Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.29 no.2 Brasília 2020 Epub 04-Maio-2020
http://dx.doi.org/10.5123/s1679-49742020000200027
Original article
Adverse event occurrence following use of tetanus, diphtheria and acellular pertussis adsorbed vaccine – Tdap –, São Paulo, SP, Brazil, 2015-2016 *
1Secretaria de Estado da Saúde, Coordenadoria de Ciência, Tecnologia e Insumos Estratégicos de Saúde, São Paulo, SP, Brazil
2Universidade de São Paulo, Faculdade de Medicina, São Paulo, SP, Brazil
3Universidade de São Paulo, Faculdade de Saúde Pública, SP, Brazil
4Secretaria de Estado da Saúde, Coordenadoria de Controle de Doenças, São Paulo, SP, Brazil
5Secretaria de Estado da Saúde, Coordenadoria de Controle de Doenças, São Paulo, SP, Brazil
Objective
to describe occurrence of adverse events following immunization (AEFI) with Tdap vaccine during pregnancy.
Methods
this was a descriptive study using data from reports by participants in an effectiveness and immunogenicity study conducted in two hospitals in São Paulo, SP, Brazil, from 2015 to 2016.
Results
of the 201 mothers included in the study, 48 (23.9%) had at least one AEFI; 60 symptoms related to Tdap use were identified – pain (22.4%), swelling (2.5%), fever (1.5%), somnolence (1.0%), redness (0.5%), vomiting (0.5%), headache (0.5%), local reaction (0.5%), and fatigue (0.5%); no rare, very rare, or extremely rare adverse events were reported; all events were considered to be expected, as they are described in the vaccine package insert; outcome of all events was recovery without sequelae.
Conclusion
Tdap vaccine in the form adopted by the National Immunization Program is safe; no unexpected adverse events were identified among vaccinated pregnant women.
Key words: Diphteria-Tetanus-acellular Pertusis Vaccines; Drug-Related Side Effects and Adverse Reactions; Safety; Surveillance
Introduction
