Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.29 no.2 Brasília 2020 Epub 22-Abr-2020
http://dx.doi.org/10.5123/s1679-49742020000200018
ORIGINAL ARTICLE
Occurrence of hepatitis B in pregnant women and follow-up of exposed children in the State of São Paulo, Brazil, in 2012*
1Secretaria de Estado da Saúde, Centro de Vigilância Epidemiológica ‘Professor Alexandre Vranjac’, São Paulo, SP, Brazil
2Secretaria de Estado da Saúde, Superintendência de Controle de Endemias do Estado de São Paulo, São Paulo, SP, Brazil
3Secretaria de Estado da Saúde, Instituto Adolfo Lutz, São Paulo, SP, Brazil
4Secretaria Municipal da Saúde, Divisão de Vigilância Epidemiológica, São Paulo, SP, Brazil
5Universidade de São Paulo, Instituto de Medicina Tropical, São Paulo, SP, Brazil
6Secretaria de Estado da Saúde, Centro de Vigilância Epidemiológica ‘Professor Alexandre Vranjac’, São Paulo, SP, Brazil
Objective:
to describe the occurrence of hepatitis B among pregnant women, immunoprophylaxis and vertical and perinatal transmission in children exposed to the virus in the São Paulo state primary care network, Brazil.
Methods:
this was a cross-sectional study using prenatal records of pregnant women attending health services between January and June 2012 and a cohort of newborns; the frequencies of the results were described and the estimated occurrence of hepatitis B was calculated.
Results:
6,233 pregnant women were included, of whom 53.1% were between 20-29 years old, 58.7% had 8-11 years of schooling, 53.3% were white, and 73.9% lived with a partner; occurrence of hepatitis B was 0.13% (95%CI 0.04; 0.21); of the eight children of mothers with chronic hepatitis B, six had a complete vaccination schedule, and there was no vertical or perinatal transmission.
Conclusion:
there was low occurrence of hepatitis B in pregnant women and absence of vertical or perinatal transmission.
Keywords: Hepatitis B; Pregnant Women; Prevalence; Epidemiologic Studies; Vaccination; Infectious Disease Transmission, Vertical
Introduction
Hepatitis B is caused by a DNA virus belonging to the Hepadnaviridae family. This virus is considered to be oncogenic and has liver cell tropism. Infection investigation is done by means of testing for hepatitis B surface antigens (HBsAg). The virus is most frequently transmitted during sex, although it is also transmitted parenterally (sharing needles, syringes and other sharp objects) and by dental and surgical procedures that fail to comply with biosafety norms; it can also be transmitted from mother to child, mainly at childbirth and in the perinatal period.
Hepatitis B virus (HBV) infection can cause acute or chronic hepatitis. People with chronic infection are at risk of developing hepatocellular carcinoma, cirrhosis or both.1 HBV infection is a global Public Health problem, due to its high transmissibility, although its global disease burden has reduced over the last three decades owing to large-scale vaccination of the population.2,3
The risk of infants who are infected through vertical transmission developing chronic infection is greater than 90% if they do not receive adequate immunoprophylaxis: first vaccine dose at birth; and specific hepatitis B immune globulin (HBIG), preferably in the first 12 hours of life.4
There are few population-based studies on the magnitude of hepatitis B among parturient pregnant women, nor about vertical transmission among infants exposed to the virus.5,6 At the time of submitting this article for publication, the magnitude of HBV infection in pregnant women and its transmission to newborn babies in the state of São Paulo is not known.
The objective of this study was to describe occurrence of hepatitis B among pregnant women, immunoprophylaxis and vertical and perinatal transmission to infants exposed to the virus attending Primary Healthcare services in the state of São Paulo, Brazil.
Methods
A cross-sectional study was conducted to measure occurrence of hepatitis B among pregnant women attending prenatal consultations in the state of São Paulo between January 1st and June 30th 2012. Moreover, a cohort was formed comprised of children born to mothers diagnosed as having hepatitis B, in order to assess (i) newborn baby immunoprophylaxis in maternity hospitals and (ii) follow-up at primary healthcare centers responsible for the care of these children, access to vaccination coverage and collection of blood for laboratory tests.
In 2019 the state of São Paulo had a population of 44,434,333 inhabitants (22,748,886 females and 21,566,044 males), covering an area of de 248,219.63km2 and comprised of 645 municipalities. The human development index (HDI) for that year was 0.783.7 There are around 4,400 Primary Healthcare (PHC) centers in the state and PHC population coverage is 62.9%.8
The study was conducted in three stages: the first stage involved gathering secondary data from medical records and other records of pregnant women attending National Health System Prenatal Care Network services in the state of São Paulo (SUS-SP); the second stage involved collecting secondary data providing information about infant immunoprophylaxis at the maternity hospital where mothers identified as having hepatitis B had given birth; the third stage comprised gathering secondary data on infant vaccination coverage and primary blood collection from infants for laboratory testing, with the aim of detecting diagnosis of vertical or perinatal transmission. The third and final stage of the study took place in PHC centers, shortly after the exposed infants were 18 months old. The choice of the 18 month-old age range is justified by the fact of antibodies passing through the placenta and establishing themselves in the fetus and, therefore, a positive test result prior to 18 months of age being due to the presence of maternal antibodies.9
This study took the case definition of hepatitis B in pregnant women to be: presence of reactive HBsAg in the prenatal period (excluding false positive results, or HBsAg due to vaccination), confirmed by the presence of Anti-HBc, based on information collected at PHC centers, maternity hospitals or follow-up reference services for pregnant women. HBV infection in exposed children is confirmed by reactive HBsAg test results.9
First of all a sample database was built using Prenatal Information System (SISPRENATAL) records. This system was chosen because it includes all prenatal consultations by pregnant women at PHC centers, as well as providing the number of consultations attended.
