Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Epidemiologia e Serviços de Saúde
versión impresa ISSN 1679-4974versión On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.28 no.3 Brasília set. 2019 Epub 15-Oct-2019
http://dx.doi.org/10.5123/s1679-49742019000300009
ORIGINAL ARTICLE
Evaluation of the national meningococcal disease surveillance system: Brazil, 2007-2017
1Ministério da Saúde, Secretaria de Vigilância em Saúde, Brasília, DF, Brasil
Objective:
to evaluate meningococcal disease (MD) surveillance from 2007-2017, according to its attributes.
Methods:
this was a descriptive study of the ‘timeliness’, ‘representativeness’, ‘simplicity’, ‘flexibility’, ‘data quality’, ‘acceptability’ and ‘usefulness’ attributes of the system, based on Centers for Disease Control and Prevention guidelines; we used National Notifiable Diseases Information System (SINAN) data, with onset of symptoms between 2007-2017.
Results:
the system was found to be complex with eight case definitions and six laboratory confirmations; flexible, with adequate description of epidemiological changes; good chemoprophylaxis, active tracing and serogrouping completeness; low acceptability with chemoprophylaxis found in less than 70.0% of records; timely, with excellent investigation, closure and collection of cerebrospinal fluid; representative, in its description of MD in Brazil; useful, adequately guiding control actions.
Conclusion:
different clinical presentations of MD and the need for rapid case management interfere with the system’s acceptability and complexity; the latter, however, is useful for epidemiological analysis.
Keywords: Meningococcal Infections; Program Evaluation; Epidemiologic Surveillance Services; Information Systems
Introduction
Meningococcal disease (MD) is a severe infection caused by the bacterium Neisseria meningitidis (meningococcus), which can rapidly lead to death if not treated immediately - in less than 24 hours - and has a significant epidemic potential.1,2
MD has a broad clinical spectrum and an individual with MD can range from being healthy to having fulminating meningococcemia. Its main clinical forms are meningococcal meningitis (MM), meningococcemia (MCC) and meningitis with meningococcemia (MM+MCC). MM results from hematogenic dissemination, occurs in around 50% of infected patients and is similar to other types of purulent meningitis. In turn, MCC is characterized by sudden fever and petechial rash, which can lead to purpura fulminans occurring in 5% to 20% of patients with the disease.1,3
DM occurs all over the world, with considerable incidence differences depending both on the region affected and the most frequent serogroup.4 Worldwide, it is estimated that 500,000 MD cases occur each year, causing 50,000 deaths, with serogroup A being the main cause due, among other factors, to its high incidence in the so called “African Meningitis Belt”.5 Based on its polysaccharide capsule antigenic composition, N. meningitidis is classified into 12 different serogroups, whereby serogroups A, B, C, Y, W and X account for almost all MD cases, infecting only humans.4 In Brazil, average MD incidence between 2007 and 2013 was 1.4 cases per 100,000 inhabitants, with serogroup C accounting for most cases.6
MD is endemic in Brazil, with sporadic outbreaks in municipal territories. Its behavior is also seasonal, with incidence increasing in winter and decreasing in summer, even in epidemic situations. The disease has also a secular tendency, that is to say, incidence over a given period appears to be similar to incidence in previous periods.7,8
Antibiotic chemoprophylaxis and immunization are recommended as preventive and control measures. Chemoprophylaxis is recommended for close contacts of confirmed cases and should be carried out as soon as possible, in order to prevent secondary cases, which are rare but manifest themselves 48 hours after the first case.9 The second measure, immunization, is the most effective way to prevent the disease and available vaccines are serogroup-specific or serosubtype-specific. In Brazil, meningococcal C conjugate vaccine (MenC) was implemented in 2010, for the immunization of children under 2 years of age, as part of the National Immunization Program (PNI) child immunization schedule. This vaccine is also used to control outbreaks by means of vaccine blocking or mass vaccination.10,11
MD in Brazil is a very important disease for public health given its potential to cause outbreaks. For this reason its notification has been compulsory in Brazilian territory since the publication of Health Ministry Ordinance GM/MS No. 1,271, dated 6 June 2014. This ordinance also requires health authorities to put more effort into monitoring and controlling infection. Implementation of national surveillance systems aim to ensure efficient actions to manage diseases and conditions. Evaluation of a health surveillance system therefore seeks to ensure that its main objectives will be reached, with rational and efficient use of available public resources.12,13
As MD is recognized as a severe Public Health problem, systematic evaluation of its epidemiological surveillance system is necessary. This system should be able to: produce data to contribute to knowing the disease’s epidemiological and etiological profile; monitor the epidemiological situation; detect outbreaks in advance; establish measures for prevention and control; and characterize circulating serogroups.7-9
This study aimed to evaluate MD epidemiological surveillance between 2007 and 2017, based on the system’s qualitative, quantitative and usefulness attributes.