Whooping cough is an acute bacterial respiratory disease, characterized by paroxysmal coughing. Babies and children under 2 years old comprise the group most at risk both of catching the disease and also suffering its complications and mortality.1
Adding the pertussis component to immunization coverage has led to a large reduction in whooping cough incidence globally. Notwithstanding, its transmission has never been interrupted.3
Likewise in Brazil, the increase in diphtheria, tetanus and pertussis (DTP) adsorbed vaccine coverage with effect from the 1990s was accompanied by a large reduction in whooping cough incidence. The immunization schedule used in Brazil consists of three doses in the first year of life – at 2, 4 and 6 months old – and two boosters – at 15 months and 4 years old.4
With effect from 2010, however, pertussis occurrence increased in several countries, such as the United States, Australia and Argentina; as well as in Brazil, despite high immunization coverage being maintained.1
Increased incidence was pronounced in Brazil between 2011 and 2014. A considerable proportion of cases occurred in children under 1 year old, although most cases were concentrated among the under-two-year-olds, who were too young to have the first DTP vaccine dose ( Figure 1 ).5
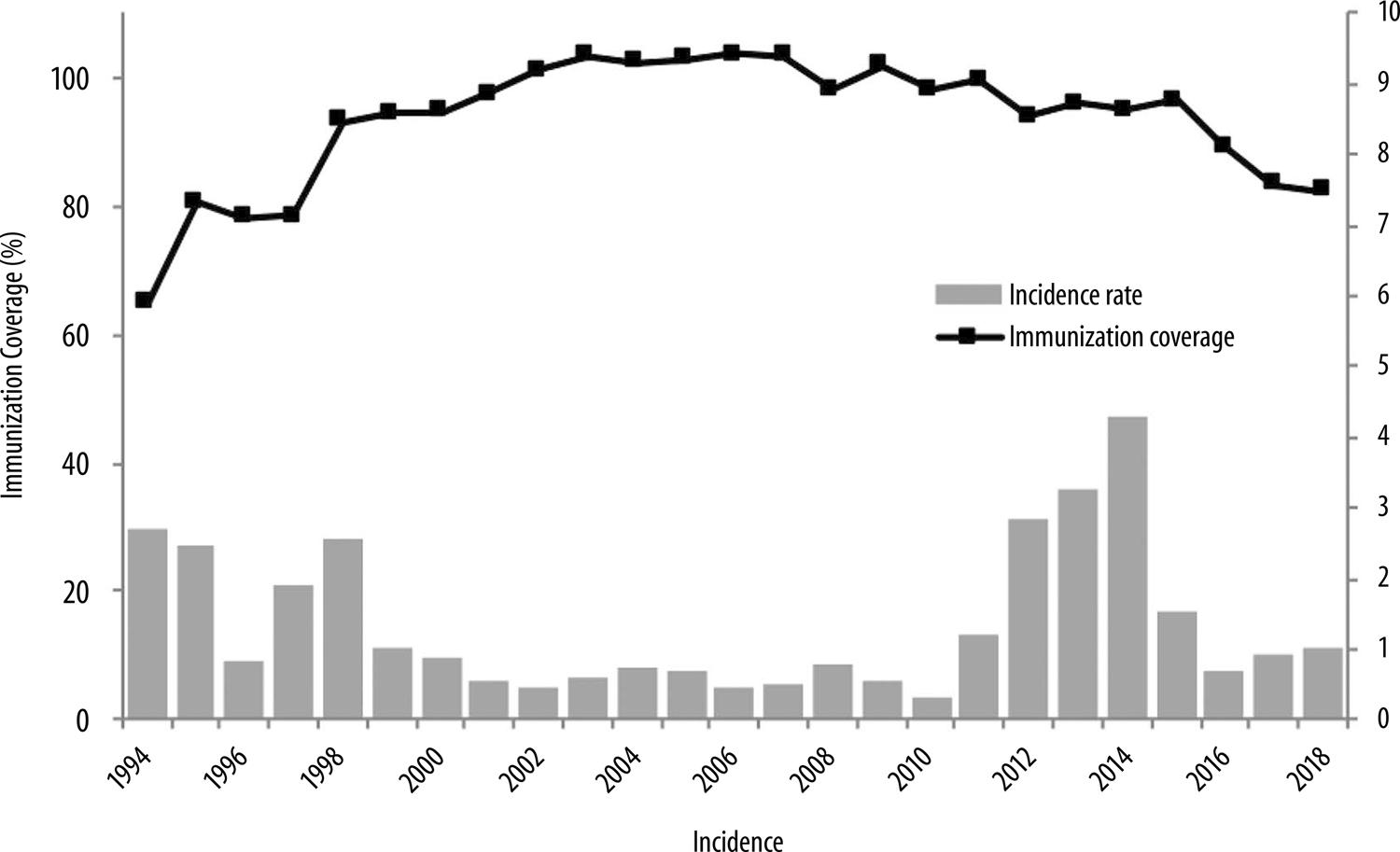
Sources:
General Coordination of Communicable Diseases, Health Surveillance Secretariat, Ministry of Health (CGDT/SVS/MS), 2018.
Meningitis Working Group.
Notifiable Health Conditions Information System (SINAN), updated as at 22/03/2019.
Figure 1 – Whooping cough incidence rate and DTP and DTP-Hib immunization coverage, Brazil, 1990-2018Notes:Incidence per 100,000 inhabitants, except neonatal tetanus – calculated per 100,000 infants under 1 year old.The general population with effect from 2016 was estimated by the Interagency Health Information Network (RIPSA) and by the Brazilian Institute of Geography and Statistics (IBGE), based on 2015 data.