The sample database included all pregnant women who attended three or more prenatal consultations in the SUS-SP Primary Healthcare Network and at Primary Healthcare centers between January 1st and June 30th 2012. These criteria were chosen in order to improve the completeness of information regarding the results of hepatitis B serological tests. The sample size was calculated based on 0.7% estimated hepatitis B prevalence among pregnant women according to previous studies,10-14 0.2% margin of error plus a further 20%. The final estimated sample size for the study was 8,000 pregnant women attending SUS-SP facilities.
The state of São Paulo is divided into 17 Regionalized Health Care Networks (RRAS). For the purposes of sampling, the Primary Healthcare centers were grouped into six strata, according to the RRAS, so as to include pregnant women using these services in all regions of the state. The sample design was of the stratified type, divided proportionally to the size of each stratum.15
The sample of pregnant women was drawn from the SISPRENATAL system which provided a list with the names of the Primary Healthcare centers and the number of consultations attended by pregnant women. In order to overcome heterogeneity in the number of consultations at the Primary Healthcare centers, the centers were grouped into each of the six strata so as to comprise a set of sample units to be drawn from, whereby each sample unit was comprised of at least 200 consultations. Information on 100 women was taken from each of these groups. In each stratum samples were drawn systematically, with an interval proportional to the number of sample units in each stratum, starting at random.
Data on the pregnant women was collected from prenatal medical records kept at the Primary Healthcare centers; as well as from other records available at the service when medical records were incomplete (matrix database of the municipality of São Paulo, pregnant women’s medical records at other health services, laboratory management systems). Structured questionnaires were prepared for this purpose. The questionnaires were validated in a pilot test and filled in by duly trained researchers responsible for collecting data in the same units so as to minimize biases.
The following variables were collected on pregnant women:
age, in years (<15; 15-19; 20-29; 30-39; 40 and over);
schooling in years of study (none; 1-7; 8-11; 12 or more);
race/skin color (white; brown; black; indigenous; yellow);
marital status (has a partner; no partner);
number of prenatal consultations (3-5; 6-11; 12 or more);
diagnosis of syphilis (yes; no);
positive HIV test result HIV (yes; no); and
hepatitis B HBsAg and Anti-HBc (total) serological test results (reactive; not reactive).
In addition, in the case of HBsAg reactive pregnant women, information was collected about serology of other hepatitis B markers, including Anti-HBc, in order to exclude false positive results (isolated HBsAg with negative Anti-HBc), presence of markers due to vaccination and checking for possible typing errors.
With regard to exposed children, the following information was gathered at the maternity hospital where the child was born: (i) 1st dose of hepatitis B vaccine at birth (yes; no; no information); and (ii) whether HBIG was administered (yes; no; no information). Information was also sought at Primary Healthcare reference centers that children were referred to for follow-up, in order to collect data on vaccination coverage and to collect blood samples for laboratory tests for all children over 18 months old, in accordance with the protocol adopted by the study. Vaccination cards and other information systems were consulted to check whether the children had received three doses of hepatitis B vaccine by the time they were 6 months old (vaccination complete: yes; no; not located), as recommended by the National Immunization Program (PNI).16
The blood samples were collected on filter paper using a finger prick. The blood was diluted in an appropriate buffer for subsequent serological testing. The tests were performed at the São Paulo Adolfo Lutz Institute Virology Center Hepatitis Laboratory (IAL). The HBsAg and Anti-HBc (total) serological markers were tested in all samples (reactive; non-reactive; not located). When the HBsAg/Anti-HBc (total) sample results were not reactive, an Anti-HBs marker test was performed to check for vaccine seroconversion; while for samples with a reactive HBsAg result, a further blood sample was collected in order to test for viral load and HBV S region amplification, for subsequent genotyping (Figure 1).