Methods
We prepared an evaluation study based on the United States Centers for Disease Control and Prevention document “Updated Guidelines for Evaluating Public Health Surveillance Systems: recommendations from the guidelines working group”,14 with analysis of the system’s quantitative attributes (timeliness and representativeness) and qualitative attributes (simplicity, flexibility, data quality, and acceptability) and its usefulness.
We used data from the Notifiable Diseases Information System (SINAN) Net version, providing information on confirmed MD cases with onset of symptoms between 2007 and 2017. This information was used to calculate the incidence coefficient per year and per Federative Unit, as well as to calculate measurements expressed in percentages, according to the evaluation criteria for each attribute evaluated. The system was updated for the last time and data were retrieved for the study in May 2018.
When evaluating the simplicity attribute, we used descriptive data of the surveillance system, including analysis of pertinent case definitions, aggregated laboratory methods and control measures. We analyzed simplicity subjectively, taking system classification as being either simple or complex as our parameter for doing so.
The “flexibility” attribute was evaluated based on changes in annual meningococcal disease incidence coefficients arising due to new surveillance interventions implemented in the period studied. This study considers new interventions to be the decentralization of the polymerase chain reaction (PCR) technique to the state-level Central Public Health Laboratories (Lacen) and the introduction of the meningococcal C conjugate vaccine on the national immunization schedule. Both these processes started in 2010. If the MD surveillance system was able to properly detect and describe changes in annual MD incidence coefficients owing to the interventions described above, it was classified as flexible.
The “data quality” attribute was evaluated based on analysis of completeness of certain essential fields, both compulsory and non-compulsory, of the MD notification forms. These fields were chosen because they reflect essential aspects regarding the quality of health surveillance team intervention when MD secondary cases occur in the community, in order to avoid the spread of the disease among the population. Well completed data mean that surveillance team intervention was adequate. We selected “chemoprophylaxis use” for analysis from among the compulsory fields, and the “MenC immunization”, “contact with suspected case”, “secondary case” and “serogroup” variables from among the non-compulsory fields. In the case of analysis of chemoprophylaxis use, “good” data quality was taken to be average completeness of 100% for the period analyzed (2007 to 2017), since the information system does not save notification data if this information is missing. In the case of the other non-compulsory variables, the parameters established were: excellent = ≥90.0%; good = 80.0 to 89.9%; regular = 70.0 to 79.9%; and poor = <70.0%. It is important to recall that the information system accepts notification even when non-compulsory variable data is missing, although these data are essential to MD surveillance, both for evaluating intervention quality and also for building epidemiological and operational indicators. For this reason a good percentage of field completeness is needed.15,17 The evaluation criteria we used were: three or more fields evaluated as “good” classified the system as having “good quality”, while fewer than three fields evaluated as “good” classified the system as having “poor quality”.
The acceptability attribute was evaluated by average timeliness - over the period studied - of chemoprophylaxis for close contacts of suspected MD cases, which should be done in up to 48 hours from the date of notification. Therefore, we analyzed the “notification date” and “chemoprophylaxis date” fields of the investigation form, with the aim of estimating the period between notification and contacts receiving chemoprophylaxis procedures. Contact chemoprophylaxis necessarily implies a set of measures to be taken by health teams: investigation of the total contacts to be medicated; chemoprophylaxis medication stock control and use; monitoring secondary cases or cases with medication side effects; and active tracing of no-show contacts. Correct recording of the intervention on the notification form informs as to chemoprophylaxis timeliness. It is an important indicator of health professionals’ readiness to undertake with expertise activities recommended by MD surveillance, whilst also describing the system’s acceptability for these professionals.15,17 As an evaluation criterion, we considered the system to be highly acceptable if timeliness of average chemoprophylaxis in the period studied was greater than or equal to 70.0%, given that timely delivery will not always be possible due to operational difficulties related to active tracing (remote or hard-to-reach areas), staff difficulties (not trained or insufficient number) and technology difficulties (lack of medication supplies or kits for laboratory exams).6,8
When analyzing the “timeliness” attribute we observed:
period of time between onset of symptoms and case being admitted to hospital (up to 24 hours);
period of time between hospital admission and case notification to the surveillance service (up to 24 hours);
period of time between notification and commencement of investigation (up to 24 hours);
period of time between hospital admission and lumbar puncture, for cases that had cerebrospinal fluid (CSF, also known as liquor) collection (up to 24 hours);
period of time between notification and chemoprophylaxis for close contacts (up to 48 hours); and
period of time between case notification and case closure (up to 60 days).
The parameters we used to analyze system "timeliness” were average percentages calculated in the study period, with timeliness being defined as: excellent, when greater than or equal to 90.0%; good, between 70.0% and 89.9%; regular, between 50.0% and 69.9%; and poor when less than 50.0%. The system was considered to meet the evaluation criteria set by the Meningitis Surveillance Systems Data Quality Evaluation Handbook for timeliness when three or more fields were rated as “good”, otherwise it was not considered to be timely.15-17
The “representativeness” attribute was evaluated based on the capacity of the system to precisely describe (time, place and person) the MD event in Brazil from SINAN data in the defined study period, focusing on the calculation of MD incidence coefficients stratified by year, Federative Unit, administrative region and most affected age groups.