In view of the reemergence of whooping cough and cases being concentrated among children younger than the age indicated for the vaccine to be administered, several countries adopted new strategies, in particular vaccinating pregnant women with vaccines containing acellular pertussis: tetanus, diphtheria and acellular pertussis adsorbed vaccine, known as Tdap vaccine.7
Vaccinating pregnant women enables transplacental transfer of antibodies to their babies before birth, protecting them during this period of greater care and monitoring until they reach vaccination age. In 2014, the Brazilian National Immunization Program started offering acellular vaccine (Tdap) to pregnant women at between 27 and 36 weeks of pregnancy. In 2017, the recommendation was extended to include pregnant women with effect from the 20th week of pregnancy.1
Considering that national data on vaccinating pregnant women with Tdap was virtually inexistent, the São Paulo State Immunization Program decided to conduct studies to enable evaluation of the effectiveness and safety of this procedure. One of these studies evaluated vaccination immunogenicity and safety.10
When the possibility of administering Tdap vaccine to pregnant women was first considered, knowledge about its safety in this case was limited. As such, the Advisory Committee on Immunization Practice (ACIP) of the United States Centers for Disease Control and Prevention (US CDC) reviewed data available on the Vaccine Adverse Events Reporting System (VAERS/US CDC), pregnancy records kept by the producers of Tdap vaccine and the results of smaller studies. Based on its review of those studies, the ACIP concluded that high risk or uncommon patterns of adverse events among pregnant women who received Tdap or among their newborn babies had not been found; the Committee considered it to be unlikely that the few severe adverse events reported had been caused by the vaccine.8 The same can be stated about booster doses among pregnant women who showed good tolerance to the product, whereby the event with most recorded occurrences was pain at the injection site. Moreover, none of the few severe adverse events reported were attributed to the vaccine. These findings are written on the package insert prepared by the producer of the tetanus, diphtheria and acellular pertussis adsorbed vaccine – Tdap –, which is the vaccine used by the Brazilian National Immunization Program.
The objective of this study was to describe the occurrence of adverse effects following immunization (AEFI) with Tdap during pregnancy.
Methods
This was a descriptive study of occurrence of adverse effects following immunization –AEFI – among pregnant women vaccinated with Tdap participating in an immunogenicity evaluation cohort study.11
The municipality of São Paulo, SP, is Brazil’s most populous municipality, with 11,253,503 inhabitants in 2010, according to the Brazilian Institute of Geography and Statistics (IBGE).12 It is one of the municipalities that concentrate Brazil’s economic activities and has a high human development index (HDI) (0.805 in 2010), although it is characterized by considerable socio-economic inequality.13
This study was conducted in two public state-level hospitals located in the outskirts of the city, namely the Leonor Mendes de Barros General and Maternity Hospital and the Interlagos General and Maternity Hospital, both of which deliver around 400 babies a month. This study is part of a larger study called ‘Immunization of pregnant women with adult acellular vaccine (Tdap): evaluation of reduction in whooping cough occurrence in babies and immune response of children’. Reduction in whooping cough occurrence was assessed by means of a case-control study.10 Immune response was analyzed by means of a cohort study of pregnant women and their children born in two public hospitals located in the municipality of São Paulo.11
Pregnant women were recruited when they were admitted to hospital to have their babies. Their socio-economic and demographic, obstetric and prenatal Tdap vaccination data were collected, as well as blood samples in order to study whooping cough antibody titers. Their babies were followed up from 2 to 7 months of age. Blood samples were also taken from the babies: from the umbilical cord; before the first childhood dose of vaccine containing the pertussis component (2 months old); and one month after the third dose of the vaccine. This study analyzed data related to AEFI due to Tdap administered during prenatal care of the cohort study participants.
Parturient women aged 18 and over receiving care in the above mentioned hospitals between July 2015 and August 2016 were eligible to take part in the study. However, the following were considered to be ineligible: (i) parturient women who received a blood transfusion or blood products in the three months prior to childbirth; (ii) those who received Tdap vaccine in the last 14 days of pregnancy; (iii) those who were diagnosed or suspected of having whooping cough during pregnancy; (iv) those whose gestational age was <37 weeks; (v) those with no information about Tdap vaccination date or epidemiological data; and (vi) mothers of babies whose birth weight was less than 2500 grams.