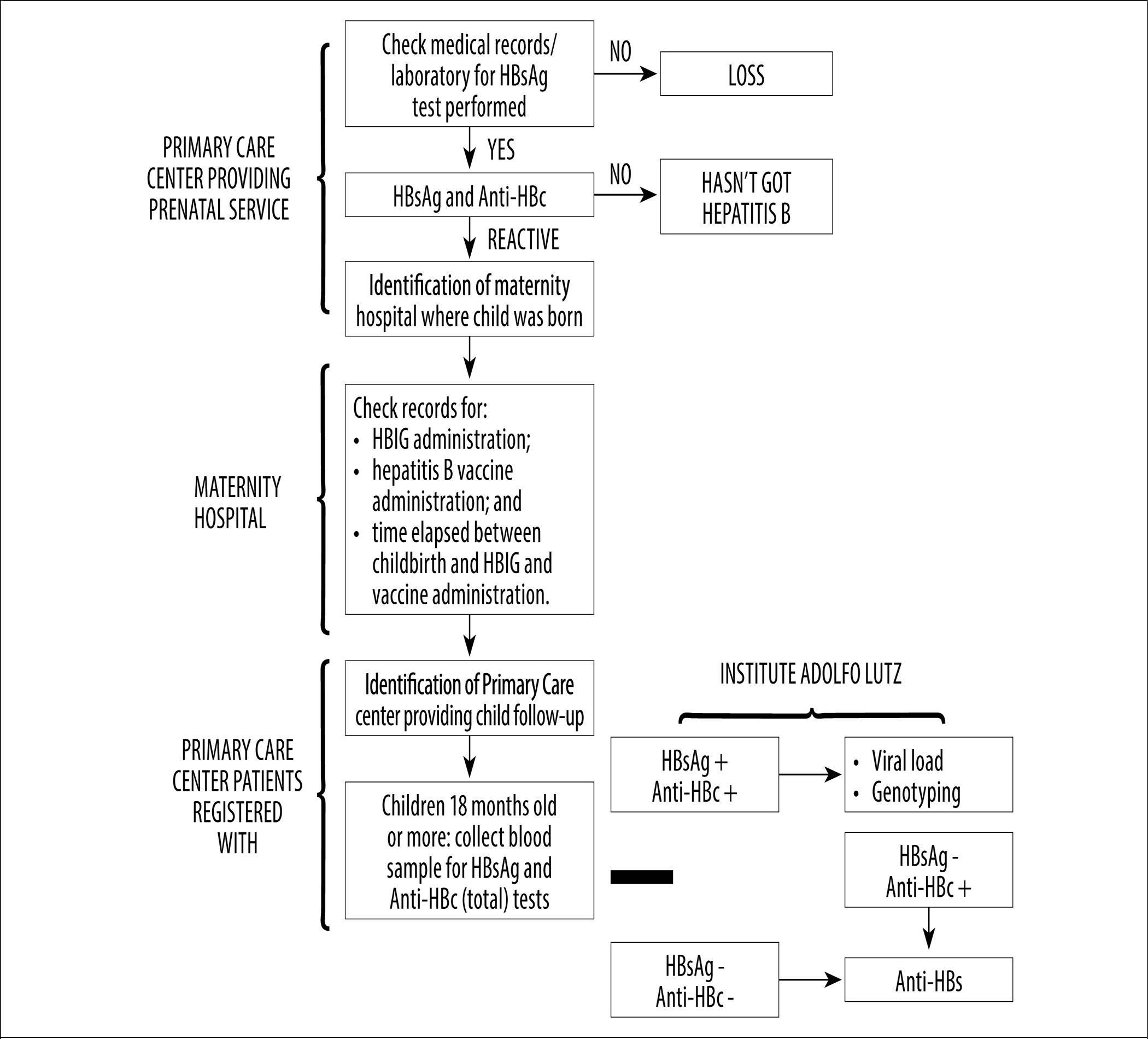
Figure 1 - Flowchart of hepatitis B virus investigation in pregnant women and children attending National Health System Prenatal Care Network services in the state of São Paulo, January-June 2012
The data were analyzed using Statistical Package for Social Science (SPSS), IBM, version 26.0, for Windows.
The characteristics of the pregnant women are shown in absolute and relative frequencies. The following were calculated: (i) estimated occurrence of hepatitis B in pregnant women (number of pregnant women with confirmed diagnosis/number of pregnant women in the sample x 100), taking 95% confidence intervals (95%CI); (ii) number of HBV vertical/perinatal transmission cases among exposed infants; and (iii) proportion of newborn babies receiving immunoprophylaxis in the first 24 hours of life.
The study project was approved by the São Paulo City Health Department Research Ethics Committee (Opinion No. 328.584, dated 08/07/2013) and by the São Paulo Adolfo Lutz Institute Research Ethics Committee (Protocol No. 008/2013, dated 17/09/2013). All mothers and their children who were asked to provide blood samples were informed about the procedure protocol and were invited to take part in the study following their formal agreement by signing a Free and Informed Consent form.
In accordance with National Health Council Resolution No. 441, dated May 12th 2011, all biological samples collected and sent to IAL were stored in appropriate freezers and only used for the purposes of the study. The samples were duly disposed of when the project ended.
Results
The sample database was comprised of 110,253 consultations recorded on the SISPRENATAL system at 2,552 Primary Healthcare centers. The sample obtained from this was 8,000 consultations by pregnant women at 597 centers. The data on 7,982 pregnant women who had had prenatal care via the SUS Primary Healthcare network in the state of São Paulo (99.8% of the planned sample) were studied and 6,233 (78%) of them were considered eligible for analysis (Figure 2). There were 1,749 pregnant women whose medical records or other results did not contain serological test results and, therefore, they were not included.
