting a reduction in the hospitalization rate due to these causes in children.14,21-22 In our study in Santa Catarina, this reduction is more evident in children aged under 1 year old when compared to children aged between 1 and 4 years old. This fact may possibly be related to the time period analyzed in which PCV10 had only been implanted for 5 years.
Despite the vaccine only having impact on reducing hospitalizations due to pneumonia caused by Streptococcus pneumoniae serotypes or by serotypes corresponding to them,14 and present in its composition, Domingues et al. reported that in 2014 in ten Brazilian states, vaccination with three doses of PCV10 was associated with an 84% reduction (95%CI: 66;92) in invasive pneumococcal disease (IPD), through serotypes present in the vaccine (serotypes 1, 4, 5, 6B, 7F, 9V, 14, 18C, 19F and 23F), as well as a 78% reduction (95%CI: 41;92) in the serotypes corresponding to the vaccine (6A and 19A), when immunological cross-reactivity was reported.15,23 In turn, research conducted in Salvador in 2002, with strains isolated from the blood of children and adolescents hospitalized with pneumonia and fever without localized symptoms, identified predominance of serotypes 14, 5, 6A, 6B, 19F, 9V, 18C and 23F in that population. All these serotypes are present in the vaccine, thus demonstrating the importance of PCV10 in reducing pneumonia cases in Brazil.24 The choice of serotypes present in the vaccine is a determinant of its impact on reducing cases of the disease.
In addition to age, other factors such as climate and use of solid fuels in households may be associated with the occurrence of the disease.24 These factors, which were not analyzed in our study, could explain why there continued to be high hospitalization rates in the Serra Catarinense region, this being one of the state’s coldest regions where the use of solid fuels is predominant in 38.2% of households.25
Regarding other limitations of this study, we highlight the difference between the years of pre- and post-vaccination data collection, as well as the short period following vaccine implantation available for analysis. Nevertheless, comparison of mean rates for the period before and after vaccine implementation is very appropriate for data on the type of counting. When analyzing long time series, the effect of the different vaccination schedules can be controlled and the impact of group immunity can be assessed. Furthermore, it is important to highlight that comparative time series - before and after - assume homogeneity between the groups being compared, principally with regard to the vaccine not being used. Although 23-valent antipneumococcal polysaccharide vaccine and PCV7 vaccine have been used by the National Immunization Program in special situations, in children at greater risk of becoming ill and in children being cared for in private clinics, the number of children vaccinated before 2010 was very low from the population point of view.26
Another point to be considered is the recording of hospitalizations due to pneumonia, which may possibly be influenced by other respiratory tract infections not preventable with PCV10,27,28 such as pneumonias due to other causes and complications arising from type A influenza (H1N1).29 This scenario did not interfere with the results of our study, as there were few records of children being hospitalized due to H1N1 in Santa Catarina.30
Introducing PCV10 vaccine is not intended to eliminate pneumonia caused by Streptococcus pneumoniae but rather to reduce pneumonia hospitalization rates, as was demonstrated over the years covered by our study. It must be emphasized that PCV10 implantation and maintaining high coverage were not capable on their own of reducing hospital morbidity in all regions of Santa Catarina. The need therefore exists to identify the specificities of each population and to carry out other interventions beyond implanting PCV10 vaccine.
Based on the results described here, we concluded that pneumonia hospitalizations of children aged under 1 year old were predominant. There was an important reduction in hospitalizations in the period following PCV10 vaccine implantation, in the different regions of Santa Catarina. The distribution of the reduction was unequal, drawing attention to the Serrano Plateau region, where there were fewer beds per 1,000 inhabitants and lower regional HDI. This finding can contribute to PNI evaluation of PCV10 implantation strategies in the state of Santa Catarina, and also with regard to implantation of new vaccines such as PCV13.