The minimum sample size was calculated using the following formula:
Where:
n = minimum sample size
z = confidence coefficient, the value of which was 1.96 for an alpha of 0.05
p = outcome frequency; we used p=15% AEFI occurrence
q = complement of that proportion (q=1-p)
e = absolute value of maximum error – 0.05
This resulted in:
The data were collected by means of an interview using a semi-structured questionnaire, administered during admission to hospital for child delivery. The questionnaire contained sociodemographic, clinical and epidemiological variables related to the pregnant women:
a) sociodemographic variables
- age range (in years: <20; 20-34; ≥35);
- schooling (in years of study: less than 8; more than 8; unknown);
- race/skin color (white; black; yellow; brown; indigenous);
- monthly family income (less than BRL 1,576; BRL 1,576 to BRL 3,151; more than BRL 3,152);
b) clinical and epidemiological variables
- gestational age at time of vaccination (in weeks: 17-26; 27-29; 30-33; 34-40; unknown);
- number of prenatal consultations (<7; ≥7; unknown); and
- adverse event occurrence and duration (in hours following vaccination: up to 24h; up to 48h; up to 72h; up to 96h; unknown).
Body mass index (BMI) was examined as a continuous variable, calculated based on self-reported maternal pre-pregnancy weight and height.
The interviews were conducted between July 2015 and August 2016.
The vaccine used was Boostrix (GSK Biologicals, Rixensart, Belgium), licensed in Brazil as Refortrix. The product contains 20 International Units (IU) of tetanus toxoid, 2 IU of diphtheria toxoid, 8 µg of inactivated pertussis toxoid, 8 µg of filamentous hemagglutinin (FHA) and 2.5 µg of pertactin.11
AEFI occurrence was investigated based on a list of adverse reactions related to Tdap vaccine use, classified as follows: very common (>10% of vaccinated patients); common (1% to 10% of vaccinated patients); uncommon (0.1% to 1.0% of vaccinated patients); Rare (0.1% to 0.01%); and very rare (under 0.01% of vaccinated patients). This classification was based on the date of onset and the duration of each reaction. The adverse event classification criteria used here as the same as those found on the vaccine package insert.13Figure 2 shows the list of AEFI classified in each category, applied by the researchers based on an adaptation of information found on the package insert and the Adverse Event Following Immunization Manual, published by the Ministry of Health.14
