Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Epidemiologia e Serviços de Saúde
versión impresa ISSN 1679-4974versión On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.28 no.2 Brasília jun. 2019 Epub 27-Jun-2019
http://dx.doi.org/10.5123/s1679-49742019000200002
PROFILE OF NATIONAL HEALTH DATABASES
Special Tuberculosis Treatment Information System (SITE-TB) in Brazil: background, description and perspectives*
1Universidade de Brasília, Programa de Pós-Graduação em Medicina Tropical, Brasília, DF, Brasil
2Universidade Federal do Rio de Janeiro, Instituto de Estudos em Saúde Coletiva, Rio de Janeiro, RJ, Brasil
3Ministério da Saúde, Secretaria de Vigilância em Saúde, Brasília, DF, Brasil
4Fundação Oswaldo Cruz, Escola Nacional de Saúde Pública Sergio Arouca, Rio de Janeiro, RJ, Brasil
The Special Tuberculosis Treatment Information System (SITE-TB) arose mainly from the need to routinely monitor all persons with drug-resistant tuberculosis (DR-TB) in Brazil, as well as to qualify tuberculosis’ drug control. Developed by the Professor Hélio Fraga Reference Center and the Management Sciences for Health/Brazil Project, this online system was implemented in 2013 in all Brazilian states. In addition to DR-TB, the system registers people with drug-sensitive tuberculosis with special regimen indications, and those with nontuberculous mycobacterial infections identified by differential diagnosis of tuberculosis. All confirmed tuberculosis cases should be notified on the Notifiable Diseases Information System (SINAN). In situations where treatment with special regimens is necessary, the case is closed on SINAN and notified on SITE-TB. Professionals from tuberculosis reference centers report and monitor these cases on the system, as well as manage tuberculosis’ drugs.
Keywords: Information Systems; Tuberculosis, Multidrug-Resistant; Surveillance; Epidemiology
Introduction
Brazil is not considered a multidrug-resistant tuberculosis (MDR-TB) high burden country. Nevertheless, detecting these cases is a challenge for Brazil. Between 2014 and 2016, the MDR-TB detection rate for Brazil, according to WHO, ranged between 40% and 63%.1)-(3 The first reports of drug-resistant TB (DR-TB) in Brazil were in Rio de Janeiro city and are dated from 1958-1959.4 In the years that followed, similar reports were found in other Brazilian state capitals, revealing a problem on a national scale.4 Up until the 1990s, DR-TB surveillance had only been performed by the I National Survey of Anti-TB Drug Resistance, undertaken between 1995 and 1997.5
In the year 2000, upon the initiative of the Professor Hélio Fraga Reference Center (CRPHF), which, at the time, was part of the organizational structure of the Health Surveillance Secretariat (SVS) of the Ministry of Health, Brazil began passive epidemiological surveillance of MDR-TB, based on cases notified by the TB reference services.6 At that time, the definition of a confirmed MDR-TB case was patients with diagnosis confirmed by culture and identification of Mycobacterium tuberculosis and drug sensitivity test (DST) with resistance to rifampicin (R) and isoniazid (H) plus another drug. Probable MDR-TB cases were those with retreatment regimen failure (category III), with sensitivity to R or H.6 Diagnosis of resistance to first-line drugs (R and H) was done by the state-level reference laboratories, also known as Central Public Health Laboratories (LACEN), and by the CRPHF national-level reference laboratory.7
Data computerization, including people diagnosed since 1994, began with a typed Microsoft Office Excel® spreadsheet. In 2004, via a formal agreement between CRPHF and the Management Sciences for Health project (MSH/Brazil), funded by the United States Agency for International Development (USAID), the main tool for monitoring MDR-TB in Brazil was developed: the MR-TB System.8 The goals were to improve notification and monitor people with MDR-TB, to strengthen and decentralize the MDR-TB Epidemiological Surveillance Program, to enable evaluation and research to be carried out by keeping the information system updated and controlling the logistics of specific drugs at the central and peripheral levels.8 Considering the evolution of resistant TB diagnosis, the need to monitor all people with resistant TB and not just those with MDR-TB, the need to change the DR-TB surveillance model, including passive surveillance based on notifications, the need to control anti-TB drug dispensing, in addition to the need to produce epidemiological information about DR-TB in Brazil, CRPHF and the MSH/Brazil project enhanced the MR-TB System, which changed its name to Special Tuberculosis Treatment Information System (SITE-TB). Thus, since 2013, all persons with special TB treatment indication must be notified on this system. Special TB cases are considered those not having use of a first-line regimen prescribed because of the occurrence of severe adverse reactions, drug interaction, comorbidities or resistance to an anti-TB drug. Special cases also include notified cases of nontuberculous mycobacterial (NTM) infections identified by differential diagnosis of TB.9
In order to evaluate the evolution of resistance to anti-TB drugs, the II National Survey of Anti-TB Drug Resistance was conducted in 2006 and 2007. Comparison with the results of the I Survey identified an increase in primary resistance to H, from 4.4% to 6.0% (95% confidence interval [95%CI]: 5.2%;6.8%).10 The II survey results are used by WHO to estimate MDR-TB cases based on notified pulmonary cases.3
As a consequence of the II Survey, in 2008, during the IV National Tuberculosis Meeting held in Salvador city, an announcement was made to the scientific community that the addition of ethambutol (E) in the intensive phase of treatment had been approved and the reasons why. In December 2009, the sensitive TB treatment regimen was altered in Brazil, replacing the triple regimen - comprised of R, H and pyrazinamide - with the quadruple fixed-dose combination (FDC) regimen, which added E to the other drugs already used.11),(12
In 2011, the National Tuberculosis Control Program (NTP) published the Brazilian Guidelines for Tuberculosis Control, with new indications for DR-TB diagnosis and treatment. Following WHO recommendation, this manual changed the MDR-TB national definition to the universally used definition: resistance to at least R and H identified by DST, which may, at least temporarily, have caused increased system sensitivity as a direct consequence of the case definition alteration. Moreover, (i) mono-resistance cases were defined as those with resistance to one drug, (ii) poly-resistance cases were defined as those with resistance to two or more drugs, except R and H association, and (iii) extensively resistant cases were defined as resistance to R, H and another fluoroquinolone as well as to an injectable second-line drug.12
In 2012 and 2013, all the Brazilian states were trained for SITE-TB implantation and use. Exceptionally, the state of São Paulo notified people with MDR-TB only on SITE-TB, until October 2017. Since then, all people with TB being treated with special regimens were notified on SITE-TB.
