Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.29 no.1 Brasília 2020 Epub 19-Fev-2020
http://dx.doi.org/10.5123/s1679-497420120000100010
ORIGINAL ARTICLE
Evaluation of the Drug-Resistant Tuberculosis Surveillance System, Brazil, 2013-2017*
1Ministério da Saúde, Secretaria de Vigilância em Saúde, Programa de Treinamento em Epidemiologia Aplicada aos Serviços do Sistema Único de Saúde, Brasília, DF, Brazil
2Ministério da Saúde, Secretaria de Vigilância em Saúde, Coordenação Geral de Vigilância das Doenças de Transmissão Respiratória de Condições Crônicas, Brasília, DF, Brazil
Methods:
this was an evaluative study, following Centers for Disease Control and Prevention guidelines, using national data from the Special Tuberculosis Treatment Information System (SITETB), and the Notifiable Diseases Information System (SINAN), from 2013 to 2017.
Results:
average data completeness was 95% (schooling [89.1%; 5,417/6,078], nationality [94.7%; 5,754/6,078], race/skin color [99.1%; 6,023/6,078], type of resistance [98.6%; 5,995/6,078], clinical form [100%; 6,078/6,078], and HIV test [87%; 5,289/6,078]); average proportion of cases with sputum cultures performed was 65.7% (culture 1 [94.8%; 5,764/6,078], culture 2 [69.8%; 4,241/6,078], culture 3 [54.7%, 3,324/6,078], and culture 4 [43.6%; 2,652/6,078]); DRTB-SS reported 52% (1,197/2,300) of multi-resistant cases estimated by the World Health Organization in 2015, 41.3% (990/2,400) in 2016, and 45.8% (1,100/2,400) in 2017.
Keywords: Tuberculosis, Multidrug-Resistant; Information Systems; Epidemiological Monitoring; Public Health Surveillance
Introduction
Among the leading causes of death worldwide, tuberculosis is considered to be a priority problem on the global agenda. Ten million cases were estimated to have been registered in 2017 and 1.6 million deaths were estimated to have been caused by tuberculosis in the same period.1
Tuberculosis resistant to one or more drugs, referred to as drug-resistant tuberculosis (DRTB), represents a Public Health crisis and a health security and tuberculosis prevention threat.1,2 Moreover, resistance to rifampicin and isoniazid, which are the strongest first-line drugs, is classified as multi-drug-resistant tuberculosis (MDR-TB).2
It is estimated that 558,000 new rifampicin-resistant tuberculosis cases were laboratory-diagnosed worldwide in 2017, 82% of which were estimated to be MDR-TB cases.1 In the same year, Brazil was estimated to have 2,000 MDR-TB rifampicin-resistant cases, 1,110 of which were laboratory-confirmed.1
The Drug-Resistant Tuberculosis Surveillance System (DRTB-SS) is universal and passive, comprised of cases that should be notified on the Notifiable Diseases Information System (SINAN) and also on the Special Tuberculosis Treatment Information System (SITETB). The Multi-Resistant Tuberculosis (MRTB) Epidemiological Surveillance Program came into being in Brazil in the year 2000.3-5 The MRTB Information System was implemented in 2004, having as its objectives (I) to enhance MRTB case notification and follow-up, (II) to strengthen and decentralize the MRTB Epidemiological Surveillance Program, (III) to enable evaluation and research by keeping the information system up to date and (IV) to control shipment of stocks of specific drugs at central and peripheral level.4 The SITETB system was implemented in 2012/2013 and replaced the MRTB Information System. It is currently the online information system used in Brazil to achieve the objectives mentioned above:5 initially the patient is notified on the SINAN system; when DRTB is diagnosed, the case is closed as a DRTB case on the SINAN system and is notified on the SITETB system.2
The purpose of evaluating surveillance systems is (i) to verify performance and whether surveillance needs are being met, (ii) to verify whether important Public Health problems are being monitored efficiently and effectively, and (iii) to propose recommendations for system improvement.6 The Centers of Disease Control and Prevention (CDC) Guidelines propose surveillance system evaluation based on qualitative and quantitative attributes.6
Diverse studies have been conducted with the aim of evaluating the tuberculosis surveillance system, principally with regard to the quality of data available on the SINAN system.7-9 However, few studies relating to DRTB-SS evaluation have been conducted in Brazil and they have always been related to system decentralization, its history and control measures.3,5,10 Considering the epidemiological relevance of DRTB within Public Health and the fact that the DRTB-SS has not been widely evaluated from a national perspective and on a national scale, the objective of this study was to evaluate the system, its qualitative and quantitative attributes and its usefulness. This evaluation will enable greater knowledge of DRTB-SS, recommendations for improvements to its performance and fulfilling the needs it is intended to address, bringing benefits for system users and drug-resistant tuberculosis patients.
Methods
This was an evaluative study of the DRTB-SS based on descriptive analysis of DRTB cases diagnosed between 2013 and 2017 in Brazil, using the evaluation guidelines proposed by CDC.6 The qualitative attributes evaluated were simplicity, data quality, flexibility and acceptability, while the quantitative attributes evaluated were sensitivity, representativeness, timeliness and stability. DRTB-SS usefulness was also evaluated.