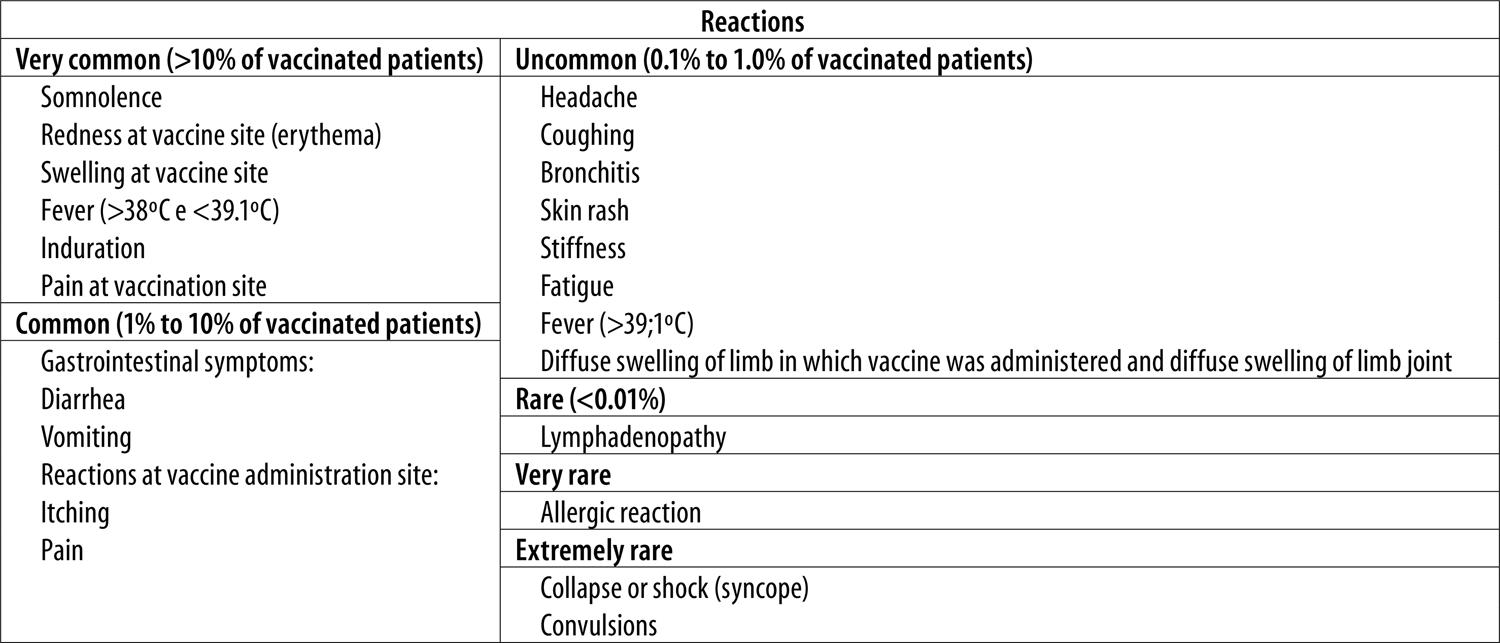
Figure 2 – List of adverse events following immunization with tetanus, diphtheria and acellular pertussis adsorbed vaccine – TdapNote:List adapted from the Adverse Event Following Immunization Epidemiological Surveillance Manual and from the vaccine package insert.
Adverse event frequency was calculated by dividing the number of AEFIs by the number of vaccinated people included in the study (n=201):
In an exploratory manner, the prevalence ratio (PR) was estimated directly, calculated from the product of AEFI prevalence among the parturient women exposed to each of the factors and the corresponding prevalence among non-exposed parturient women. Pearson’s chi-square test was used to test for association between occurrence of AEFI and the following variables: maternal age; gestational age when vaccine was administered; first pregnancy; schooling; race/skin color; monthly family income; and BMI.
The study project was approved by the Adolfo Lutz Institute Research Ethics Committee as per Certification of Submission for Ethical Appraisal (CAAE) No. 37581114.0.0000.0059, issued on 23/03/2017. All respondents were informed about the objective of the study and their participation depended on their agreement and their signing the Free and Informed Consent form.
Results
Two hundred and one out of the 218 parturient women identified were included. Seventeen were excluded from the analysis for the following reasons: vaccination interval <15 days before childbirth (2 parturient women: 11.8%); gestational age <37 weeks (4 parturient women: 23.5%); no record of vaccination date (5 parturient women: 9.4%); birth weight below 2500 grams (3 parturient women: 1.4%); no epidemiological record form (3 parturient women: 17.6%).
Of the 201 women included, 75.1% were between 20 and 34 years old, 53.7% had eight or more years of schooling, 44.8% were of brown race/skin color, 40.3% were at between 30 and 33 gestational weeks when they were vaccinated, and the records of 84.1% showed fewer than seven prenatal consultations ( Table 1 ).