The implantation of SITE-TB and progress with diagnosis strengthened DR-TB surveillance. From the beginning of the use of the system, NTP has been responsible for both the surveillance of people with TB with special treatment prescribed and for managing the system. One of the main actions undertaken by NTP in relation to the epidemiological situation of DR-TB in Brazil was the implantation of the rapid molecular test for TB (RMT-TB), which indicates whether there is resistance to R at the time of TB diagnosis.13 In 2015, the first year in which RMT-TB was used, one year after its implantation had begun,6 Brazil had the largest detection rate of people with MDR-TB (63%) in the last three years.1)-(3 The changes found in the epidemiological situation of DR-TB in the country enabled to monitor the RMT-TB implantation results, since SITE-TB implemented in 2013, and given its characteristics, was quickly updated to collect the necessary information.
Due to the implantation of RTM-TB, in 2014 NTP launched the Informative Note No. 9 containing recommendations on the use of the new technology, as well as on treatment of R resistant TB (RR-TB) diagnosed by RMT-TB, even before the DST result.14 In 2016, DR-TB treatment was updated again by Informative Note No. 8 and its new recommendations for MDR-TB treatment, which are in accordance with WHO recommendations,15 are still in force as at the date of the publication of this article (Figure 1).
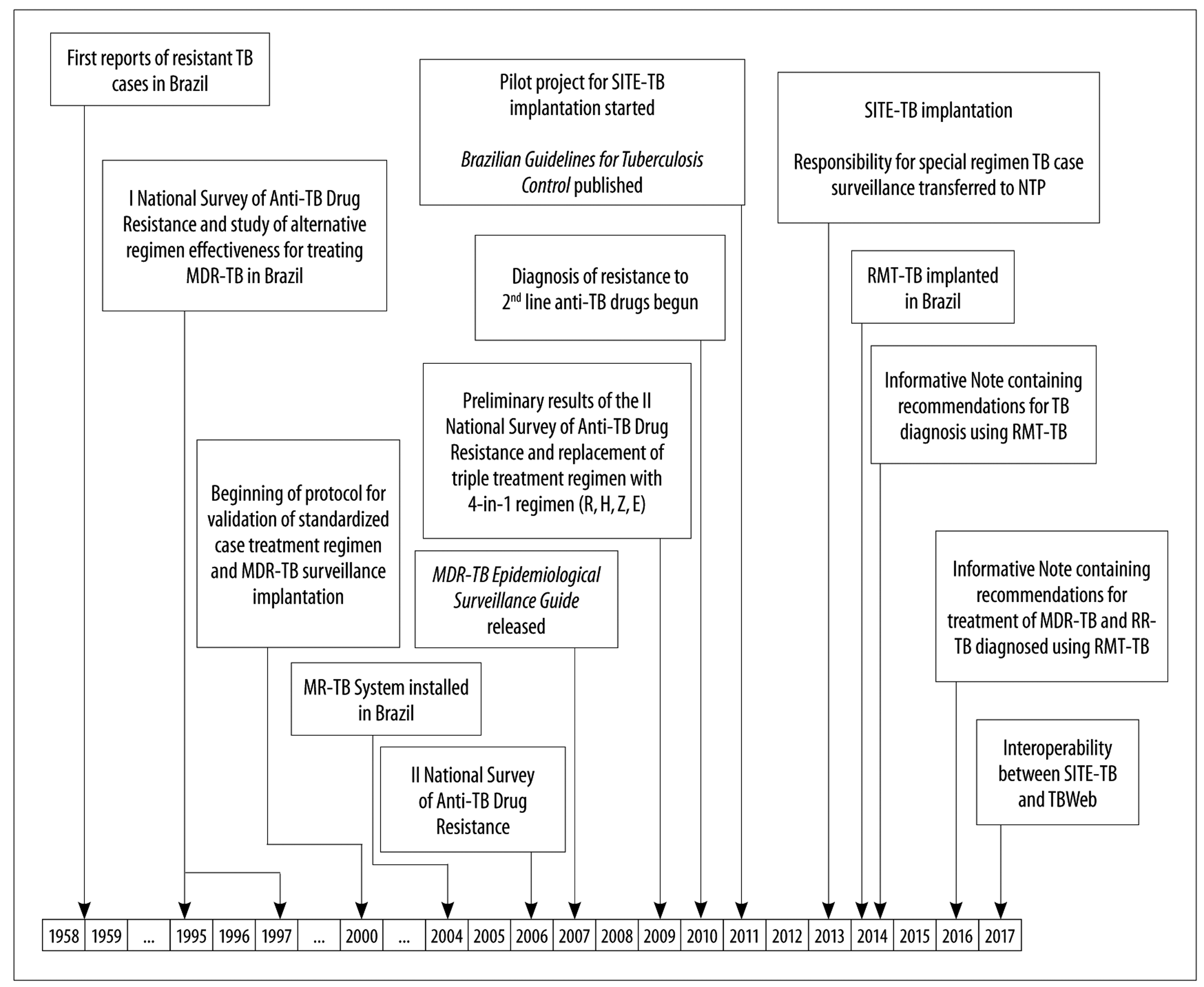
Legend:
TB: tuberculosis.