Brazil is the largest South American country in terms of territorial size and has 5,570 municipalities spread over 27 Federative Units and five geographical regions. In 2019 it had an estimated population of 210,659,013 inhabitants.11
According to the surveillance action routine, DRTB cases are initially notified and then closed on the SINAN system and subsequently notified on the SITETB system. This latter system requires at least 110 variables to be filled in. Based on clinical and laboratory information and information on treatment proposed, cases are assessed by a team of specialist physicians, responsible for validating treatment indicated. Case follow-up is done by means of periodic medical consultations and laboratory tests until treatment is finalized.
Data and technical information were retrieved from the SITETB and SINAN systems and tuberculosis surveillance manuals.2,4 The scientific literature,12,13 and National Tuberculosis Control Program technical staff were also consulted. Data was also taken from a satisfaction survey of SITETB system users by means of a semi-structured questionnaire available on the FormSUS® platform. Data analysis was based on simple and relative frequency measurements, central and scatter trends, using a 95% confidence interval (95%CI). The following applications were used: Microsoft Excel, Epi Info 7, ReclinK III 3.1.6.3160, Stata 12.0.
Qualitative attributes
With regard to the SITETB system, DRTB-SS simplicity was evaluated based on the following items:
DRTB case notification/validation/follow-up and closure flowchart.
Number of case notification variables (obligatory and key variables).
Number of health professionals and health facilities registered on SITETB.
Data quality was evaluated using the following criteria:
a) Duplication - identified using the RecLink III duplication routine, for new cases diagnosed in the study period.
b) Completeness - calculated for sociodemographic variables (schooling; nationality; race/skin color) and DRTB case clinical variables (type of resistance; clinical form; HIV test date);8 completeness was assessed taking the average proportion (%) of complete fields in relation to total cases, while incomplete fields were taken to be fields left blank or for which information was missing.7-9,14
c) Inconsistency - assessed taking average invalid data recorded for DRTB cases, based on disagreement between categories of the following variables,
- human immunodeficiency virus (HIV) test with negative result but presence of AIDS-related comorbidity;8
- diagnosis date - treatment start date with result in <0 days -;
- X-ray with presentation (bilateral, or unilateral cavitation or non-cavitation) and clinical form (extrapulmonary).
Further information about the formula for calculating the indicators mentioned above can be found in Figure 2, shown in the chapter describing the Results of this study.
Flexibility was calculated based on the following items:
Period (in days) between rapid molecular tuberculosis test (RMT) rollout in the laboratory network and inclusion of the test result collection variable on the SITETB system.
Health technology post-rollout evaluation - proportion (%) of DRTB cases having RMT in the first year following rollout (rollout occurred in September 2014).
Number of variables relating to treatment options included (per annum) on the SITETB system and existence of variables to be included.
Difference between variables on the paper form and the online form for notification on the SITETB system.
Satisfaction survey with SITETB system users, assessed by the proportion (%) of alterations suggested by system users that had been incorporated.
Acceptability was evaluated by calculating the following items:
a) Sputum culture tests performed to monitor DRTB cases with the purpose of estimating the proportion (%) of cases with sputum culture collection and follow-up data filled in, out of the total number of cases diagnosed in the period. Sputum culture monitoring tests are recommended every three months, up until the 12th month after treatment has ended.2 We assessed the data for the first four quarterly DRTB case follow-up culture tests.
b) Proportion (%) of new cases testing for HIV (positive or negative), out of total new cases diagnosed in the period.9 HIV testing is recommended for all individuals with tuberculosis.2
c) Probabilistic linkage between all cases classified as DRTB cases notified on the SITETB system and cases notified on the SINAN system and closed as DRTB cases on SINAN, diagnosed between 2013 and 2017. The objective of this procedure was to evaluate agreement between the two information systems.15-16 Based on the Venn diagram, we identified the number of DRTB cases on the SITETB system (S1), the number of cases on the SINAN system closed as DRTB cases (S2) and the number of cases on both systems (A).17 The estimated total number of DRTB cases (Ne) was calculated using the Chapman formula,18 where
Taking the estimated number of cases, the proportion of cases notified by the SITETB system was then calculated.

a) RMT: rapid molecular test.
b) SITETB: Special Tuberculosis Treatment Information System.
c) HIV: human immunodeficiency virus.
Figure 1 - Description of the items for evaluation, classification and overall evaluation of attributes of the Drug-Resistant Tuberculosis Surveillance System, Brazil, 2013-2017
d) Average proportion (%) of ‘I agree totally’ and ‘I agree partially’ answers to the following items of the satisfaction survey:
Quantitative attributes
Sensitivity was evaluated according to capacity to notify cases on the DRTB-SS, i.e., cases notified on the SITETB and the SINAN systems, out of total cases estimated by the World Health Organization (WHO) based on prevalence surveys conducted in Brazil.1,6 This indicator was calculated as the average ratio between MDR-TB cases (n) detected by the DRTB-SS system in a year and total MDR-TB cases (n) estimated by WHO, multiplied by 100, in 2015, 2016 and 2017.1
Representativeness was evaluated according to case characterization, as follows.
a) Person - distribution (%) of new MDR-TB cases by sex and age group; and distribution (%) of rifampicin-resistant cases diagnosed by RMT by sex, age group and TB-HIV coinfection in Brazil versus international literature.12,13
b) Time - new DRTB cases (n) according to month and year of diagnosis.