The system’s “usefulness” attribute was evaluated according to its provision of indicators that were efficient in describing MD magnitude in Brazil and in determining and guiding MD control and monitoring measures.14 As per the Health Surveillance Guide, published by the Brazilian Ministry of Health in 2017, the objectives of the MD surveillance system are:9
To monitor the epidemiological situation of meningococcal disease in Brazil.
To detect outbreaks in advance.
To guide the use and evaluate the effectiveness of prevention and control measures.
To monitor the prevalence of circulating N. meningitidis serogroups and serotypes.
To monitor the bacterial resistance profile of N. meningitidis strains.
To produce and disseminate epidemiological information.
If the MD surveillance system is capable of providing relevant epidemiological data to fulfill these objectives, it will be considered useful.
For data analysis and processing we used Tabwin® 3.6b, Epi Info™ 7.1.5, Microsoft® Excel 2010 and Recklink® 3.1.6.
As this was an epidemiological surveillance evaluation study based on analysis of secondary data retrieved from an anonymous national database, it was exempt from appraisal by a Research Ethics Committee, whilst duly complying with the ethical requirements of National Health Council Resolution CNS No. 466, dated 12 December 2012; and Resolution CNS No. 510, dated 7 April 2016. Access to the database was granted by institutional consent. The study results were aggregated and are presented as follows.
Results
We studied 23,930 confirmed MD cases in Brazil with onset of symptoms between 2007 and 2017. The system uses three suspected case definitions based on clinical evaluation criteria, four confirmed case definitions based on clinical laboratory criteria, and one discarded case definition, based on confirmed case exclusion criteria (if the suspected case does not meet the MD confirmation criteria, it is automatically discarded).14
Suspected MD cases also have six different models of laboratory confirmation (chemical and cytological, bacterioscopy, counterimmunoelectrophoresis, latex agglutination, PCR or culture), mostly performed by municipal and state health services.
Measures to control new cases depend on medication and immunobiologicals, with 48-hour chemoprophylaxis being used for sporadic case contacts; in the event of an MD outbreak, vaccine blocking is carried out in addition to chemoprophylaxis.
MD epidemiological surveillance has high case detection sensitivity, using eight case definitions, together with six different models of laboratory confirmation, as well as a system of control and prevention measures using medication and immunobiologicals. Because of these characteristics, MD epidemiological surveillance was considered to be complex.