MDR-TB: multidrug-resistant tuberculosis.
MR-TB: multiresistant tuberculosis.
RR-TB: rifampicin resistant tuberculosis tested with RMT-TB.
RMT-TB: rapid molecular test for tuberculosis.
TBWeb: Tuberculosis Case Notification and Monitoring System.
SITE-TB: Special Tuberculosis Treatment Information System.
R: rifampicin.
H: isoniazid.
Z: pyrazinamide.
E: ethambutol.
PNCT: National Tuberculosis Control Program.
Anti-TB: anti-tuberculosis.
Figure 1 - Timeline of drug-resistant tuberculosis epidemiological surveillance implantation in Brazil
In addition to enabling the current situation of DR-TB in Brazil to be monitored, SITE-TB has been used for the development of operational research.16),(17 The system provides timely data that reveal the magnitude of DR-TB in Brazil and the quality of care offered to people with DR-TB. However, its functionalities and limitations have not been described. In addition to providing an historical report on the implantation of the system, this article aims to describe its main characteristics and perspectives for its use (Figure 2).
Characteristics of the Special Tuberculosis Treatment Information System (SITE-TB)
Data collection
All confirmed TB cases must be notified on the Notifiable Diseases Information System (SINAN). In situations in which treatment with a special regimen is necessary, the case must be closed on SINAN as regimen change, failure or DR-TB, and need to be notified on SITE-TB. Health professionals at TB reference services, either secondary or tertiary, notify these cases and monitor them on the system, as well as manage medication.9 It is an online tool that allows the direct filling in of data on the system. Alternatively, data is entered from notification sheets and follow-up cards.18
Information obtained during treatment follow-up must be entered at least monthly.9 Patient transfer in between services can be done via SITE-TB. Health professionals registered at the health center receiving the case can view the transfer on the system and accept it, accessing the data and providing treatment continuity.18
If NTM cases are entered mistakenly on SINAN, they must be closed as cases that have had their diagnosis changed and then notified on SITE-TB; or notified directly on SITE-TB, as long as they are identified by differential diagnosis of TB. Additionally, H or R mono-resistance cases must be closed on SINAN as DR-TB cases and, a posteriori, be notified on SITE-TB, regardless of the treatment regimen used. TB-HIV coinfection cases where R is replaced by rifabutin are not considered to be special treatment cases; nor are those having treatment complication with the basic regimen where the regimen is introduced with one drug at a time. These latter cases must only be notified on SITE-TB after the definitive special regimen has been defined (Figure 3).9
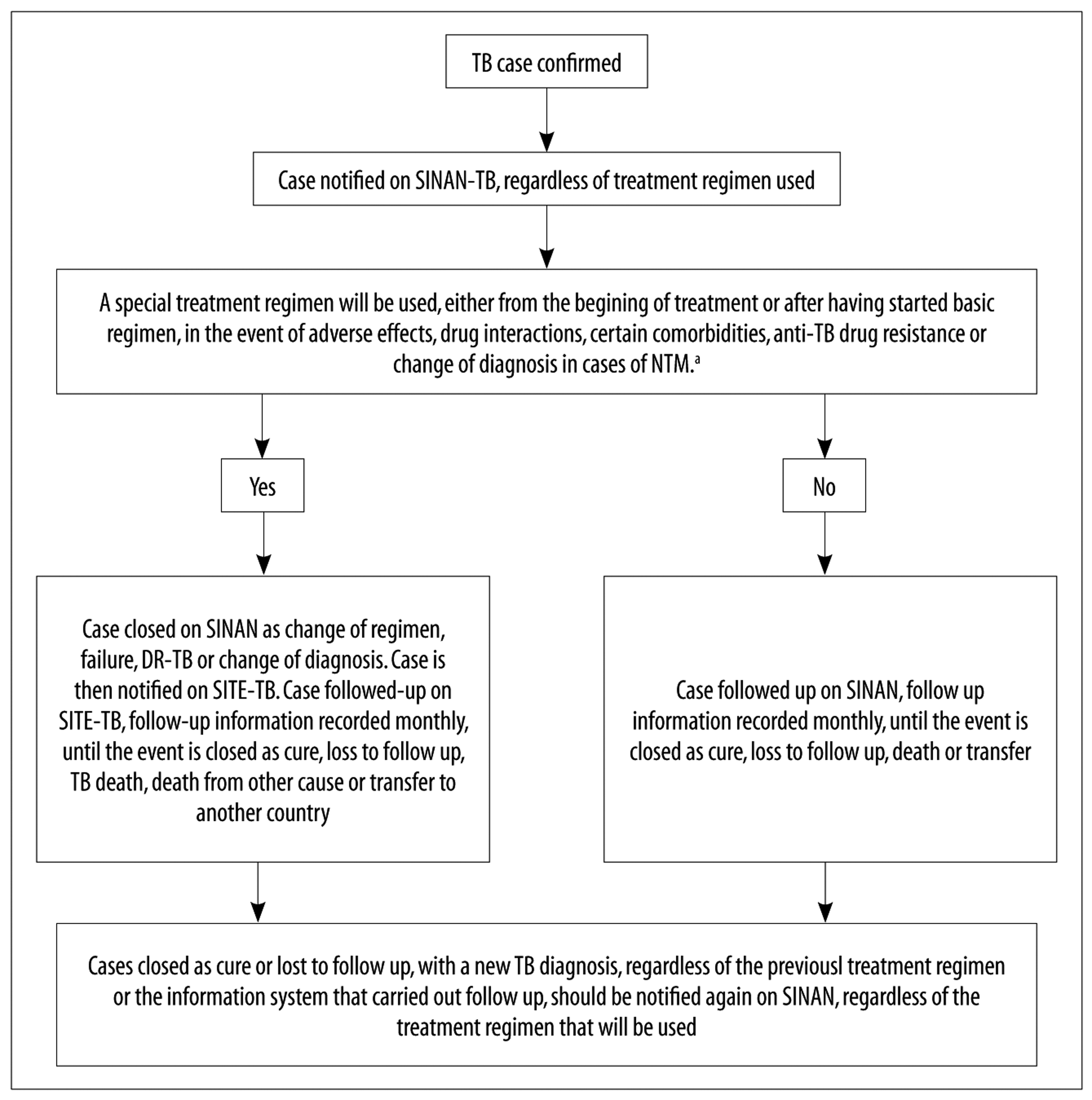
Legend:
TB: tuberculosis
DR-TB: drug-resistant tuberculosis
SINAN: Notifiable Diseases Information System
SITE-TB: Special Tuberculosis Treatment Information System
NTM: nontuberculous mycobacterial infections
Anti-TB: anti-tuberculosis
a) Cases with mono-resistance to isoniazid or rifampicin should be closed on SINAN as DR-TB and then notified on SITE-TB, regardless of the treatment regimen used. The following will not be considered to be special treatment (i) TB-HIV coinfection cases in which rifampicin is replaced by rifabutin and (ii) cases with some kind of complication during treatment with basic regimen which led to the drug-by-drug regimen. The latter cases should only be notified on SITE-TB after definition of the definitive special regimen.