Timeliness was evaluated based on the following indicators created for this study:
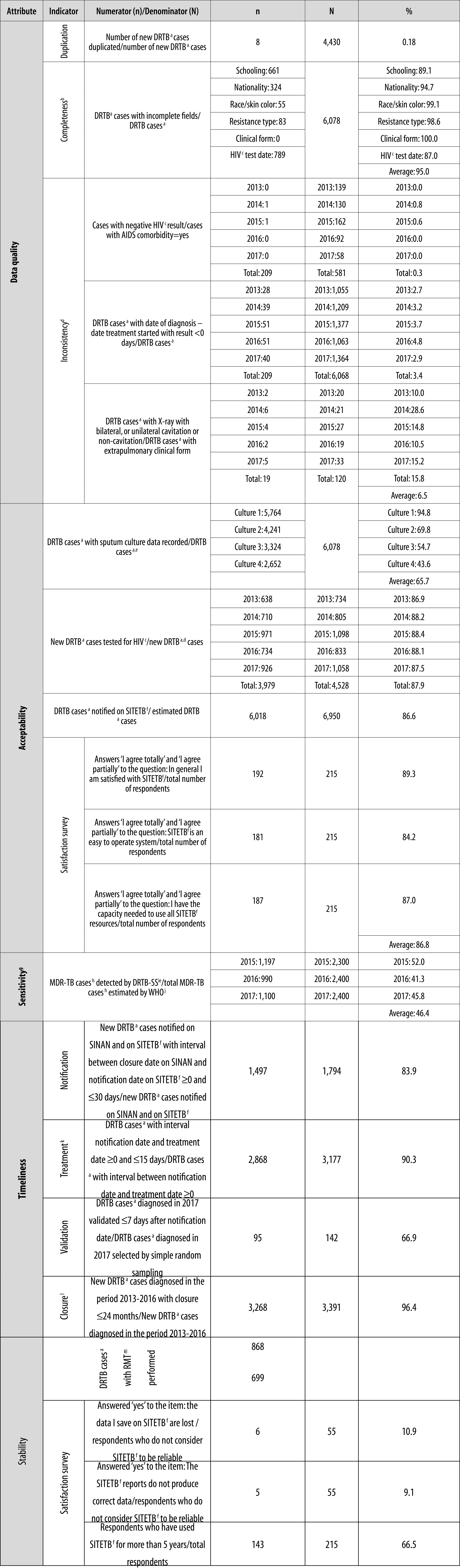
a) DRTB: drug-resistant tuberculosis.
b) Indicators calculated for each key variable (schooling, nationality, race/skin color, resistance type, clinical form, HIV test date).
c) HIV: human immunodeficiency virus. d) Indicators calculated per year of diagnosis (2013-2017).
e) Indicators calculated per sputum culture performed (cultures 1 to 4).
f) SITETB: Special Tuberculosis Treatment Information System.
g) Indicators calculated per year of diagnosis (2015-2017).
h) MDR-TB: multi-drug-resistant tuberculosis.
i) DRTB-SS: Drug-Resistant Tuberculosis Surveillance System.
j) WHO: World Health Organization.
k) Cases that had no treatment start date were removed.
l) Extensively resistant cases were removed.
m) RMT: rapid molecular test.
Figure 2 - Evaluation of Drug-Resistant Tuberculosis Surveillance System data quality, acceptability, sensitivity, timeliness and stability, Brazil, 2013-2017
a) Notification - proportion (%) of new DRTB cases with an interval between closure date on the SINAN system and date of notification on the SITETB system ≥0 and ≤30 days.
b) Treatment - proportion (%) of DRTB cases with an interval between notification date and treatment start date ≥0 and ≤30 days.
c) Validation - period between case notification and treatment validation by specialist physicians. A simple random sample of new DRTB cases notified in 2017 was calculated (expected frequency = 50%; margin of error = 5%). For the selected cases we calculated the proportion of cases with an interval between notification date and validation date ≤7 days; for this item we used an 80% confidence interval (8%CI) as it was an estimate based on the use of probabilistic sampling.
d) Closure - proportion (%) of new DRTB cases diagnosed in the period 2013-2016, with an interval between treatment start date and closure date ≤24 months; we excluded extensively drug resistant cases, for which treatment periods may be greater than 24 months.
Stability was evaluated by means of the following items:
a) DRTB cases (n) with RMT performed (taking into consideration RMT stockout in 2015).
b) Average proportion (%) of “Yes” answers to the satisfaction survey questions, calculated in relation to participants who considered that the SITETB was not reliable, namely:
c) Proportion (%) of survey respondents who had used SITETB for more than five years.
Classification and overall evaluation of the qualitative and quantitative attributes and their respective items are shown in Figure 1. DRTB-SS usefulness was evaluated considering the objectives of the MRTB Information System.4
The study project was approved by the National Research Ethics Committee (CONEP) - Consolidated Report No. 2.756.559, dated July 6th 2018 -, in accordance with National Health Council Resolution No. 466, dated December 12th 2012.
Results
A total of 6,078 DRTB cases were notified on the SITETB system in the period assessed. Of these, 4,528 were new cases, distributed mainly in the Southeast region (n=1,783; 39.4%). The most frequent closure types for new cases diagnosed between 2013 and 2016 were favorable outcome (cure/treatment completed) (n=2,196; 63.7%) and treatment abandonment (n=675; 19.6%). Closure due to death was recorded in 7.3% (n=252) of cases (Table 1).
Simplicity
In January 2019, 895 users and 889 health facilities were registered on SITETB and DRTB-SS was considered to be complex.