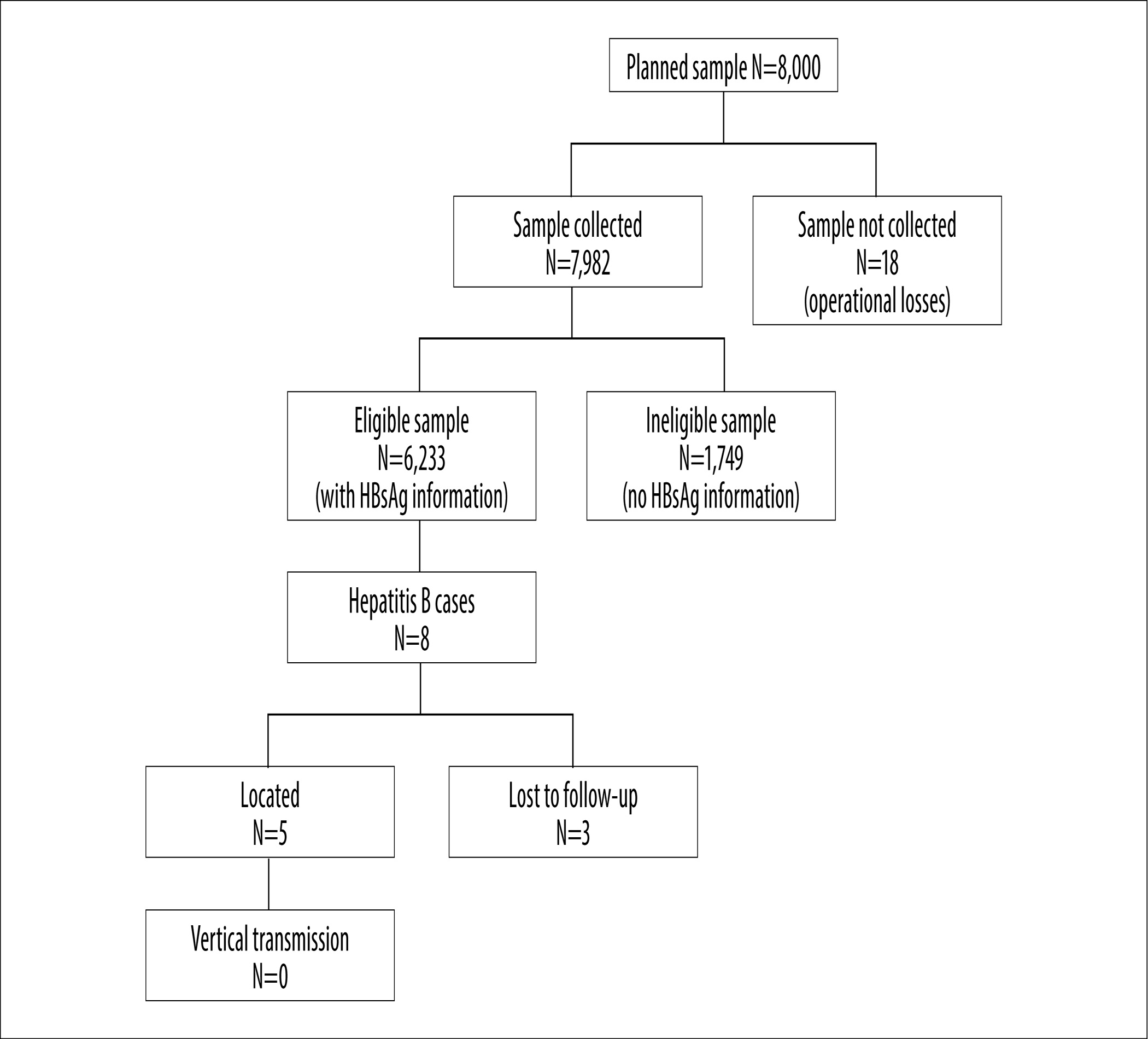
Figure 2 - Flowchart of the sample collected from pregnant women attending Brazilian National Health System Primary healthcare prenatal services and results of vertical and perinatal transmission investigation in children born to mothers with the hepatitis B virus, São Paulo state, January-June 2012
With regard to the characteristics of the pregnant women, the majority were 20-29 years old (53.1%), had 8-11 years of schooling (58.7%), were of white race/skin color (53.3%) and had a partner (73.9%). Approximately 80,0% (4,988) of them had had six or more prenatal consultations. The proportion of pregnant women with records of positive syphilis test results during pregnancy, either current or previous, was 1.6%; while the proportion of women with positive HIV test results was 0.4% (Table 1). The medical records and other data sources of 2,917 (46.8%) pregnant women contained information showing that they had had full hepatitis B vaccination (three doses).
Table 1 - Sociodemographic and clinical characteristics of pregnant women attending Brazilian National Health System Primary Healthcare Prenatal services in the state of São Paulo, January-June 2012
| Sociodemographic and clinical characteristics | n | % |
|---|---|---|
| Age range (in years | ||
| <15 | 46 | 0.7 |
| 15-19 | 1,118 | 18.1 |
| 20-29 | 3,270 | 53.1 |
| 30-39 | 1,605 | 26.0 |
| ≥40 | 127 | 2.1 |
| Total | 6,166 | 100.0 |
| Schooling (in years of study) | ||
| None | 26 | 0.6 |
| 1-7 | 1,108 | 28.7 |
| 8-11 | 2,269 | 58.7 |
| ≥12 | 463 | 12.0 |
| Total | 3,866 | 100.0 |
| Race/skin color | ||
| White | 2,563 | 53.3 |
| Brown | 1,727 | 36.0 |
| Black | 454 | 9.4 |
| Indígena | 12 | 0.2 |
| Amarela | 54 | 1.1 |
| Total | 4,810 | 100.0 |
| Marital status | ||
| Has a partner | 2,847 | 73.9 |
| No partner | 1,007 | 26.1 |
| Total | 3,854 | 100.0 |
| Number of prenatal consultations | ||
| 3-5 | 1,235 | 19.9 |
| 6-11 | 4,247 | 68.2 |
| ≥12 | 741 | 11.9 |
| Total | 6,223 | 100.0 |
| Diagnosis of syphilis | ||
| Yes | 78 | 1.6 |
| No | 4,701 | 98.4 |
| Total | 4,779 | 100.0 |
| Positive HIVa test result | ||
| Sim | 20 | 0.4 |
| Não | 5,498 | 99.6 |
| Total | 5,518 | 100.0 |
a)HIV: human immunodeficiency virus.