Figure 3 - Flowchart for confirmed tuberculosis case notification on the Notifiable Diseases Information System (SINAN) and on the Special Tuberculosis Treatment Information System (SITE-TB)
Data processing
The thoroughness of notification and the treatment regimen proposed for cases notified on SITE-TB are assessed by a team of specialist physicians from several different Brazilian states. They are referred to as ‘validators’ and their activities are organized by NTP. The validator is responsible for checking data ranging from history of previous treatment, clinical characteristics, laboratory tests and treatment proposed. Once all the doubts of the validator are clarified, the patient’s record is validated, given an identification number and the system then authorizes the medication request, and data from this registry are made available for epidemiological analyses.18),(19 Every case notified more than once receives the same identifier number, plus an extra digit to identify the type of treatment.
In addition to case validation, SITE-TB also identifies inconsistencies based on predefined rules and uses red tags to inform the professionals who use the system when one of more of the following situations occur: no patient contacts assessed; missing follow-up culture; missing radiology control examination; missing SINAN number; late recording of medical consultations; pregnant woman taking risk medication; amikacin prescription; probable duplicate record. The tag is removed when the inconsistency is solved.
The system also generates blue tags which are included and removed by health professionals with the aim of monitoring patients or enabling communication between professionals, drawing attention to any given peculiarity.
Variables
When filling in the notification form, case type is selected first: drug sensitive TB with special regimen prescribed; DR-TB; or NTM. Figure 4 describes the blocks that comprise the notification form and those of the follow-up card, as well as the respective variables available.18

Legend:
SUS card: Brazilian National Health System service user card
SINAN: Notifiable Diseases Information System
DOT: directly observed treatment
RMT-TB: rapid molecular test for tuberculosis
ST: sensitivity test
DR-TB: drug-resistant tuberculosis
HIV: human immunodeficiency virus
AIDS: acquired immunodeficiency syndrome
Figure 4 - Variables of the notification form and follow up form of the Special Tuberculosis Treatment Information System (SITE-TB)
Uses
SITE-TB is used to notify and follow up on TB cases with special treatment prescribed and NTM cases. It is also used for drug management and epidemiological and operational analysis. Follow-up of notified people is done by accessing each individual’s record and filling in the follow-up card every time there is a medical consultation; for example, if an individual is in directly observed treatment, a record will be made of their response to treatment and how they are submitted to new examinations. It is also possible to follow up on records awaiting validation, pending, pending item related to, waiting to start treatment or in treatment, in the process of being transferred to another service, or closed cases, as well as by viewing records marked with red or blue tags.
The health professional responsible for controlling drugs can forward new orders, accompany pending orders, view backtracks of orders and stock transfers, dispense drugs and monitor drug dispensing. The system also provides standardized reports on drug movement and stock levels.