Data quality
Probabilistic linkage identified eight (0.2%) duplications among new DRTB cases. Average completeness of the ‘schooling’ (89.1%), ‘nationality’ (94.7%), ‘race/skin color’ (99.1%), ‘resistance type’ (98.6%), ‘clinical form’ (100%) and ‘HIV test date’ (87.0%) variables was 95.0% for DRTB cases. Only 0.3% of cases had inconsistent data on existence of AIDS and 3.4% had inconsistent data on diagnosis date and tuberculosis treatment start date. However, data on clinical form was inconsistent for 15.8% of people with DRTB (Figure 2), resulting in excellent data quality.
Flexibility
The RMT variable was included on the SITETB system approximately 488 days after RMT rollout in the laboratory network. In 2015, one year after RMT rollout, 62.8% of cases were diagnosed using this test. All variables related to new treatment options (n=23) had been included on SITETB when this evaluation was conducted. Comparison between printed paper and online notification instruments identified differences in relation to 12 variables. Out of the 215 satisfaction survey respondents, 37 (17.2%) suggested alterations for the SITETB system and 18 (48.6%) of these respondents felt that they had been taken into consideration regarding their satisfaction
Table 1 - New drug-resistant tuberculosis cases by region of residence and drug-resistant tuberculosis case closure status, Brazil, 2013-2017
| Variables | Year of diagnosis | Total | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2013 | 2014 | 2015 | 2016 | 2017 | ||||||||
| N | % | N | % | N | % | N | % | N | % | N | % | |
| Region of residence | ||||||||||||
| North | 71 | 9.7 | 89 | 11.1 | 150 | 13.7 | 100 | 12.0 | 176 | 16.6 | 586 | 12.9 |
| Northeast | 179 | 24.4 | 224 | 27.8 | 273 | 24.9 | 211 | 25.3 | 230 | 21.7 | 1,117 | 24.7 |
| Southeast | 320 | 43.6 | 302 | 37.5 | 468 | 42.6 | 300 | 36.0 | 393 | 37.2 | 1,783 | 39.4 |
| South | 149 | 20.3 | 162 | 20.1 | 183 | 16.7 | 197 | 23.6 | 235 | 22.2 | 926 | 20.5 |
| Midwest | 15 | 2.0 | 28 | 3.5 | 24 | 2.2 | 25 | 3.0 | 24 | 2.3 | 116 | 2.6 |
| Total | 734 | 16.2 | 805 | 17.8 | 1,098 | 24.2 | 833 | 18.4 | 1,058 | 23.4 | 4,528 | 100.0 |
| Closure status a | ||||||||||||
| Cure/treatment completed | 469 | 64.0 | 511 | 63.9 | 704 | 64.4 | 512 | 62.4 | - | - | 2,196 | 63.7 |
| Treatment abandonment | 140 | 19.1 | 153 | 19.1 | 221 | 20.2 | 161 | 19.6 | - | - | 675 | 19.6 |
| Death/death from other cause | 52 | 7.1 | 60 | 7.5 | 74 | 6.8 | 66 | 8.0 | - | - | 252 | 7.3 |
| Failure | 55 | 7.5 | 49 | 6.1 | 55 | 5.0 | 31 | 3.8 | - | - | 190 | 5.5 |
| Regimen change | 9 | 1.2 | 13 | 1.6 | 24 | 2.2 | 17 | 2.1 | - | - | 63 | 1.8 |
| In treatment | 1 | 0.1 | 5 | 0.6 | 11 | 1.0 | 29 | 3.5 | - | - | 46 | 1.3 |
| DRTB b | 6 | 0.8 | 9 | 1.1 | 3 | 0.3 | 2 | 0.2 | - | - | 20 | 0.6 |
| Transfers | 1 | 0.1 | - | - | 2 | 0.2 | 3 | 0.4 | - | - | 6 | 0.2 |
| Total | 733 | 21.3 | 800 | 23.2 | 1,094 | 31.7 | 821 | 23.8 | - | - | 3,448 | 100.0 |
a) Closure of cases diagnosed in 2017 was not evaluated (treatment in progress).
b) DRTB: drug-resistant tuberculosis with the system. When combining the items evaluated, system flexibility was found to be moderate.
Acceptability
Of the 6,078 notified DRTB cases, 94.8% carried out sputum culture 1, 69.8% carried out sputum culture 2, 54.7% carried out sputum culture 3 and 43.6% carried out sputum culture 4. The average proportion of DRTB cases having data on sputum culture collection in the period was 65.7%. Among new DRTB cases, 87.9% (n=3,979) tested for HIV (Figure 2). Based on 6,018 DRTB cases notified on SITETB and 4,035 cases notified on SINAN, probabilistic linkage identified 3,494 pairs (cases on both systems). Using the Chapman formula, 6,950 DRTB cases were estimated for the period, 86.6% (n=6,018) of which were notified on SITETB. Regarding those who answered the satisfaction survey (n=215), 89.3% (n=192) were satisfied with the system, 84.2% (n=181) considered SITETB easy to operate and 87.0% (n=187) stated that they were capable of using all its resources. Overall acceptability was moderate.
Sensitivity
In 2015, 1,197 rifampicin-resistant MDR-TB cases were notified on DRTB-SS, i.e. 52.0% of the 2,300 cases estimated by WHO for the same period. In 2016, 41.3% (990/2,400) of estimated cases were notified, while in 2017, 45.8% (1,100/2,400) of estimated cases were notified (Figure 2). The overall average calculated for the three-year period was 46.4%, resulting in low sensitivity.