Reactive HBsAg serological results were found on the prenatal medical records of 14 pregnant women (0.2%). Following investigation of serology information, eight reactive Anti HBc cases were confirmed, resulting in occurrence of 0.13% (95%CI: 0.04;0.21).
When comparing the characteristics of pregnant women who had information about HBsAg with pregnant women for whom HBsAg data was missing, those for whom there was no information were found to have less schooling, a lower number of prenatal consultations and a higher proportion of recorded syphilis cases. The remaining characteristics had no important comparable differences among the 6,233 eligible pregnant women.
Table 2 shows the results of the analysis of data from the maternity hospitals and follow-up at Primary Healthcare centers in relation to the eight infants born to women diagnosed as having chronic hepatitis B. Seven newborn babies were found to have received their first dose of hepatitis B vaccine at the maternity hospital following childbirth, and four received HBIG in the first 24 hours of life. Three infants did not receive immune globulin due to a Ministry of Health stockout during the period studied. With regard to vaccination against hepatitis B, six infants had a complete vaccination schedule: 3 doses by the time they were 6 months old.
Table 2 - Results of immunoprophylaxis at maternity hospital, child follow-up and serological tests on newborn babies exposed to the hepatitis B virus among pregnant women attending Brazilian National Health System Primary Healthcare Prenatal services in the state of São Paulo, January-June de 2012
| Case | Care at maternity hospital | Follow-up at Primary Healthcare centera | ||||
|---|---|---|---|---|---|---|
| HBIGb | First dose of hepatitis B vaccine | Hepatitis B vaccine | Serological test result | |||
| HBsAgc | Anti-HBc (total)d | Anti-HBse | ||||
| 1 | No | Yes | Completef | NRh | NRh | Ri |
| 2 | No | Yes | Completef | NRh | NRh | Ri |
| 3 | No | Yes | NLg | NLg | NLg | NLg |
| 4 | Yes | Yes | Completef | NRh | NRh | Ri |
| 5 | Yes | Yes | Completef | NRh | NLg | NLg |
| 6 | Yes | Yes | Completef | NRh | NRh | Ri |
| 7 | Yes | Yes | Completef | NRh | NRh | NRh * |
| 8 | No information | No information | NLg | NLg | NLg | NLg |
a) Primary Healthcare center: (Unidade Básica de Saúde - UBS).
b) HBIG: specific hepatitis B immune globulin.
c) HBsAg: hepatitis B surface antigens.
d) Anti-HBc (total): antibody to hepatitis B core antigen.
e) Anti-HBs: antibody against the surface antigens of the hepatitis B virus.
f) Complete: 3 doses of vaccine.
g) NL: not located at the time of the study.
h) NR: not reactive.
i) R: reactive.
*Case in which seroconversion was not found and the vaccine schedule was repeated, with reactive Anti-HBs result.
Among the eight cases of mothers identified as having hepatitis B, three children were not located for follow-up. In one case, the child had moved to another state and information was obtained that it was having follow-up at a Primary Healthcare center, with a complete vaccination schedule and non-reactive serology for hepatitis B. In the second case, the mother was a foreign migrant and returned to her country of origin after the child was born and no further information was obtained. In the third case, the mother was in a situation of social vulnerability and was lost to follow-up as attempts to ensure she attended the health service were unsuccessful. Five of the eight cases were located for interview and blood sample collection and all these children had non-reactive HBsAg and Anti-HBc serological test results. Four of the children had hepatitis B vaccine seroconversion (reactive Anti-HBs). With regard to the case for which seroconversion was not found, the Primary Healthcare center was advised to repeat the schedule: a new Anti-HBs serological test was performed (between 30 and 60 days after the last dose), and the result was reactive.
Discussion
The pregnant women studied here were, for the most part, young, of white race/skin color and had eight or more years of schooling. The majority of them lived with a partner and had had an adequate number of prenatal consultations, suggesting that they were women with a profile of social inclusion who adhere or have access to prenatal care in Primary Healthcare centers.
Less schooling, a lower number of consultations and a higher percentage of syphilis in current or previous pregnancy among the women for whom information about hepatitis B was not found, may suggest that they were not tested because of less adherence to the service and/or greater social vulnerability, this hindering prenatal follow-up.
Low hepatitis B occurrence was found in pregnant women in the SUS-SP Prenatal Care Network. The main findings from follow-up of the children exposed to HBV revealed adequate coverage of the first dose of vaccine at birth, adequate complete vaccination coverage and inexistence of vertical or perinatal transmission.