The analysis of the epidemiological and operational situation assists health professionals working at reference services and tuberculosis programs in monitoring the TB situation in their territory, with the aim of defining surveillance actions and decision-making, based on the data for their catchment area.
Updates
SITE-TB is a result of the improvement of the former MR-TB System. The main changes made to achieve its current status were:
- Increased MDR-TB case notification to include all TB-DR cases diagnosed: This change contributed to the knowledge of some case characteristics, DR-TB magnitude and distribution nationwide, support for the logistics and purchase of supplies and well as being able to know the treatment outcome of all DR-TB cases.
- Notification on SITE-TB of drug-sensitive TB cases with special regimens prescribed: This measure enabled to monitor all special regimens used in TB treatment.
- Correction of problems presented by the anti-TB drug management module: These corrections allowed more accurate monitoring of the drug stock in each TB reference service registered on the system, preventing failures in drug logistics and distribution, as well as losses due to expired medication.
All these alterations have contributed to the system generating more relevant data and, as a consequence, control of TB cases with special regimens prescribed.
In 2013, when SITE-TB was implanted nationwide, all MDR-TB cases notified on the MR-TB System migrated to SITE-TB. Since then, SITE-TB has undergone a large number of updates to correct errors or implement improvements. One of these changes worthy of mention is the inclusion of the variable for RMT-TB data collection, in addition to new standardized treatments for DR-TB which were defined following the implantation of rapid molecular testing for tuberculosis. NTP, as the national program responsible for these updates, maintains direct contact with the health professionals who use the system, thus facilitating identification of necessary updates. Having a systems developer working in NTP facilitates this work.
Coverage
In January 2018, the system had 1,589 health professionals registered with active password. They were located in 503 institutions and had diverse access profiles, defined according to their activities(Table 1). System viewing by each professional is limited to the records of the health center where they work; local health service managers can view the records notified by the health centers under their municipality, state or federal level responsibility.
Table 1 - Users, access profiles and number of institutions registered on the Special Tuberculosis Treatment Information System (SITE-TB), by Federative Unit and national region, Brazil, January 2018
| Federative Unit and national region | Users | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Access profiles | ||||||||||
| Notification with or without access to the drug module | Drug control with or without access to the case module | Other | Total | Institutions | ||||||
| N | % | N | % | N | % | N | % | N | % | |
| Rondônia | 5 | 83.3 | 1 | 16.7 | - | - | 6 | 0.4 | 4 | 0.8 |
| Acre | 7 | 87.5 | 1 | 12.5 | - | - | 8 | 0.5 | 4 | 0.8 |
| Amazonas | 11 | 84.6 | - | - | 2 | 15.4 | 13 | 0.8 | 4 | 0.8 |
| Roraima | 7 | 100.0 | - | - | - | - | 7 | 0.4 | 5 | 1.0 |
| Pará | 10 | 55.6 | 6 | 33.3 | 2 | 11.1 | 18 | 1.1 | 3 | 0.6 |
| Amapá | 11 | 84.6 | 2 | 15.4 | - | - | 13 | 0.8 | 4 | 0.8 |
| Tocantins | 12 | 66.7 | 6 | 33.3 | - | - | 18 | 1.1 | 3 | 0.6 |
| North | 63 | 75.9 | 16 | 19.3 | 4 | 4.8 | 83 | 5.2 | 27 | 5.4 |
| Maranhão | 10 | 47.6 | 4 | 19.0 | 7 | 33.3 | 21 | 1.3 | 3 | 0.6 |
| Piauí | 10 | 71.4 | 3 | 21.4 | 1 | 7.1 | 14 | 0.9 | 4 | 0.8 |
| Ceará | 17 | 58.6 | 11 | 37.9 | 1 | 3.4 | 29 | 1.8 | 8 | 1.6 |
| Rio Grande do Norte | 3 | 100.0 | - | - | - | - | 3 | 0.2 | 2 | 0.4 |
| Paraíba | 10 | 90.9 | 1 | 9.1 | - | - | 11 | 0.7 | 5 | 1.0 |
| Pernambuco | 25 | 43.9 | 22 | 38.6 | 10 | 17.5 | 57 | 3.6 | 21 | 4.2 |
| Alagoas | 16 | 66.7 | 7 | 29.2 | 1 | 4.2 | 24 | 1.5 | 8 | 1.6 |
| Sergipe | 9 | 81.8 | 2 | 18.2 | - | - | 11 | 0.7 | 3 | 0.6 |
| Bahia | 57 | 67.1 | 16 | 18.8 | 12 | 14.1 | 85 | 5.3 | 21 | 4.2 |
| Northeast | 157 | 61.6 | 66 | 25.9 | 32 | 12.5 | 255 | 16.0 | 75 | 14.9 |
| Minas Gerais | 87 | 48.9 | 44 | 24.7 | 47 | 26.4 | 178 | 11.2 | 46 | 9.1 |
| Espírito Santo | 8 | 57.1 | 5 | 35.7 | 1 | 7.1 | 14 | 0.9 | 7 | 1.4 |
| Rio de Janeiro | 183 | 59.8 | 97 | 31.7 | 26 | 8.5 | 306 | 19.3 | 68 | 13.5 |
| São Paulo | 107 | 54.0 | 66 | 33.3 | 25 | 12.6 | 198 | 12.5 | 81 | 16.1 |
| Southeast | 385 | 55.3 | 212 | 30.5 | 99 | 14.2 | 696 | 43.8 | 202 | 40.2 |
| Paraná | 98 | 61.6 | 50 | 31.4 | 11 | 6.9 | 159 | 10.0 | 63 | 12.5 |
| Santa Catarina | 104 | 71.2 | 42 | 28.8 | - | - | 146 | 9.2 | 79 | 15.7 |
| Rio Grande do Sul | 96 | 64.9 | 47 | 31.8 | 5 | 3.4 | 148 | 9.3 | 30 | 6.0 |
| South | 298 | 65.8 | 139 | 30.7 | 16 | 3.5 | 453 | 28.5 | 172 | 34.2 |