Representativeness
MDR-TB cases notified on SITETB occurred most frequently among young adults aged 20-39 (44.4%), the majority of whom were male (67.4%), thus corroborating results available in the international literature. A similar pattern was found in rifampicin-resistant cases notified on SITETB and also in the literature consulted, as well as the same proportion of HIV coinfection (14.1%).
DRTB case distribution per month of diagnosis was constant throughout the period and there was no seasonal pattern (Figure 3). Representativeness was considered to be high.
Timeliness
Notification on SITETB was considered to be timely for 83.9% of new DRTB cases. Treatment was started on a timely basis for 90.3% of DRTB cases notified in the period. Validation was timely for 66.9% (80%CI 61.9;71.9) of new cases notified in 2017, selected using random sampling. 96.4% of new cases notified between 2013 and 2016 were closed on a timely basis (Figure 2). This attribute was considered to be moderate.
Stability
In 2015, the year in which RMT stockout occurred, there was a reduction in the number of tests performed (n=868 in 2014; n=699 in 2015). In the satisfaction survey, among those who did not consider the SITETB system to be totally reliable (n=55), 6 (10.9%) stated
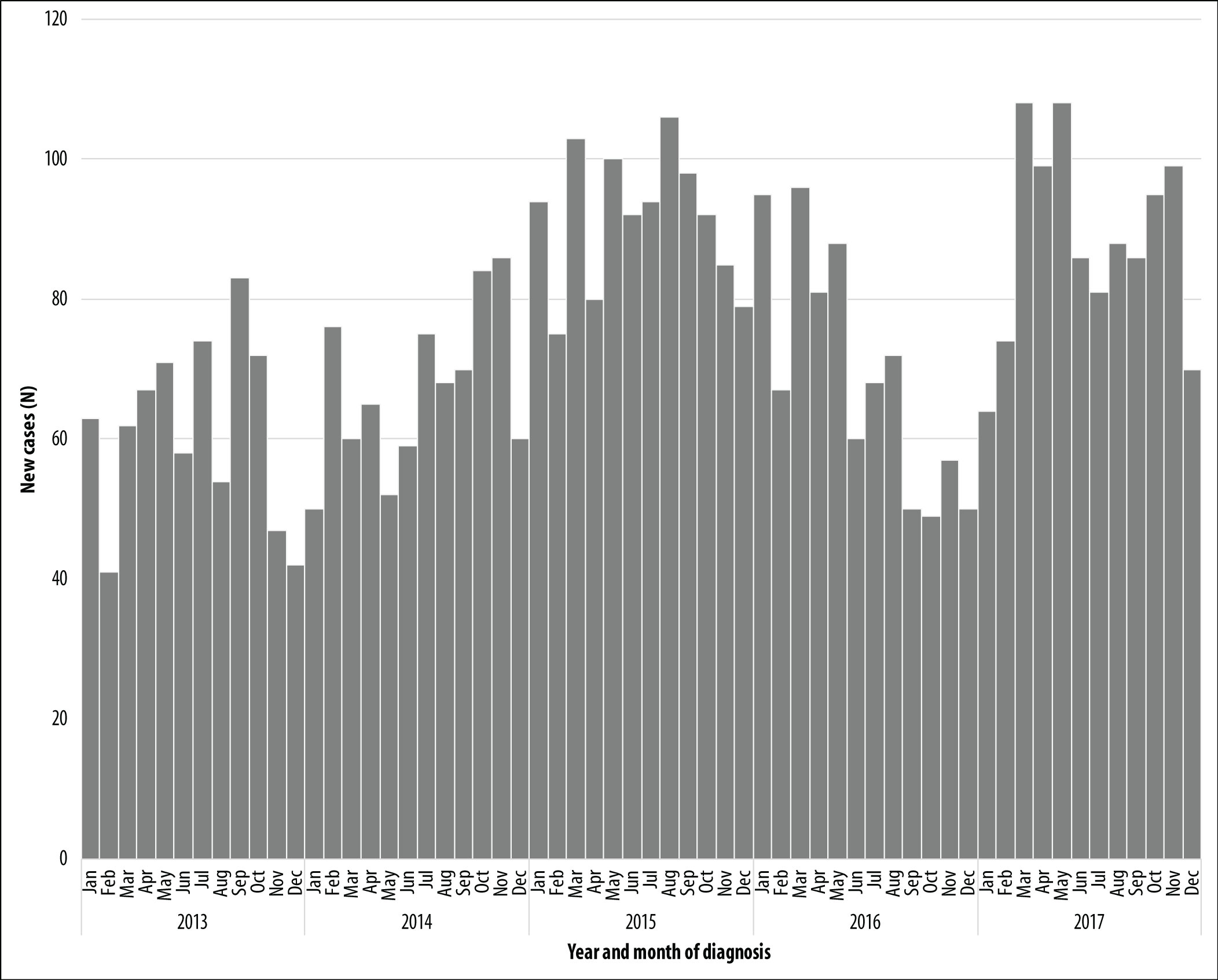
Figure 3 - Number of new drug-resistant tuberculosis cases (N=4,528) by year of diagnosis, Brazil, 2013-2017
that the data they saved on SITETB were lost, and 5 (9.1%) stated that SITETB reports did not produce correct data. Taking the survey participants as a whole (n=215), 143 (66.5%) had used SITETB for more than five years (Figure 2). Overall stability was considered to be moderate.