REFERENCES
1. United Nations Children's Fund. Committing to child survival: a promise renewed - progress report 2015 [Internet]. New York: United Nations Children's Fund; 2015 [cited 2019 Oct 18]. 96 p. Available in: Available in: https://www.unicef.org/publications/files/APR_2015_9_Sep_15.pdf [ Links ]
2. Victora CG. Intervenções para reduzir a mortalidade infantil pré-escolar e materna no Brasil. Rev Bras Epidemiol. 2001 abr;4(1):1-67. doi: 10.1590/S1415-790X2001000100002. [ Links ]
3. United Nations Children's Fund. Levels & trends in child mortality: estimates developed by the UN Inter-agency Group for Child Mortality Estimation. Report 2015 [Internet]. New York: United Nations Children's Fund; 2015 [cited 2018 Oct 18]. 32 p. Available in: Available in: http://documents.worldbank.org/curated/pt/306381468000931160/pdf/99335-REVISED-9-8-AR-PUBLIC-disclosed-9-8-2015-Box393197B-IGME-Report-9-3-LR-Web.pdf [ Links ]
4. Rudan I, Boschi-Pinto C, Biloglav Z, Mulholland K, Campbell H. Epidemiology and etiology of childhood pneumonia. Bull World Health Organ. 2008 May;86(5):408-15. [ Links ]
5. Cardoso AM. A persistência das infecções respiratórias agudas como problema de Saúde Pública. Cad Saúde Pública. 2010 jul;26(7):1270-71. [ Links ]
6. Cesar JA, Horta BL, Gomes G, Shehadeh I, Chotolina J, Rangel L, et al. Utilização de serviços de saúde por menores de cinco anos no extremo sul do Brasil. Cad Saúde Pública. 2002;18(1):299-305. [ Links ]
7. Xu Z, Liu Y, Ma Z, Li S, Hu W, Tong S. Impact of temperature on childhood pneumonia estimated from satellite remotesensing. Environ Res. 2014 Jul;132:334-41. doi: 10.1016/j.envres.2014.04.021. [ Links ]
8. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Nota Informativa n° 149, de 7 de dezembro de 2015. Informa as mudanças no calendário nacional de vacinação para o ano de 2016 [Internet]. Brasília: Ministério da Saúde; 2015 [citado 2018 out 18]. Disponível em: Disponível em: http://www.aids.gov.br/pt-br/legislacao/nota-informativa-no-1492015 [ Links ]
9. Constenia CO. Economic impact of pneumococcal conjugate vaccination in Brazil, Chile, and Uruguay. Rev Panam Salud Pública. 2008 Aug;24(2):101-12. [ Links ]
10. Gomez JA, Tirado JC, Navarro Rojas AA, Castrejon Alba MM, Topachevskyi O. Cost-effectiveness and cost utility analysis of three pneumococcal conjugate vaccines in children of Peru. BMC Public Health. 2013 Oct;13:1025. doi: 10.1186/1471-2458-13-1025. [ Links ]
11. Pírez MC, Algorta G, Chamorro F, Romero C, Varela A, Cedres AM, et al. Changes in hospitalizations for pneumonia after universal vaccination with pneumococcal conjugate vaccines 7/13 valent and Haemophilus influenzae type b conjugate vaccine in a Pediatric Referral Hospital in Uruguay. Pediatr Infect Dis. 2014 Jul;33(7):753-9. doi: 10.1097/INF.0000000000000294. [ Links ]
12. Espinosa de los Monteros LE, Aguilar-Ituarte F, Jiménez-Juárez RN, Rodríguez-Suárez RS, Gómez Barreto D. Reemplazo de serotipos de Streptococcus pneumoniae en niños con vacuna conjugada antineumocóccica 7V en México. Salud Publica Mex. 2010 ene-feb;52(1):4-13. [ Links ]
13. Marti SG, Colantonio L, Bardach A, Galante J, Lopez A, Caporale J, et al. A cost-effectiveness analysis of a 10-valent pneumococcal conjugate vaccine in children in six Latin American countries. Cost Eff Resour Alloc. 2013 Aug;11(1):21. doi: 10.1186/1478-7547-11-21. [ Links ]
14. Abrão WM, Mello LM, Silva AS, Nunes AA. Impact of the antipneumococcal conjugate vaccine on the occurrence of infectious respiratory diseases and hospitalization rates in children. Rev Soc Bras Med Trop. 2015 Jan-Feb;48(1):44-9. doi: 10.1590/0037-8682-0007-2015. [ Links ]
15. Domingues CM, Verani JR, Montenegro Renoiner EI, Cunto Brandileone MC, Flannery B, et al. Effectiveness of ten-valent pneumococcal conjugate vaccine against invasive pneumococcal disease in Brazil: a matched case-control study. Lancet Respir Med. 2014 Jun;2(6):464-71. doi: 10.1016/S2213-2600(14)70060-8. [ Links ]