| Mato Grosso do Sul | 17 | 65.4 | 9 | 34.6 | - | - | 26 | 1.6 | 10 | 2.0 |
| Mato Grosso | 15 | 83.3 | 1 | 5.6 | 2 | 11.1 | 18 | 1.1 | 7 | 1.4 |
| Goiás | 11 | 47.8 | 1 | 4.3 | 11 | 47.8 | 23 | 1.4 | 6 | 1.2 |
| Distrito Federal | - | - | - | - | - | - | 35 | 2.2 | 4 | 0.8 |
| Midwest | 43 | 42.2 | 11 | 10.8 | 13 | 12.7 | 102 | 6.4 | 27 | 5.4 |
| Total | 946 | 59.5 | 444 | 27.9 | 164 | 10.3 | 1.589 | 100.0 | 503 | 100.0 |
When the system was being implanted, the state of São Paulo requested NTP to enable interoperability between SITE-TB and the Tuberculosis Case Notification and Monitoring System (TBWeb). São Paulo state uses the latter system to record notifications of people with TB. This regional process was finalized in September 2017. Since then, all patients with special regimens prescribed and notified on TBWeb migrate automatically to SITE-TB, thus reducing the workload of professionals at the São Paulo state TB reference services, since migration does not require double typing of cases.
Data quality and limitations
Most of the variables on the notification form are required fields that must be filled in. This ensures greater completeness and quality of the information generated by notifications. In 2016, among the main non-mandatory variable entry fields, ‘Extrapulmonary TB’ and ‘Nationality’ had more than 90% completeness, while ‘Probable place of infection’, had 76% completeness. Completeness for ‘SUS card number’ and ‘SINAN number’ was less than 50%. On the other hand, the required ‘Race/skin color’ and ‘Schooling’ field variables were filled in as unknown in 13% and 1% of cases, respectively. It is not possible to assess the completeness of the data generated by the follow-up cards since the system only exports data that has been filled in.
With regard to the data, one of the limitations to be taken into consideration lies in how comorbidities, associated factors and adverse drug effects are filled in. As these are variables that cannot be filled in simply by choosing ‘yes’ or ‘no’, when they are not filled in, it becomes impossible to differentiate between absence of comorbidity, associated factor or adverse effect, and lack of data collection.
Finally, the system does not export all data on included cases, such as treatment regime, in the case of individualized regimens, or changes to the initially proposed regimen.
Data analysis
Analysis of data entered onto the system can be done via standardized reports generated by the system’s data analysis tool which enables any indicator of interest to be calculated. Data can also be exported to Excel, enabling analysis with a variety of statistical packages.
SITE-TB has two functionalities intended to minimize duplicate data and ensure higher quality when analyzing the data recorded on it. The first is checking prior to notification whether there are possible previous records for the same patient based on patient name and date of birth. The second functionality is the red tag generated by SITE-TB itself to identify possible duplicate data, based on the patient’s name, their mother’s name and date of birth.
Cases closed because of change of diagnosis should be excluded from analysis.
It is important to note that data about directly observed treatment was only included in the second semester of 2017.
It is also necessary to evaluate the consistency with which the date of diagnosis is filled in, because it is important information for the epidemiological analysis of TB. Users frequently fill in this variable with the date of initial diagnosis of the disease rather than the date of resistant diagnosis, failure or need to change regimens.
Access
The system is available at the following URL: sitetb.saude.gov.br
Health professionals involved in diagnosis and treatment, both of people with TB on special regimens and also cases of NTM, have access to SITE-TB when authorized by the state-level TB control program coordinators. Researchers interested in unidentified data generated by the system can file requests via the Electronic Citizens’ Information Service System (e-SIC) at this URL:
http://www.acessoainformacao.gov.br/sistema/site/primeiroacesso.html
National data with patient identification should be requested according to the procedures of the Health Surveillance Secretariat of the Ministry of Health (SVS/MS) via the following URL: http://portalms.saude.gov.br/vigilancia-em-saude/indicadores-de-saude/bancos-de-dados-nominais
Municipal and state-level data should be requested from the respective municipal or state Health Departments.
Final considerations
A satisfaction survey conducted in 2016 demonstrated that 80% of health professionals who use SITE-TB are satisfied with it.20 Its flexibility enables frequent updating, incorporation of new improvements to the quality of its standardized reports and its data analysis tool, as well as revision of the notification and follow-up forms.