Discussion
DRTB-SS was considered to be a complex, high quality and representative system. Its flexibility, acceptability, timeliness and stability were considered to be moderate, while its sensitivity was considered to be low. It was found to be useful, based on its ability to meet the objectives of the MRTB Information System.4
This study has some limitations. Medication management - a process which forms part of DRTB-SS - was not evaluated and cases which had ‘death’ as their outcome were not checked on the official Mortality Information System (SIM). Failure to identify, confirm and/or evaluate this data and information can compromise the results of a study. Moreover, data incompleteness may have been overestimated owing to fields with missing information, which may possibly represent unknown information rather than failure to fill in the field. Case closure classification errors on SINAN may have influenced system acceptability, as only cases closed as DRTB cases were used for probabilistic linkage with SITETB data. This analysis did not use Laboratory Environment Manager data, which would have enabled unnotified DRTB cases to be identified and this would have increased DRTB-SS acceptability.
Various results regarding the simplicity of systems similar to DRTB-SS in other countries can be found in the literature. In the Yemen, a study conducted in sentinel units evaluated DRTB-SS as having com moderate simplicity.18 In the United States, a pilot instrument prepared to obtain basic information on DRTB cases was considered to be simple.19 In comparison with the results of these two studies, DRTB-SS can be considered to have been implemented throughout Brazil, having consolidated workflows and activities which, to a certain extent, justify its complexity.
The high data quality found reflects technical staff knowledge about activities recommended for DRTB control. This is fundamental not only for data quality, but also for achieving favorable outcomes for DRTB cases, as per the conclusion reached by a study conducted in Niterói, RJ.20 HIV testing percentages were higher than those found on the treatment-sensitive tuberculosis surveillance system.21 This reiterates the existence of technical staff surveillance knowledge and actions and can collaborate with timely starting of antiretroviral therapy and reducing mortality caused by TB-HIV coinfection.2
Incorporation of new variables and new technologies on SITETB demonstrates the flexibility of this information system. Different results were reported by a DRTB-SS evaluation carried out in the United States between 2012 and 2015, when concern was expressed regarding incorporation of technology and DRTB case surveillance into local electronic systems.19 However, the low percentage of incorporation of alterations suggested by SITETB users found via the satisfaction survey demonstrates the importance of interlocution between administrators and users with the aim of improving system functioning.
Among the items evaluated for the ‘acceptability’ attribute, low percentages were found for follow-up sputum culture. Similar results have been documented for sensitive tuberculosis follow-up examinations and surveillance system evaluations.7-9 As such, we propose evaluation of work processes, laboratory services and case follow-up, with the aim of identifying difficulties and proposing strategies to overcome them. SITETB case detection capacity (calculated based on cases notified on SINAN, cases notified on SITETB and real case estimation) was assessed as being moderate. Case detection improvement is possible, given that, according to the Ministry of Health recommendation, DRTB cases notified on SITETB should also be notified on SINAN and closed on SINAN as DRTB cases.2 Acceptability as assessed by system users, when compared to the 2016 satisfaction survey, showed a larger number of individuals who considered themselves to be capable of using SITETB resources and who stated that they were satisfied with the system.22
Low DRTB-SS sensitivity, when compared to WHO estimates, suggests the existence of gaps in case detection. As such, strategies aimed at identifying areas for structuring the laboratory network and expanding DRTB diagnosis can help to achieve greater knowledge about resistance to tuberculosis drugs in Brazil. Low DRTB-SS sensitivity has already been documented in other studies, highlighting the need to improve access to diagnosis and treatment.18
Moderate notification timeliness and low validation timeliness indicate that these processes can be carried out in a shorter space of time, although there is no standard timeliness for these items. Untimely validation, together with other factors, such as the time taken to make laboratory test results available,23 can delay the start of treatment, contributing to dissemination of resistant bacilli and, consequently, worsening of the patient’s condition.
DRTB-SS was considered to be useful because it offers conditions for notifying and monitoring cases on SINAN and, above all, on SITETB. Strengthening and decentralizing this surveillance system are goals that have already been achieved, to the extent that the system is comprised of decentralized and integrated workflows, with the participation of health service managers from different government levels and health facilities. Identification of studies and evaluations using SITETB data,5,10,24,25 as well as stock control and control of medication shipments, which are activities routinely carried out by SITETB, enable the system’s usefulness to be recognized.
In order for DRTB-SS to be enhanced and strengthened, we recommend that national health service managers (i) review and update system objectives, (ii) prepare indicators and targets for DRTB-SS, (iii) update at the same time the paper form and the online form for notification on the SITETB system, (iv) evaluate the diagnosis network and identify priority areas for DRTB and (v) propose periodic evaluations of DRTB cases and data. We recommend that State and Municipal Tuberculosis Control Programs (i) periodically monitor cases and data, (ii) evaluate the diagnosis network and integration of information between SITETB and SINAN, as well as (iii) periodically providing training for SITETB users.
Acknowledgements
We thank Walter Ataalpa Freitas and Stefano Barbosa Codenotti for their collaboration with this study.