As of the year 2010, the introduction of MenC as part of the Brazilian basic immunization schedule reduced MD serogroup C incidence in the population, and the decentralization of the Polymerase Chain Reaction (PCR) technique to state-level laboratories reduced incidence of MD with no serogroup identification (Figure 1). The epidemiological surveillance system was considered to be flexible, as it was able to describe these changes resulting from the interventions that took place.
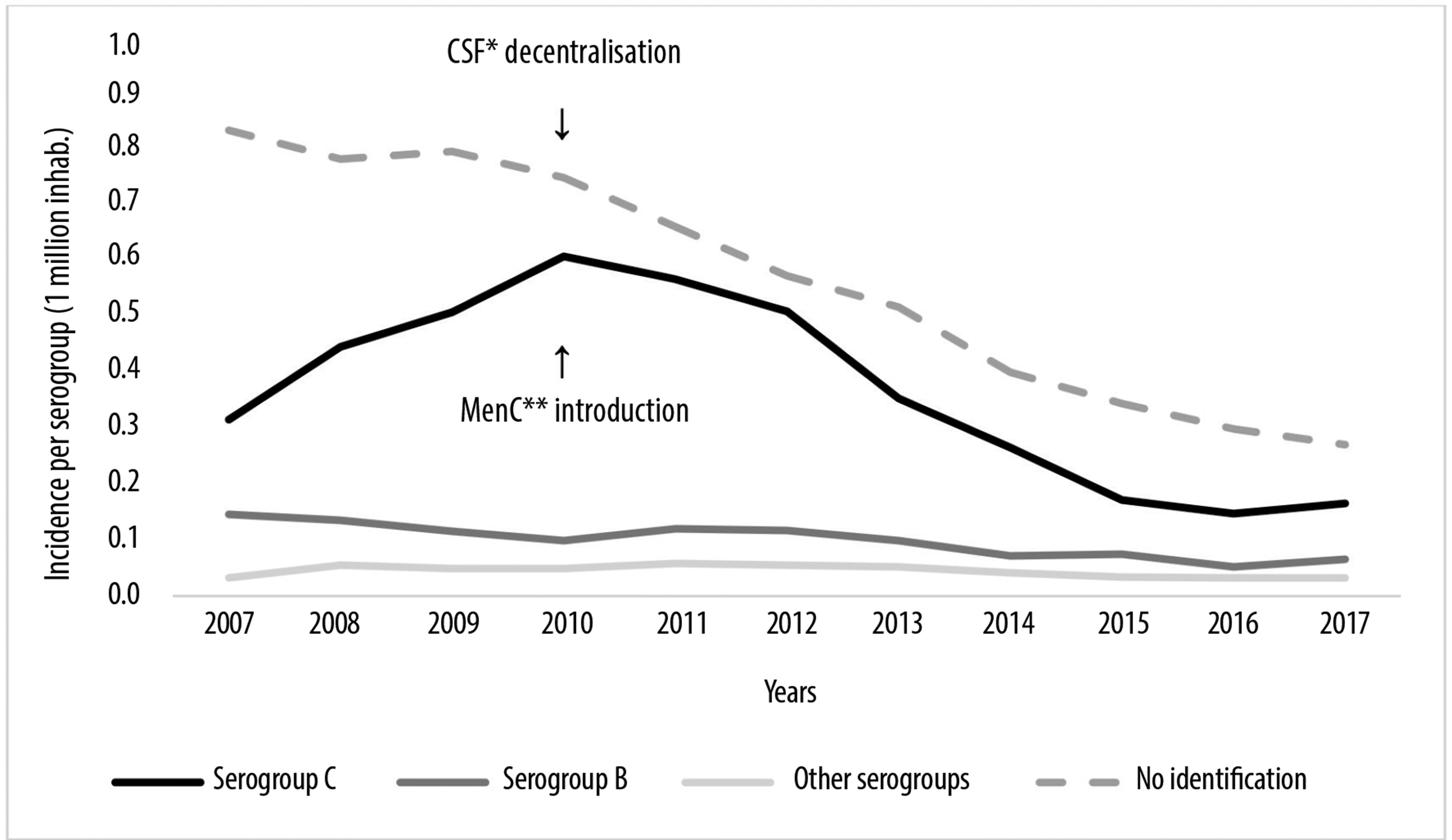
* Polymerase Chain Reaction (PCR) test.
** Meningococcal C conjugate vaccine.
Source: Notifiable Diseases Information System (SINAN); data collected in May 2018.
Figure 1 - Meningococcal disease incidence per serogroup, Brazil, 2007-2017
Table 1 shows that completeness of the “chemoprophylaxis use” field was good in all the period analyzed. Completeness of the “contact with suspected case” and “serogroup” fields was excellent, “immunization with MenC” was regular and “secondary case” was poor. As three out of five fields had completeness evaluated as good or excellent, data quality of the MD surveillance system was considered high.
Table 2 shows that we found an average percentage of 56.3% of records with timely chemoprophylaxis for contacts (up to 48 hours following suspected case symptoms); average percentage of records with timely chemoprophylaxis was not greater than 60.0% in any of the years evaluated. As chemoprophylaxis was timely in less than 70.0% of the records, system acceptability was considered to be low.
Table 1 - Completeness of compulsory and non-compulsory variables on the meningococcal disease epidemiological surveillance system, Brazil, 2007-2017
| Variables (fields) | Completeness (%) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | Average | Evolution | |
| Compulsory | |||||||||||||
| Chemoprophylaxis use | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | Good |
| Non-compulsory | |||||||||||||
| MenC immunization a | 75.6 | 74.8 | 74.9 | 76.9 | 76.2 | 77.5 | 76.0 | 79.1 | 80.9 | 81.1 | 80.7 | 77.6 | Regular |
| Contact with suspected case | 93.7 | 93.6 | 93.7 | 92.7 | 92.0 | 93.3 | 92.6 | 93.7 | 94.5 | 95.8 | 94.2 | 93.6 | Excellent |
| Secondary case | 3.2 | 2.2 | 2.6 | 2.9 | 2.5 | 3.1 | 1.8 | 2.4 | 3.0 | 2.3 | 2.1 | 2.6 | Poor |
| Serogroup | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | Excellent |
a) Meningococcal C conjugate vaccine.
Source: Notifiable Diseases Information System (SINAN); data collected in May 2018.
The average rates of investigation timeliness (96.6%) and closure timeliness (93.5%) were excellent, LCR collection timeliness (87.7%) was good, and notification (68.3%), service (58.1%) and chemoprophylaxis (56.3%) timeliness was regular. The MD epidemiological surveillance system was therefore considered to be timely (Table 2).