The implantation of SITE-TB, together with the incorporation of RMT-TB in Brazil, are two of the main actions undertaken to qualify surveillance of TB cases with special regimens prescribed. In addition to supporting disease control actions, the individualized data registered on the system comprise a national database which can be analyzed and used in observational studies and which is accessible to researchers interested in and dedicated to the issue. Drugs control made available to each individual is one of the ways of sustaining controlled resistance to anti-TB drugs, thus keeping Brazil off the list of countries with a high burden of DR-TB cases.3
The progress made this far needs to be accompanied by improvements in the public health care network, which includes the main actors of tuberculosis surveillance, in the sense of continuous enhancement of heath surveillance in Brazil. SUS Primary Care, which coordinates and organizes the actions and services provided by the public health service network,21 should be prepared to identify special cases in need of reference services. Primary Care should also take on the responsibility to follow-up of treatment of patients with drug-sensitive TB for whom the basic regimen has been prescribed, thus enabling the other health care levels - secondary and tertiary - to be dedicated to care of special cases.9
The laboratory network needs to be strengthened in order to meet the TB diagnosis algorithms by offering RMT-TB, culture test and DST to indicated cases. Other molecular tools, apart from RMT-TB, need to be incorporated into the Public Health laboratories, in order to enable, for example, identification of possible aggregated cases in populations at high risk of DR-TB, apart from the detection and mobility of new potentially resistant strains in Brazil. Genome sequencing can be useful for mapping the transmission chain and investigating outbreaks in populations and environments at greater risk of developing TB.22 All of this information needs to be present on SITE-TB so that this system can be used even more as the official source of anti-TB drug resistance surveillance in Brazil.
By strengthening the Public Health care network, qualifying surveillance tools and incorporating molecular epidemiology into TB control actions, it will be possible to detect all estimated TB cases, qualify and center care on people with TB and, as a consequence, meet the targets set by both the Sustainable Development Goals and the National Plan to End Tuberculosis as a Public Health Problem in Brazil.23),(24
REFERENCES
1. World Health Organization. Global tuberculosis report 2016 [Internet]. Geneva: World Health Organization; 2016 [cited 2019 Feb 19]. Available from: Available from: https://www.who.int/tb/publications/global_report/en/ [ Links ]
2. World Health Organization. Global tuberculosis report 2015 [Internet]. Geneva; World Health Organization; 2015 [cited 2019 Feb 19]. Available from: Available from: https://apps.who.int/iris/handle/10665/191102 [ Links ]
3. World Health Organization. Global tuberculosis report 2017 [Internet]. Geneva; World Health Organization; 2017 [cited 2019 Feb 19]. 147 p. Available from: Available from: https://www.who.int/tb/publications/global_report/gtbr2017_main_text.pdf [ Links ]
4. Maciel MS, Mendes PD, Gomes AP, Siqueira-Batista R. A história da tuberculose no Brasil: os muitos tons (de cinza) da miséria. Rev Soc Bras Clin Med [Internet]. 2012 maio-jun [citado 2017 nov 3];10(3):226-30. Disponível em: Disponível em: http://files.bvs.br/upload/S/1679-1010/2012/v10n3/a2886.pdf [ Links ]
5. Braga JU, Barreto AMW, Hijjar MA. Inquérito epidemiológico da resistência às drogas usadas no tratamento da tuberculose no Brasil 1995-97 - IERDTB. Parte I: aspectos metodológicos. Bol Pneumol Sanit [Internet]. 2002 jun [citado 2019 fev 19];10(1):65-73. Disponível em: Disponível em: http://scielo.iec.gov.br/pdf/bps/v10n1/v10n1a10.pdf [ Links ]
6. Dalcolmo MP, Andrade MKN, Picon PD. Multiresistant tuberculosis in Brazil: history and control. Rev Saúde Pública [Internet]. 2007 set [citado 2019 fev 19];41(suppl 1):34-42. Disponível em: Disponível em: http://www.scielo.br/pdf/rsp/v41s1/6570.pdf . Doi: 10.1590/S0034-89102007000800006 [ Links ]
7. Barreto AMW, Martins FM. Laboratório de tuberculose do CRPHF quinze anos de atividades. Bol Pneumol Sanit [Internet]. 1999 dez [citado 2019 fev 19];7(2):41-51. Disponível em: Disponível em: http://scielo.iec.gov.br/pdf/bps/v7n2/v7n2a05.pdf [ Links ]
8. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Centro de Referência Professor Helio Fraga. Projeto MSH. Tuberculose multirresistente: guia de vigilância epidemiológica [Internet]. Rio de Janeiro: Ministério da Saúde; 2007 [citado 2019 fev 19]. Disponível em: Disponível em: http://www5.ensp.fiocruz.br/biblioteca/dados/txt_670024370.pdf [ Links ]
9. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Guia de vigilância em saúde [Internet]. 2. ed. Brasília: Ministério da Saúde; 2017 [citado 2019 fev 19]. 750 p. Disponível em: Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2017/outubro/06/Volume-Unico-2017.pdf [ Links ]