REFERENCES
1. World Health Organization. Global tuberculosis report 2018 [Internet]. Geneva: World Health Organization; 2018 [cited 2018 Out 22]. Available from: Available from: https://www.who.int/tb/publications/global_report/en/ [ Links ]
2. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Coordenação Geral de Desenvolvimento da Epidemiologia em Serviços. Guia de vigilância em saúde [Internet]. 3 ed. Brasília: Ministério da Saúde; 2019 [citado 2019 nov 8]. 740 p. Disponível em: Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2019/junho/25/guia-vigilancia-saude-volume-unico-3ed.pdf [ Links ]
3. Dalcolmo MP, Andrade MKDN, Picon PD. Tuberculose multirresistente no Brasil: histórico e medidas de controle. Rev Saúde Pública [Internet]. 2007 set [citado 2019 nov 8];41 Suppl 1:34-42. Disponível em: Disponível em: http://www.scielo.br/pdf/rsp/v41s1/6570.pdf . doi: 10.1590/S0034-89102007000800006 [ Links ]
4. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde.Centro de Referência Professor Hélio Fraga. Projeto MSH. Tuberculose multirresistente - guia de vigilância epidemiológica [Internet]. Rio de Janeiro: Ministério da Saúde; 2007 [citado 2019 nov 8]. 90 p. Disponível em: Disponível em: http://www5.ensp.fiocruz.br/biblioteca/dados/txt_670024370.pdf [ Links ]
5. Bartholomay P, Pinheiro RS, Pelissari DM, Arakaki-Sanchez D, Dockhorn F, Rocha JL, et al. Sistema de informação de tratamentos especiais de tuberculose: (SITETB): histórico, descrição e perspectivas. Epidemiol Serv Saúde [Internet]. 2019 jun-set [citado 2019 nov 8]; 28(2):e2018158. Disponível em: Disponível em: http://www.scielo.br/pdf/ress/v28n2/2237-9622-ress-28-02-e2018158.pdf . doi: 10.5123/s1679-49742019000200002 [ Links ]
5. Guidelines Work Group, German RR, Westmoreland D. Updated guidelines for evaluating public health surveillance systems. MMWR Morb Mortal Wkly Rep [Internet]. 2001 Jul [cited 2019 Nov 8];50(RR13):1-35. Available from: Available from: https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5013a1.htm [ Links ]
6. Malhão TA, Oliveira GP, Codennoti S, Moherdaui F. Avaliação da completitude do Sistema de Informação de Agravos de Notificação da Tuberculose, Brasil, 2001-2006. Epidemiol Serv Saúde [Internet]. 2010 jun-set [citado 2019 nov 8];19(3):245-56. Disponível em: Disponível em: http://scielo.iec.gov.br/pdf/ess/v19n3/v19n3a07.pdf . doi: 10.5123/S1679-49742010000300007 [ Links ]
7. Oliveira PB, Oliveira GP, Codenotti SB, Saraceni V, Nóbrega AA, Sobel J. Avaliação do sistema de vigilância da tuberculose no município do Rio de Janeiro, 2001 a 2006. Cad Saúde Coletiva [Internet]. 2010 [citado 2019 nov 8];18(3):337-46. Disponível em: Disponível em: http://www.cadernos.iesc.ufrj.br/cadernos/images/csc/2010_3/artigos/CSCv18n3_pag337-46.pdf.pdf [ Links ]
8. Silva GDM, Bartholomay P, Cruz OG, Garcia LP. Avaliação da qualidade dos dados, oportunidade e aceitabilidade da vigilância da tuberculose nas microrregiões do Brasil. Ciênc Saúde Coletiva [Internet]. 2017 out [citado 2019 nov 8];22(10):3307-19. Disponível em: Disponível em: http://www.scielo.br/pdf/csc/v22n10/1413-8123-csc-22-10-3307.pdf . doi: 10.1590/1413-812320172210.18032017 [ Links ]
9. Wilhelm D, Rodrigues MV, Nakata PT, Godoy SC, Blatt CR. Descentralização do acesso ao sistema de informações de tratamentos especiais em tuberculose. Rev Baiana Enferm [Internet]. 2018 [citado 2019 nov 8];32:e25134. Disponível em: Disponível em: https://portalseer.ufba.br/index.php/enfermagem/article/view/25134 . doi: 10.18471/rbe.v32.25134 [ Links ]
10. Ministério da Saúde (BR). Departamento de Informática do SUS (Tabnet): demográficas e socioeconômicas. Projeção da população do Brasil por sexo e idade simples [Internet]. Brasília: Ministério da Saúde ; 2019. Disponível em: http://www2.datasus.gov.br/DATASUS/index.php?area=0206 [ Links ]
11. Zhao Y, Xu S, Wang L, Chin DP, Wang S, Jiang G, et al. National survey of drug-resistant tuberculosis in China. N Engl J Med [Internet]. 2012 Jun [cited 2019 Nov 8];366(23):2161-70. Available from: Available from: https://www.nejm.org/doi/full/10.1056/NEJMoa1108789 . doi: 10.1056/NEJMoa1108789 [ Links ]
12. Kuyinu YA, Odugbeni BA, Salisu-Olatunji SO, Adepoju FO, Odusanya OO. Characteristics of mycobacterium tuberculosis positive patients screened for drug-resistant tuberculosis at a tertiary health facility in Lagos, Nigeria. J Natl Med Assoc [Internet]. 2017 Feb [cited 2019 Nov 8];110(1):88-91. Available from: Available from: https://www.sciencedirect.com/science/article/abs/pii/S0027968417300147?via%3Dihub . doi: 10.1016/j.jnma.2017.04.007 [ Links ]