The results showed that hepatitis B occurrence in pregnant women was similar to occurrence found in developed countries and in areas of low endemicity in Brazil. A study conducted in Norway, which recruited pregnant women between October 2003 and October 2004 (n=1,668), revealed HBsAg prevalence of 0.1%.17 In the United States, hepatitis B prevalence in a representative sample of childbirths throughout the entire country between 1998 and 2011 among women who were 50 years old or less was 0.09%.18
A literature review of hepatitis B distribution in Brazil, which included 25 studies conducted between 2000 and 2014, showed prevalence in pregnant women varying between 0.0% and 8.7%.19 The data found in our study corroborate data reported by studies conducted in Brazil in the second half of the 2000s in areas of low hepatitis B prevalence among pregnant women.19,20 A study conducted in 2012 in the municipality of Itajaí, in Southern Brazil, found hepatitis B prevalence of 0.4% among 10,147 pregnant women.5 Another study, based on health system records about HBsAg testing in pregnant women having prenatal care between 2007 and 2012 in the municipality of Maceió, in Northeastern Brazil, also found prevalence of 0.4%.21
It is noteworthy that prevalence of HBV infection in pregnant women is commonly used as a proxy for the general population geral.22 A population-based study conducted in state capital cities in Brazil’s Northern, Southeastern and Southern regions between 2005 and 2009, found HBsAg prevalence of 0.37% in the 10 to 69 year-old population.23,24
Adequate coverage of the first dose of vaccine at birth meets the recommendations for close care during pregnancy and prevention of vertical transmission. Vaccination against hepatitis B was introduced in Brazil in 1992 for specific groups,25 and since then has gradually expanded until being made universally available to all age ranges in 2016.16 In the state of São Paulo, the vaccine was introduced in 2005 for all liveborn babies in the first 12 hours of life. The care procedures for pregnant and puerperal women in the SUS-SP recommend serological screening in the prenatal period, using the HBsAg marker, with the aim of preventing vertical transmission.
In addition to the first dose of vaccine, administration of specific immune globulin is prescribed for newborns exposed to HBV in the first 24 hours following childbirth.26,27 However, provision of HBIG in the first 24 hours was found to be inadequate in this study. Immunoprophylaxis with HBIG within 24 hours after child delivery, combined with hepatitis B vaccination of the newborn, stops mother-to-child HBV transmission in 80 to 95% of cases.3 By itself, hepatitis B vaccination is 75 to 95% effective in preventing vertical transmission of hepatitis B when administered within 24 after childbirth.28
The World Health Organization’s (WHO) plan for the elimination of viral hepatitis has targets to reduce new infections by 90% and hepatitis mortality by 65%, between 2015 and 2030. The five strategic objectives to achieve elimination of infection include immunization against hepatitis B and prevention of mother-to-child HBV transmission (vertical or perinatal transmission).2
Despite the relevance of the data presented for Public Health, limitations of this study need to be highlighted. The population of pregnant women was studied based on secondary data which are subject to potential biases found in this type of data collection from official records.29 There was no information recorded about HBsAg serologies during pregnancy for some 22% of the pregnant women. As losses were not random, the sample can only be considered to be representative of pregnant women cared for on the SUS with assessment recorded on their medical records. Another limitation of this study relates to the possibility of records and notes on the variables being modified at different times. Furthermore, missing data that cannot be checked with the person who filled in the medical records, or inadequate records, can sometimes lead to incorrect classifications and information bias.29 Nevertheless, the use of large official databases to comprise the sample, as well as other information available through the Brazilian National Health System, arising from service provided to pregnant women via the Prenatal Care Network at state level, reinforce the importance of this study.
In conclusion, low occurrence of hepatitis B in pregnant women was found, with no vertical or perinatal transmission to babies exposed to the virus in the SUS-SP Primary Healthcare Network. It is important to emphasize that the results obtained can provide input for the SUS, informing its managers and staff as to analysis and evaluation of the situation, designing and implementing actions aimed at viral hepatitis prevention and control in mother and child healthcare. Finally, it is important to stress the need to improve the quality of information within National Health System Prenatal Care Network services in the state of São Paulo.