10. Kritski AL. Multidrug-resistant tuberculosis emergence: a renewed challenge. J Bras Pneumol [Internet]. 2010 Mar-Apr [cited 2019 Feb 19];36(2):157-8. Available from: Available from: http://www.scielo.br/pdf/jbpneu/v36n2/en_v36n2a01.pdf . Doi: 10.1590/S1806-37132010000200001 [ Links ]
11. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica.. Nota técnica sobre as mudanças no tratamento da tuberculose no Brasil para adultos e adolescentes - Versão 2 [Internet]. Brasília: Ministério da Saúde ; 2009 [citado 2019 fev 19]. Disponível em: Disponível em: http://www1.saude.rs.gov.br/dados/1293729099101Nota%20T%E9cnica%20-%202%AA%20vers%E3o%20%28corrigida%20em%2022-10%29.pdf [ Links ]
12. Brasil. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual de recomendações para o controle da tuberculose no Brasil [Internet]. Brasília: Ministério da Saúde ; 2011 [citado 2019 fev 19]. 284 p. Disponível em: Disponível em: http://bvsms.saude.gov.br/bvs/publicacoes/manual_recomendacoes_controle_tuberculose_brasil.pdf [ Links ]
13. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Rede de teste rápido para tuberculose: primeiro ano de implantação [Internet]. Brasília: Ministério da Saúde ; 2015 [citado 2019 fev 19]. 63 p. Disponível em: Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2016/janeiro/19/rtr-tb-15jan16-isbn-web.pdf [ Links ]
14. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Nota informativa CGPNCT/DEVEP/SVS/MS nº 09, de 11 de dezembro de 2014. Recomendações sobre o diagnóstico da tuberculose por meio do teste rápido molecular para tuberculose [Internet]. Brasília: Ministério da Saúde ; 2014 [citado 2019 fev 19]. Disponível em: Disponível em: http://www.riocomsaude.rj.gov.br/Publico/MostrarArquivo.aspx?C=tOjQoGNWGNw%3D [ Links ]
15. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Nota informativa CGPNCT/Devit/SVS/MS nº 08/2016. Brasília: Ministério da Saúde ; 2016. [ Links ]
16. Fregona G, Cosme LB, Moreira CMM, Bussular JL, Dettoni VV, Dalcolmo MP, et al. Risk factors associated with multidrug-resistant tuberculosis in Espírito Santo, Brazil. Rev Saúde Pública [Internet]. 2017 [cited 2018 Feb 13];51. Available from: Available from: http://www.scielo.br/scielo.php?script=sci_abstract&pid=S0034-89102017000100230&lng=en&nrm=iso&tlng=en . Doi: 10.1590/s1518-8787.2017051006688 [ Links ]
17. Bastos ML, Cosme LB, Fregona G, Prado TN, Bertolde AI, Zandonade E, et al. Treatment outcomes of MDR-tuberculosis patients in Brazil: a retrospective cohort analysis. BMC Infect Dis [Internet]. 2017 Nov [cited 2019 Feb 19];17(1):718. Available from: Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5686842/ . Doi: 10.1186/s12879-017-2810-1 [ Links ]
18. Ministério da Saúde (BR). Manual do usuário do Sistema de Informação de Tratamentos Especiais de Tuberculose - SITE-TB [Internet]. Brasília: Ministério da Saúde ; 2017 [citado 2017 dez 12]. Disponível em: Disponível em: http://sitetb.saude.gov.br/download.html [ Links ]
19. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. O controle da tuberculose no Brasil: avanços, inovações e desafios. Bol Epidemiol [Internet]. 2014 [citado 2019 fev 19];45(2):1-13. Disponível em: Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2014/abril/10/Boletim-Tuberculose-2014.pdf [ Links ]
20. Ministério da Saúde (BR). Systems for improved access to pharmaceuticals and services. Brasil: pesquisa de avaliação dos usuários do SITETB. Brasília: Ministério da Saúde ; 2016. [ Links ]
21. Brasil. Ministério da Saúde. Portaria MS/GM no 2.436, de 21 de setembro de 2017. Aprova a Política Nacional de Atenção Básica, estabelecendo a revisão de diretrizes para a organização da Atenção Básica, no âmbito do Sistema Único de Saúde (SUS) [Internet]. Diário Oficial da União, Brasília (DF), 2017 set 9 [citada 2018 mar 25]; Seção I:68. Disponível em: Disponível em: http://bvsms.saude.gov.br/bvs/saudelegis/gm/2017/prt2436_22_09_2017.html [ Links ]
22. Dara M, Zachariah R. Ending tuberculosis calls for leaving no one behind. Lancet Infect Dis [Internet]. 2018 Apr [cited 2019 Feb 19];18(4):365-6. Available from: Available from: https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(17)30746-6/fulltext . Doi: 10.1016/S1473-3099(17)30746-6 [ Links ]
23. United Nations. Transforming our world: the 2030 agenda for sustainable development [Internet]. Geneva: United Nations; 2015 [cited 2019 Feb 19]. Available from: Available from: https://sustainabledevelopment.un.org/post2015/transformingourworld [ Links ]
24. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Brasil livre da tuberculose: plano nacional pelo fim da tuberculose como problema de saúde pública [Internet]. Brasília: Ministério da Saúde ; 2017 [citado 2019 fev 19. 52 p. Disponível em: Disponível em: http://bvsms.saude.gov.br/bvs/publicacoes/brasil_livre_tuberculose_plano_nacional.pdf [ Links ]
*Article from a doctoral thesis titled "Tuberculosis Drug Resistant in Brazil" presented by the author Patricia Bartholomay in the Graduate Program in Tropical Medicine, Faculty of Medicine, University of Brasília. Financed with public funds, made available by the Coordination for the Improvement of Higher Education Personnel (CAPES), Ministry of Education (MEC).
Received: November 08, 2018; Accepted: February 11, 2019










 texto en
texto en 
 Curriculum ScienTI
Curriculum ScienTI