13. Correia LOS, Padilha BM, Vasconcelos SML. Métodos para avaliar a completitude dos dados dos sistemas de informação em saúde do Brasil: uma revisão sistemática. Ciênc Saúde Coletiva [Internet]. 2014 nov [citado 2019 nov 8];19(11):4467-78. Disponível em: Disponível em: http://www.scielo.br/pdf/csc/v19n11/1413-8123-csc-19-11-4467.pdf . doi: 10.1590/1413-812320141911.02822013 [ Links ]
14. Santos ML. Fatores associados à subnotificação de TB e Aids, durante os anos de 2001 a 2010, a partir do Sinan [dissertação]. Recife (PE): Fundação Instituto Oswaldo Cruz; 2014. Disponível em: https://www.arca.fiocruz.br/handle/icict/12257 [ Links ]
Souza VMM, Brant JL, Arsky MLS, Araújo WN. Avaliação do sistema nacional de vigilância epidemiológica da leptospirose - Brasil, 2007. Cad Saúde Coletiva [Internet]. 2010 [citado 2019 nov 8];18(1):95-105. Disponível em: Disponível em: http://www.cadernos.iesc.ufrj.br/cadernos/images/csc/2010_1/artigos/Modelo%20Livro%20UFRJ%209-a.pdf [ Links ]
15. Dunn J, Andreoli SB. Captura e recaptura: nova metodologia para pesquisas epidemiológicas. Rev Saúde Pública [Internet]. 1994 dez [citado 2019 nov 8];28(6):449-53. Disponível em: Disponível em: http://www.scielo.br/pdf/rsp/v28n6/09.pdf . doi: 10.1590/S0034-89101994000600009 [ Links ]
16. Abdolmugnhi J, Abdulwareth Y, Hamady A, Mahyoub E, Alagbari A, Al-Serouri A. Evaluation of multi drug resistant tuberculosis surveillance system, Yemen. Iproc [Internet]. 2018 Mar [cited 2019 Nov 8];4(1):e10538. Available from: Available from: https://www.iproc.org/2018/1/e10538/ [ Links ]
17. Belanger A, Morris BS, Brostrom R, Yost D, Goswami N, Oxtoby M, et al. Introduction and evaluation of multidrug-resistant tuberculosis supplemental surveillance in the United States. J Clin Tuberc Other Mycobact Dis [Internet]. 2019 May [cited 2019 Nov 8];15(2019):100090. Available from: Available from: https://www.sciencedirect.com/science/article/pii/S2405579418300895 . doi: 10.1016/j.jctube.2019.01.005 [ Links ]
18. Costa PV, Machado MTC, Oliveira LGD. Adesão ao tratamento para Tuberculose Multidroga-Resistente (TBMDR): estudo de caso em ambulatório de referência, Niterói (RJ), Brasil. Cad Saúde Coletiva [Internet]. 2019 jan-mar [citado 2019 nov 8];27(1):108-15. Disponível em: Disponível em: http://www.scielo.br/pdf/cadsc/v27n1/1414-462X-cadsc-1414-462X201900010292.pdf . doi: 10.1590/1414-462x201900010292 [ Links ]
19. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Brasil livre da tuberculose: evolução dos cenários epidemiológicos e operacionais da doença. Bol Epidemiol [Internet]. 2019 mar [citado 2019 nov 8];50(9):1-18. Disponível em: Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2019/marco/22/2019-009.pdf [ Links ]
20. Ministério da Saúde (BR). Agency for International Development (USA). System for improved access to pharmaceuticals and services. Brasil - pesquisa de avaliação dos usuários do SITE-TB, 2016. Arlington (VA): [S.n.]; 2016. [ Links ]
21. Ramalho DMP, Miranda PFC, Andrade MK, Brígido T, Dalcolmo MP, Mesquita E, et al. Outcome in patients with presumed drug resistant tuberculosis in five reference centers in Brazil. BMC Infect Dis [Internet]. 2017 Aug [cited 2019 Nov 8];17(1):571. Available from: Available from: https://bmcinfectdis.biomedcentral.com/articles/10.1186/s12879-017-2669-1 . doi: 10.1186/s12879-017-2669-1 [ Links ]
22. Viana PVS, Redner P, Ramos JP. Fatores associados ao abandono de tuberculose drogarresistente (TBDR) atendidos em um centro de referência no Rio de Janeiro, Brasil. Cad Saúde Pública [Internet]. 2018 [citado 2019 nov 8];34(5):e00048217. Disponível em: Disponível em: http://www.scielo.br/pdf/csp/v34n5/1678-4464-csp-34-05-e00048217.pdf . doi: 10.1590/0102-311x00048217 [ Links ]
23. Fregona G, Cosme LB, Moreira CMM, Bussular JL, Dettoni VV, Dalcolmo MP, et al. Fatores associados à tuberculose resistente no Espírito Santo, Brasil. Rev Saúde Pública [Internet]. 2017 abr [citado 2019 nov 8];51:41. Disponível em: Disponível em: http://www.scielo.br/pdf/rsp/v51/pt_0034-8910-rsp-S1518-87872017051006688.pdf . doi: 10.1590/s1518-8787.2017051006688 [ Links ]
Received: June 26, 2019; Accepted: October 18, 2019











 texto em
texto em 