Table 2 - Evaluation of timeliness of meningococcal disease epidemiological surveillance, Brazil, 2007-2017
| Timeliness | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | Average 2007-2017 | Classification |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Notification ≤24h | 67.0 | 67.9 | 67.8 | 69.1 | 69.8 | 70.7 | 70.6 | 68.0 | 67.9 | 66.8 | 65.3 | 68.3 | Regular |
| Service ≤24h | 61.1 | 59.1 | 62.2 | 58.9 | 60.4 | 57.1 | 54.6 | 58.0 | 55.4 | 57.9 | 54.7 | 58.1 | Regular |
| Epidemiological investigation ≤24h | 95.8 | 96.4 | 96.0 | 96.2 | 97.0 | 97.3 | 97.1 | 97.0 | 95.9 | 97.7 | 96.6 | 96.6 | Excellent |
| CSF collection a ≤24h | 88.2 | 89.1 | 87.3 | 89.1 | 87.7 | 88.6 | 88.5 | 87.6 | 85.5 | 86.9 | 86.7 | 87.7 | Good |
| Chemoprophylaxis ≤48h | 53.9 | 54.9 | 56.5 | 59.8 | 57.8 | 58.5 | 59.8 | 54.2 | 55.1 | 55.9 | 52.7 | 56.3 | Regular |
| Closure up to 60 days | 86.5 | 87.9 | 93.5 | 95.0 | 94.3 | 94.8 | 96.1 | 95.6 | 95.2 | 93.9 | 95.5 | 93.5 | Excellent |
Figure 2 shows that in the period from 2007 to 2017 for Brazil as a whole, the highest incidence of MD cases occurred in the under 5 years old age groups, particularly among children under 1 year old. We observed progressive incidence reduction as of 2010, based on the hypothesis that there was case reduction due to MenC being introduced on the Brazilian immunization schedule.
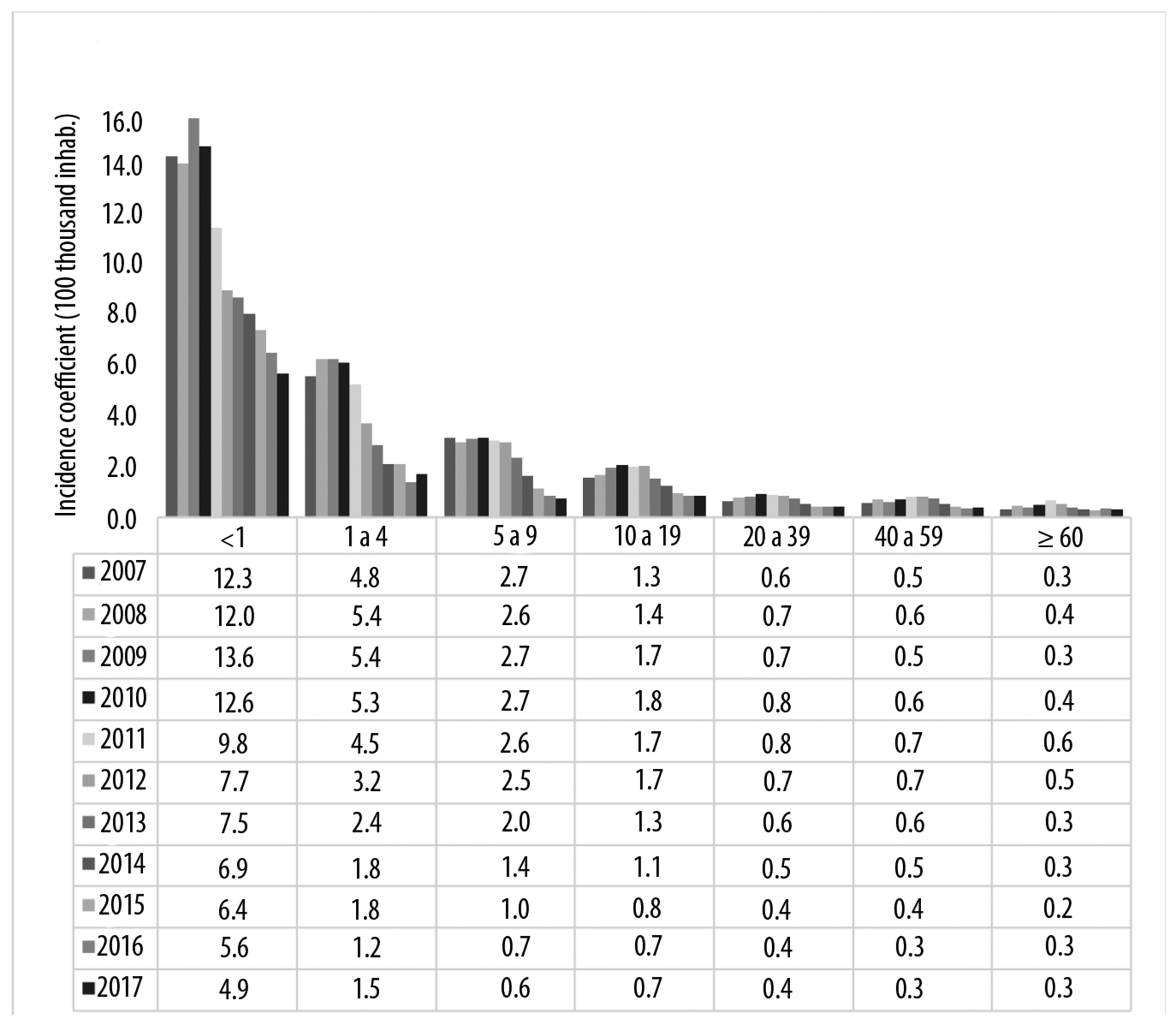
Source: Notifiable Diseases Information System (SINAN); data collected in May 2018.