REFERENCES
1. McMahon BJ. Epidemiology and natural history of hepatitis B. Semin Liver Dis [Internet]. 2005;25Suppl1:3-8. Available from: https://doi.org/10.1055/s-2005-915644. [ Links ]
2. World Health Organization. Global hepatitis report, 2017 [Internet]. Geneva: World Health Organization; 2017 [citado 2019 Jan 27]. Available from: Available from: https://www.who.int/hepatitis/publications/global-hepatitis-report2017/en/ [ Links ]
3. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Protocolo clínico e diretrizes terapêuticas para hepatite B e coinfecções [Internet]. Brasília: Ministério da Saúde; 2017 [citado 2020 mar 29]. Disponível em: Disponível em: http://www.aids.gov.br/pt-br/pub/2016/protocolo-clinico-e-diretrizes-terapeuticas-para-hepatite-b-e-coinfeccoes [ Links ]
4. Schillie S, Vellozzi C, Reingold A, Harris A, Haber P, Ward JW, et al. Prevention of hepatitis B virus infection in the United States: recommendations of the advisory committee on immunization practices. MMWR Recomm Rep [Internet]. 2018 Jan [cited 2020 Mar 29];67(1);1-31. Available from: Available from: https://doi.org/10.15585/mmwr.rr6701a1 [ Links ]
5. Kupek E, Oliveira JF. Transmissão vertical do HIV, da sífilis e da hepatite B no município de maior incidência de AIDS no Brasil: um estudo populacional no período de 2002 a 2007. Rev Bras Epidemiol [Internet]. 2012 set [citado 2020 mar 29];15(3):478-87. Disponível em: Disponível em: https://doi.org/10.1590/S1415-790X2012000300004 [ Links ]
6. Liell AP, Weber D, Toscan C, Fornari F, Madalosso LF. Prevalência do HBsAg em gestantes de Passo Fundo, RS: estudo comparativo entre os sistemas de saúde público e privado. Arq Gastroenterol. 2009;46:75-7. Doi.org/10.1590/S00048032009000100018. [ Links ]
7. Fundação Sistema Estadual de Análise de Dados (SP). Portal de estatísticas do Estado de São Paulo [Internet]. São Paulo: Fundação Sistema Estadual de Análise de Dados; 2020 [citado 2020 jan 24]. Disponível em: Disponível em: http://seade.gov.br [ Links ]
8. Secretaria da Saúde do Estado de São Paulo. Atenção básica: organização do trabalho na perspectiva da longitudinalidade e da coordenação do cuidado [Internet]; XXX Congresso dos Secretários Municipais de Saúde do Estado de São Paulo; 2016 abr 13; São Paulo. São Paulo: Secretaria de Estado da Saúde; 2016 [citado 2020 jan 27]. Disponível em: Disponível em: http://www.cosemssp.org.br/downloads/Arnaldo-Sala.pdf [ Links ]
9. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância, Prevenção e Controle das Doenças Sexualmente Transmissíveis, Aids e Hepatites Virais. Manual técnico para o diagnóstico das hepatites virais [Internet]. Brasília: Ministério da Saúde; 2018 [citado 2019 ago 15]. Disponível em: Disponível em: http://www.aids.gov.br/pt-br/pub/2015/manual-tecnico-para-o-diagnostico-das-hepatites-virais [ Links ]
10. Reiche EMV, Morimoto HK, Farias GN, Hisatsugu KR, Geller L, Gomes ACLF, et al. Prevalência de tripanossomíase americana, sífilis, toxoplasmose, rubéola, hepatite B, hepatite C e da infecção pelo vírus da imunodeficiência humana, avaliada por intermédio de testes sorológicos, em gestantes atendidas no período de 1996 a 1998 no Hospital Universitário Regional Norte do Paraná. Rev Soc Bras Med Trop [Internet]. 2000 dez [citado 2020 mar 19];33(6):519-27. Disponível em: Disponível em: https://doi.org/10.1590/S0037-86822000000600002 [ Links ]
11. Perim EB, Passos AD. Hepatite B em gestantes atendidas pelo Programa do Pré-Natal da Secretaria Municipal de Saúde de Ribeirão Preto, Brasil: prevalência da infecção e cuidados prestados aos recém-nascidos. Rev Bras Epidemiol [Internet]. 2005 set [citado 2020 mar 19];8(3):272-81. Disponível em: Disponível em: https://doi.org/10.1590/S1415-790X2005000300009 [ Links ]
12. Arraes LC, Sampaio AS, Barreto S, Guilherme MAS, Lorenzato F. Prevalência de hepatite B em parturientes e perfil sorológico perinatal. Rev Bras Ginecol Obstret [Internet]. 2003 set [citado 2020 mar 19];25(8):571-6. Disponível em: Disponível em: https://doi.org/10.1590/S0100-72032003000800005 [ Links ]
13. Vázquez-Martinez JL, Coreño-Juárez MO, Montaño-Estrada LF, Attlan M, Gómez-Dantés H. Seroprevalence of hepatitis B in pregnant women in Mexico. Salud Púb Méx [Internet]. 2003 May-Jun [citado 2020 mar 29];45(3):165-70. Available from: Available from: http://saludpublica.mx/index.php/spm/article/view/6454/7864 [ Links ]
14. Braillon A, Nguyen-Khac E, Merlin J, Dubois G, Gondry J, Capron D. HBsAg screening during pregnancy in the French province Picardy. Gynecol Obstet Fertil [Internet]. 2010 Jan [cited 2020 Mar 19];(38):13-17. Available from: Available from: https://doi.org/10.1016/j.gyobfe.2009.11.002 [ Links ]
15. Silva NN. Amostragem probabilística. São Paulo: Editora da Universidade de São Paulo; 2001. [ Links ]
16. Ministério da Saúde (BR). Secretaria de Vigilância à Saúde. Departamento de Vigilância das Doenças Transmissíveis. Programa Nacional de Imunizações. Nota informativa nº 149, de 2015/CGPNI/DEVIT/SVS/MS. Informa mudanças no calendário nacional de vacinação para o ano de 2016 [Internet]. Brasília: Ministério da Saude; 2015 [citado 2020 mar 19]. Disponível em: Disponível em: http://www.aids.gov.br/pt-br/legislacao/nota-informativa-no-1492015 [ Links ]