16. Hatisuka MFB, Arruda GO, Fernandes CAM, Marcon SS. Análise da tendência das taxas de internações por pneumonia bacteriana em crianças e adolescentes. Acta Paul Enferm. 2015 ago;28(4):294-300. doi: 10.1590/1982-0194201500051. [ Links ]
17. Ministério da Saúde (BR). Fundação Nacional de Saúde. Centro Nacional de Epidemiologia. Sistema de informações em saúde. Datasus [Internet]. 2012 [citado 2016 fev 3]. Disponível em: Disponível em: http://w3.datasus.gov.br/datasus/datasus.php?area=359A1B375C2D0E0F359G19HIJd2L2412M0N&VInclude=../site/infsaude.php [ Links ]
18. Silva SR, Mello LM, Silva AS, Nunes AA. Impacto da vacina antipneumocócica 10-valente na redução de hospitalização por pneumonia adquirida na comunidade em crianças. Rev Paul Pediatr. 2016 dez;34(4):418-24. doi: 10.1016/j.rpped.2016.02.003. [ Links ]
19. Corrêa JC. I Consenso brasileiro sobre pneumonia. J Pneumol. 1998 mar-abr;24(2):101-8. [ Links ]
20. Eskola J, Kilpi T, Palmu A, Jokinen J, Haapakoski J, Herva E, et al. Efficacy of a pneumococcal conjugate vaccine against acute otitis media. N Engl J Med. 2001 Feb;344(6): 403-9. [ Links ]
21. Hirose TE, Maluf EM, Rodrigues CO. Pneumococcal meningitis: epidemiological profile pre- and post-introduction of the pneumococcal 10-valent conjugate vaccine. J Pediatr. 2015 Mar-Apr;91(2):130-35. doi: 10.1016/j.jped.2014.07.002. [ Links ]
22. Scotta MC, Veras TN, Klein PC, Tronco V, Polack FP, Mattiello R, et al. Impact of 10-valent pneumococcal non-typeable Haemophilus influenzae protein D conjugate vaccine (PHiD-CV) on childhood pneumonia hospitalizations in Brazil two years after introduction. Vaccine. 2014 Jul;32(35):4495-9. doi: 10.1016/j.vaccine.2014.06.042. [ Links ]
23. Madhi AS. Vacina pneumocócica conjugada e variação da epidemiologia de meningite bacteriana infantil. J Pediatr. 2015 mar-abr;91(2):108-110. doi: 10.1016/j.jped.2014.11.001. [ Links ]
24. Passos BM, Simioni FJ, Deboni TL, Dalari BLSK. Características do consumo residencial de lenha e carvão vegetal. Floresta. 2016 jan-mar;46(1):21-9. doi: 10.5380/rf.v46i1.39714. [ Links ]
25. Po JY, FitzGerald JM, Carlsten C. Respiratory disease associated with solid biomass fuel exposure in rural women and children: systematic review and meta-analysis. Thorax. 2011 Mar;66(3):232-9. doi: 10.1136/thx.2010.147884. [ Links ]
26. Kupek E, Vieira ILV. O impacto da vacina pneumocócica PCV10 na redução da mortalidade por pneumonia em crianças menores de um ano em Santa Catarina, Brasil. Cad Saúde Pública. 2016 abr;32(3):e00131414. doi: 10.1590/0102-311X00131414. [ Links ]
27. Berezin EN. Vacinas pneumocócicas e pneumonias. J Pediatr. 2012 jan-fev;88(1):97-8. doi: 10.2223/JPED.2171. [ Links ]
28. Cillóniz C, Torre A. Entendimento da mortalidade em pneumonia pneumocócica bacterêmica. J Bras Pneumol. 2012 jul-ago;38(4):417-18. doi: 10.1590/S1806-37132012000400002. [ Links ]
29. Oliveira WK, Carmo EH, Penna GO, Kuchenbecker RS, Santos HB, Araujo WN, et al. Pandemic H1N1 influenza in Brazil: analysis of the first notified cases of influenza-like illness with severe acute respiratory infection (SARI). Euro Surveill. 2009 Oct;14(42):1-6. [ Links ]
30. Schuelter-Trevisol F, Dutra MC, Uliano EJM, Zandomênico J, Trevisol DJ. Perfil epidemiológico dos casos de gripe A na região sul de Santa Catarina, Brasil, na epidemia de 2009. Rev Panam Salud Publica. 2012;32(1):82-6. [ Links ]
*Article derived from the Ph.D. thesis entitled ‘Impact of conjugate vaccine against Streptococcus pneumoniae in children under Five years old in the state of Santa Catarina’, defended by Ilse Lisiane Viertel Vieira at the Santa Catarina Federal University Postgraduate Public Health Program in 2016.
Received: October 13, 2017; Accepted: September 30, 2018











 texto em
texto em