Figure 2 - Meningococcal disease incidence per age group, Brazil, 2007-2017
Figure 3 demonstrates that between 2007 and 2010 for Brazil as a whole, average MD incidence rates for the period were near to or above 1 case/100,000 inhabitants, having homogeneous distribution all over the Brazilian territory and a tendency of greater concentration in the coastal region. Between 2011 and 2014, average incidence rates for Brazil as a whole fell below 1 case/100,000 inhabitants, although states like São Paulo, Rio de Janeiro and Pernambuco still had higher average incidence rates. In the period from 2015 to 2017, average incidence was lower than 0.5 case/100,000 inhabitants in Brazil, especially in the country’s North, Northeast and Midwest regions; in the same period, average incidence rates in the states of Rio de Janeiro and São Paulo were lower than 1 case/100,000 inhabitants.
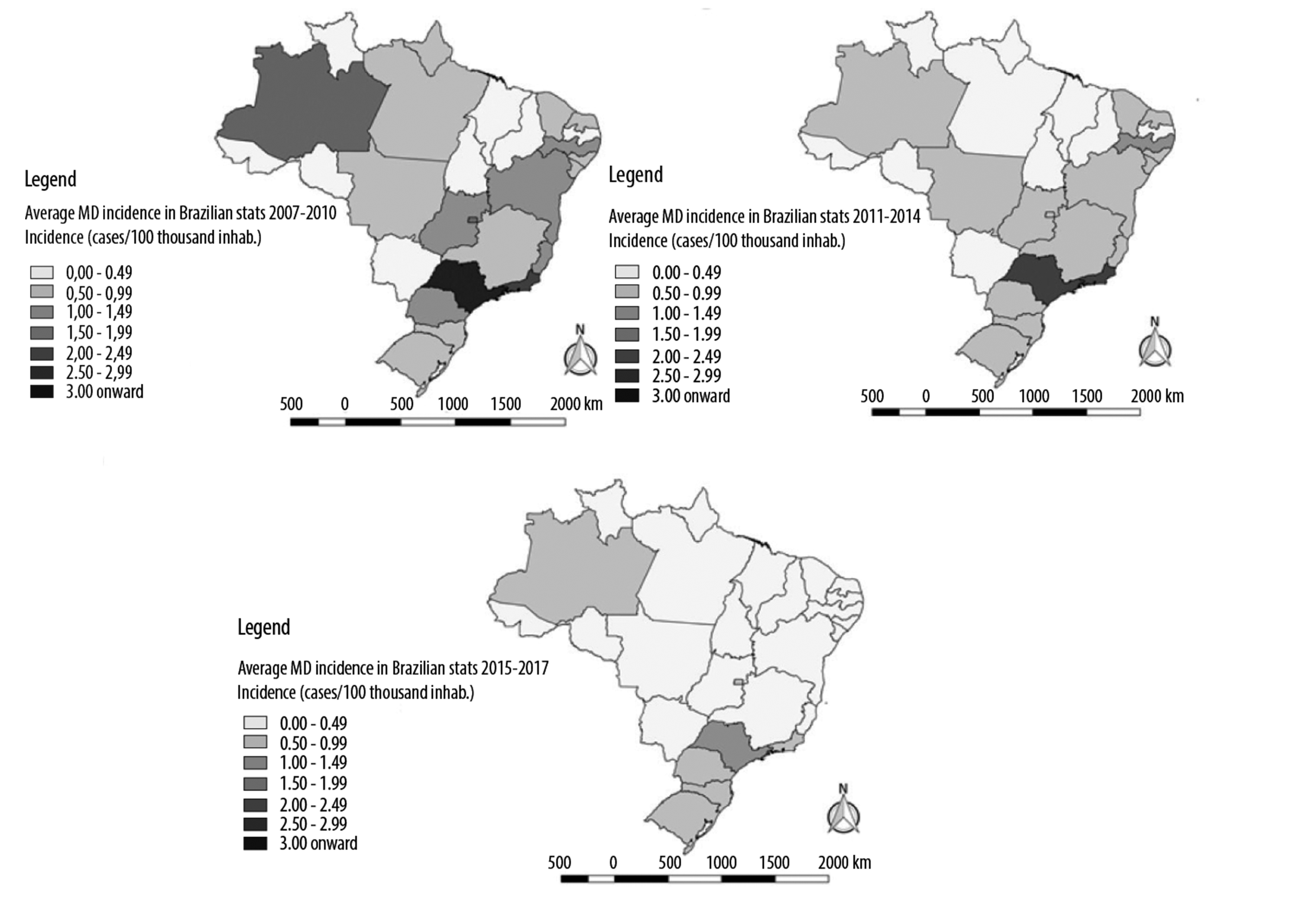
Source: Notifiable Diseases Information System (SINAN); data collected in May 2018.
Figure 3 - Meningococcal disease incidence per Federative Unit, Brazil, 2007-2017
The MD epidemiological surveillance system was considered to be representative because it allows the description of MD event time, place and persons in Brazil.