17. Kristiansen MGL, Eriksen BO, Maltau JM, Holdo B, Guttenber TJ, Mortensen L,et al. Prevalences of viremic hepatitis C and viremic hepatitis B in pregnant women in Northern Norway. Hepatogastroenterology. 2009 Jul-Aug;56(93):1141-5. [ Links ]
18. Salemi JL, Spooner KK, Mejia de Grubb MC, Aggarwal A, Matas JL, Salihu HM. National Trends of Hepatitis B and C During Pregnancy Across Sociodemographic, Behavioral, and Clinical Factors, United States, 1998-2011. J Med Virol [Internet]. 2017 Jun [cited 2020 Mar 19];89(6):1025-1032. Available from: Available from: https://doi.org/10.1590/0037-8682-0176-2015 [ Links ]
19. Souto FJD. Distribution of hepatitis B infection in Brazil: the epidemiological situation at the beginning of the 21st century. Rev Soc Bras Med Trop [Internet]. 2016 Jan-Feb [cited 2020 Mar 19];49(1):11-23. Available from: Available from: https://doi.org/10.1590/0037-8682-0176-2015 [ Links ]
20. Souza MT, Pinho TL, Santos MD, Santos AD, Monteiro VL, Fonsêca LM, et al. Prevalence of hepatitis B among pregnant women assisted at the public maternity hospitals of São Luís, Maranhão, Brazil. Braz J Infect Dis [Internet]. 2012 Nov-Dec [cited 2020 Mar 19];(16):517-20. Available from: Available from: https://doi.org/10.1016/j.bjid.2012.07.008 [ Links ]
21. Moura AA, Mello MJG, Correia JB. Prevalence of syphilis, human immunodeficiency virus, hepatitis B virus, and human T-lymphotropic virus infections and coinfections during prenatal screening in an urban Northeastern Brazilian population. Int J Infect Dis [Internet]. 2015 Oct [cited 2020 mar 19];39:10-5. Available from: Available from: https://doi.org/10.1016/j.ijid.2015.07.022 [ Links ]
22. Hofstraat SHI, Falla AM, Duffell EF, Hahné SJM, Amato-Gauci AJ, Veldhuijzen IK, et al. Current prevalence of chronic hepatitis B and C virus infection in the general population, blood donors and pregnant women in the EU/EEA: a systematic review. Epidemiol Infect [Internet]. 2017 Oct [cited 2020 Mar 19];145(14):2873-85. Available from: Available from: https://doi.org/10.1017/S0950268817001947 [ Links ]
23. Ximenes RAA, Figueiredo GM, Cardoso MRAC, Stein AT, Moreira RC, Coral G, et al. Population-based multicentric survey of hepatitis b infection and risk factors in the north, south, and southeast regions of Brazil, 10-20 years after the beginning of vaccination. Am J Trop Med Hyg [Internet]. 2015 Dec [cited 2020 Mar 19]:93(6):1341-8. Available from: Available from: https://doi.org/10.4269/ajtmh.15-0216 [ Links ]
24. Fernandes CN, Alves MM, Souza ML, Machado GA, Couto G, Evangelista RA. Prevalence of seropositivity for hepatitis B and C in pregnant women. Rev Esc Enferm USP [Internet]. 2014 Feb [cited 2020 Mar 19];48(1):91-8. Available from: Available from: https://doi.org/10.1590/S0080-623420140000100011 [ Links ]
25. Governo do Estado de São Paulo. Secretaria de Estado de Saúde. Centro de Vigilância Epidemiológica ‘Professor Alexandre Vranjac’. Bol Epidemiol Paulista [Internet]. 2006 mar [citado 2020 mar 19];3(27):27-8. Disponível em: Disponível em: http://www.saude.sp.gov.br/resources/ccd/homepage/bepa/2006/27_marco_2006.pdf [ Links ]
26. Visser M, Van der Ploeg CPB, Smit C, Hukkelhoven CWPM, Abbink F, Van Benthem BHB, et al. Evaluating progress towards triple elimination of mother-to-child transmission of HIV, syphilis and hepatitis B in the Netherlands. BMC Public Health [Internet]. 2019 Mar [cited 2020 Mar 19];19(1):353. Available from: Available from: https://doi.org/10.1186/s12889-019-6668-6 [ Links ]
27. Governo do Estado de São Paulo. Secretaria da Saúde. Coordenadoria de Planejamento em Saúde. Assessoria Técnica em Saúde da Mulher. Atenção à gestante e à puérpera no SUS-SP: manual técnico do pré-natal e puerpério [Internet]. São Paulo: SES/SP; 2010 [citado 2020 mar 19]. 234 p. Disponível em: Disponível em: http://www.saude.sp.gov.br/resources/ses/perfil/gestor/destaques/atencao-a-gestante-e-a-puerpera-no-sus-sp/manual-tecnico-do-pre-natal-e-puerperio/manual_tecnicoii.pdf [ Links ]
28. Committee on Infectious Diseases, Committee on Fetus and Newborn. Elimination of perinatal hepatitis B: providing the first vaccine dose within 24 hours of birth. Pediatrics[Internet]. 2017 Sep [cited 2020 Mar 19];140(3):e20171870. Available from: Available from: https://doi.org/ 10.1542/peds.2017-1870 [ Links ]
29. Gordis L. Epidemiologia. 4. ed. Rio de Janeiro: Revinter; 2010. [ Links ]
*The study project, entitled ‘Hepatitis B in pregnant women and children exposed to the hepatitis B virus in the State of São Paulo’, received financial support from the Health Prevention and Protection Policies Support/Special Health Fund for Mass Immunization and Disease Control (GAPS)/FESIMA): Process No. 014/16.
Received: November 12, 2019; Accepted: March 06, 2020











 texto em
texto em 
 Curriculum ScienTI
Curriculum ScienTI