Finally, regarding the system “usefulness” attribute, we found that MD surveillance is able to provide important indicators in keeping with the objectives recommended, signaling epidemiological tendencies and their specificities, especially by affected serogroups and age groups.
The incidence coefficients in this study illustrate that the system is able to provide specific data per N. meningitidis serogroup, informing not only the disease magnitude trend according to serogroup, but also guiding adequate control measures - in this case, the priority of meningococcal C conjugate vaccine use in Brazil as the main immunization preventive action, since most cases are affected by MD serogroup C.4,8,10
The specific incidence coefficients per age group calculated in this study also showed that the system is able to inform meningococcal disease magnitude and trend per affected age groups; children under 5 years old were the most affected and, consequently, this was the age group defined for MD immunization as part of the PNI immunization schedule.10 As the meningococcal disease surveillance system is able to provide efficient epidemiological data and orientate effective actions for prevention and control, it was considered to be useful.
Discussion
The MD epidemiological surveillance system was considered to be useful, complex, flexible, with good data quality, low acceptability, representative, and with timeliness varying from excellent to regular, according to the type analyzed.
The complexity of the surveillance system in terms of confirming diagnosis of meningococcal disease cases is a direct result of the dynamics of the condition of “meningitis”. Because it is a clinical syndrome, it has diverse clinical presentations and possible etiologies, requiring health team accuracy in interventions to define causal etiology.1,3,8 High sensitivity of case definition in detecting meningitis cases is necessary because this neurological syndrome is rare and severe, requires urgent health care for its treatment, besides active tracing of possible secondary cases.1,3,8,17
However, apart from the system’s complexity imposing difficulties for its operationalization by health professionals, it also interferes with its acceptability as perceived by health professionals. Great professional effort is needed for case detection, investigation and chemoprophylaxis management. It is possible that these actions may not always be carried out completely and with the quality expected from health professionals working in services on the front line of the health system.1,3,8,17
The good flexibility of the MD surveillance system in identifying MD serogroup incidence effects and changes, with effect from MenC implementation in public immunization, allowed us to identify that this system is able to perceive the effect of new procedures or diagnosis methods when they are introduced, having the ability to reliably describe new trends in MD dynamics in the Brazilian population. Another important factor that corroborates the reliability of the analyses made using the MD database is the good data quality identified in this study.6,13,17
The good representativeness of the system is another positive factor with regard to monitoring and signaling relevant new trends in MD dynamics. The system is good at detecting changes in the epidemiological pattern of MD in Brazil.1,3,8 It is important to highlight that one of the main contributions of the system to the analysis of MD trends in Brazil was the monitoring of MD incidence with effect from the introduction of MenC on the Brazilian immunization schedule.4,10
Analysis of the timeliness of the meningococcal disease surveillance system demonstrated that surveillance actions should be evaluated with effect from case notification date and not from the date of onset of symptoms, since the whole process is set in motion with effect from notification of a suspected case admitted to a hospital or emergency center. Prior to this, local epidemiological surveillance did not have enough information to start control actions. Moreover, the timeliness or quickness with which treatment and control actions are carried out depends directly on the timeliness of suspected MD case notification, so that timeliness is the system’s most important attribute.3,5-7
In relation to the limitations of this study, it can be stated that the parameters used to measure and classify the attributes analyzed are subjective and, as such, researchers who analyze the data may underestimate or overestimate results.
The meningococcal disease surveillance system in Brazil is useful for analyzing trends and monitoring the profile of this disease in Brazil. Nevertheless, health professionals need to be constantly monitored and trained, especially those who provide clinical care for meningococcal disease, training them to overcome difficulties related to the natural complexity of a surveillance system based on a clinical syndrome, strongly dependent on the availability of professionals, medication and laboratory tests.
Referências
1. Rosenstein NE, Perkins BA, Stephens DS, Popovic T, Hughes JM. Meningococcal disease. N Engl J Med [Internet]. 2001 May [cited 2019 Jun 10];344(18):1378-88.Availablefrom: Availablefrom: https://www.nejm.org/doi/full/10.1056/NEJM200105033441807?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%3dpubmed .doi: 10.1056/NEJM200105033441807 [ Links ]
2. Branco RG, Amoretti CF, Tasker RC. Meningococcal disease and meningitis. J Pediatr (Rio J) [Internet]. 2007 May [cited 2019 Jun 10];83(2 Suppl):S46-53. Available from: Available from: http://www.scielo.br/pdf/jped/v83n2s0/en_a06v83n2s0.pdf . doi: 10.1590/S0021-75572007000300006 [ Links ]
3. Nacional Institute for Health and Care Excellence (NICE). Meningitis (bacterial) and meningococcal septicaemia in under 16s: recognition, diagnosis and management [Internet]. Londres: NICE; 2015 [cited 2019 Jun 10]. Available from: Available from: https://www.nice.org.uk/guidance/cg102 [ Links ]
4. Sáfadi MP, González-Ayala S, Jäkel A, Wieffer H, Moreno C, Vyse A. The epidemiology of meningococcal disease in Latin America 1945-2010: an unpredictable and changing landscape. Epidemiol Infect [Internet]. 2012 Mar [cited 2019 Jun 10];141(3):4 Available from: Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3566594/ . doi: 10.1017/S0950268812001689 [ Links ]
5. Santos ML, Ruffino-Netto A. Doença meningocócica: situação epidemiológica no Município de Manaus, Amazonas, Brasil, 1998/2002. Cad Saúde Pública [Internet]. 2005 maio-jun [citado 2019 jun 10];21(3):823-9. Disponível em: Disponível em: http://www.scielo.br/pdf/csp/v21n3/16.pdf . doi: 10.1590/S0102-311X2005000300016 [ Links ]
6. Moraes C. Avaliação do sistema de vigilância epidemiológica da meningite, com ênfase em meningites bacterianas não-especificadas, Brasil, 2001 a 2006. [trabalho de conclusão de curso]. Brasília (DF): Episus; 2007. [ Links ]
7. Moraes JC, Barata RB. A doença meningocócica em São Paulo, Brasil, no século XX: características epidemiológicas. Cad Saúde Pública [Internet]. 2005 set-out [citado 2019 jun 10];21(5):1458-71. Disponível em: Disponível em: http://www.scielo.br/pdf/csp/v21n5/19.pdf . doi: 10.1590/S0102-311X2005000500019 [ Links ]
8. Barroso DE, Carvalho DM, Nogueira SA, Solari CA. Doença meningocócica: epidemiologia e controle dos casos secundários. Rev Saúde Pública [Internet]. 1998 fev [citado 2019 jun 10];32(1):89-97. Disponível em: Disponível em: http://www.scielo.br/pdf/rsp/v32n1/2381.pdf . doi: 10.1590/S0034-89101998000100014 [ Links ]
9. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Guia de Vigilância em Saúde [Internet]. 8. ed. Brasília: Ministério da Saúde; 2017 [citado 2019 jun 10]. v. 1. Disponível em: Disponível em: http://bvsms.saude.gov.br/bvs/publicacoes/guia_vigilancia_saude_volume_1.pdf [ Links ]
10. Laval CAB, Pimenta FC, Andrade JG, Andrade SS, Andrade ALSS. Progress towards meningitis prevention in the conjugate vaccines era. Braz J Infect Dis [Internet]. 2003 Oct [cited 2019 Jun 10];7(5):315-24. Available from: Available from: http://www.scielo.br/pdf/bjid/v7n5/18581.pdf . doi: 10.1590/S1413-86702003000500006 [ Links ]
11. Cardoso CW, Pinto LLS, Reis MG, Flannery B, Reis JN. Impact of vaccination during an epidemic of serogroup C meningococcal disease in Salvador, Brazil. Vaccine [Internet]. 2012 Aug [cited 2019 Jun 10];30(37):5541-6. Available from: Available from: https://www.sciencedirect.com/science/article/pii/S0264410X12009103?via%3Dihub . doi: 10.1016/j.vaccine.2012.06.044 [ Links ]
12. Cassia NG, Carvalhanas T. Meningites no município de São Paulo, com ênfase para doença meningocócica. CVE SP. 2012;9(97):5-25. [ Links ]
13. Fundação Instituto Oswaldo Cruz. Diretrizes para avaliação de sistemas de vigilância epidemiológica. Rio de Janeiro: Fundação Instituto Oswaldo Cruz; 2001. [ Links ]
14. Armstrong G, Birkhead GS, Horan JM, Herrera G, Lee LM, Milstein RL, Centers for Disease Control and Prevention, et al. Updated guidelines for evaluating public health surveillance systems: recommendations from the guidelines working group. MMWR. 2001 Jul [cited 2019 Jun 10];50(RR-13):1-35. Available from: Available from: https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5013a1.htm [ Links ]
15. Organização Pan-Americana da Saúde. Organização Mundial da Saúde. Informe regional de SIREVA II 2000 - 2012 [Internet]. Washington (D.C.): Organização Pan-Americana da Saúde; 2011[citado 2019 jun 10]. Disponível em: Disponível em: http://www.paho.org/hq/index.php?option=com_content&view=category&layout=blog&id=3609&Itemid=3953 [ Links ]
16. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Roteiro para uso do Sinan-NET, análise da qualidade da base de dados e cálculo de indicadores epidemiológicos e operacionais: meningite [Internet]. Brasília: Ministério da Saúde ; 2008 [citado 2019 jun 10]. Disponível em: Disponível em: http://www.saude.ba.gov.br/wp-content/uploads/2017/12/Caderno-de-analise-EXANT.pdf [ Links ]
17. Moraes GH, Carvalho ML, Monteiro L, Brondi LM . Vigilância Epidemiológica das Meningites no Brasil. 2001. Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2016/outubro/18/Revista-Memorias-EPISUS-10-anos.pdf [ Links ]
Received: October 03, 2018; Accepted: May 10, 2019










 texto en
texto en