Table 1 – Distribution of women who received Tdapaduring pregnancy and had adverse events following immunization (n=201), by sociodemographic characteristics and data on pregnancy, São Paulo, SP, 2015-2016
| Characteristics | Adverse event | ||||||
|---|---|---|---|---|---|---|---|
|
| |||||||
| Yes | No | Unknown | Total | ||||
|
| |||||||
| n=48 | n=151 | n=2 | n=201 | ||||
|
| |||||||
| n | % | n | % | n | n | % | |
| Mother’s age range (in years) | |||||||
| <20 | 2 | 4.2 | 12 | 7.9 | – | 14 | 7.0 |
| 20-34 | 35 | 72.9 | 114 | 75.5 | 2 | 151 | 75.1 |
| ≥35 | 11 | 22.9 | 25 | 16.6 | – | 36 | 17.9 |
| Mother’s schooling (in years of study) | |||||||
| <8 | 24 | 50.0 | 60 | 39.7 | 1 | 85 | 42.3 |
| ≥8 | 22 | 45.8 | 85 | 56.3 | 1 | 108 | 53.7 |
| Unknown | 2 | 4.2 | 6 | 4.0 | – | 8 | 4.0 |
| Race/skin color | |||||||
| White | 13 | 27.1 | 55 | 36.4 | 2 | 70 | 34.8 |
| Black | 14 | 29.2 | 25 | 16.6 | – | 39 | 19.4 |
| Yellow | – | – | 1 | 0.7 | – | 1 | 0.5 |
| Brown | 20 | 41.7 | 70 | 46.4 | – | 90 | 44.8 |
| Indigenous | 1 | 2.1 | – | – | – | 1 | 0.5 |
| Gestational age at time of vaccination (in weeks) | |||||||
| 17-26 | 3 | 6.3 | 16 | 10.6 | – | 19 | 9.5 |
| 27-29 | 14 | 29.2 | 57 | 37.7 | – | 71 | 35.3 |
| 30-33 | 19 | 39.6 | 61 | 40.4 | 1 | 81 | 40.3 |
| 34-40 | 12 | 25.0 | 16 | 10.6 | 1 | 29 | 14.4 |
| Unknown | – | – | 1 | 0.7 | – | 1 | 0.5 |
| Prenatal consultations | |||||||
| <7 | 40 | 83.3 | 127 | 84.1 | 2 | 169 | 84.1 |
| ≥7 | 8 | 16.7 | 23 | 15.2 | – | 31 | 15.4 |
| Unknown | – | – | 1 | 0.7 | – | 1 | 0.5 |
a) Tetanus, diphtheria and cellular pertussis adsorbed vaccine.
No significant differences were found among the women who had adverse events because of the vaccine with regard to sociodemographic or pregnancy characteristics ( Table 1 ). No association between occurrence of AEFI and the following variables was found: maternal age; gestational age when vaccine was administered; first pregnancy; schooling; race/skin color; and monthly family income. However, statistically significant association was found between AEFI occurrence and higher pre-pregnancy BMI (PR=2.25 – 95%CI 1.06;4.80).
Of the 201 mothers studied, 48 (23.9%) had at least one AEFI with Tdap. A total of 60 AEFI with Tdap related to nine signs and symptoms were identified. The following frequencies were found among AEFI classified as ‘very common’: pain (22.4%), swelling (2.5%), fever (1.5%), somnolence (1.0%) and redness (0.5%). Vomiting was recorded in relation to one participant (0.5%) among AEFI classified as ‘common’; while among AEFI classified as ‘uncommon’, one participant reported headache (0.5%), and another reported local reaction (0.5%) and fatigue (0.5%) ( Table 2 ).
Table 2 – Distribution of clinical manifestations of adverse eventsafollowing exposure to Tdapbvaccine, by length of adverse event duration, São Paulo, SP, 2015-2016
| Adverse event | ≤24 hours | ≤48 hours | ≤72 hours | ≤96 hours | Unknown | Total | Incidence rate: Adverse event x 100/201 % (95%CIb)c | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||||||
| n | % | n | % | n | % | n | % | n | % | n | % | ||
| Pain | 14 | 31.1 | 14 | 31.1 | 8 | 17.8 | 2 | 4.4 | 7 | 15.6 | 45 | 75.0 | 22.4 (17.0;28.5) |
| Somnolence | 2 | 100.0 | – | – | – | – | – | – | – | – | 2 | 3.3 | 1.0 (0.2;3.2) |
| Redness | 1 | 100.0 | – | – | – | – | – | – | – | – | 1 | 1.7 | 0.5 (0.2;2.4) |
| Swelling | 1 | 20.0 | 1 | 20.0 | 1 | 20.0 | – | – | 2 | 40.0 | 5 | 8.3 | 2.5 (0.9;5.4) |
| Fever | 3 | 100.0 | – | – | – | – | – | – | – | – | 3 | 5.0 | 1.5 (0.4;4.0) |
| Vomiting | 1 | 100.0 | – | – | – | – | – | – | – | – | 1 | 1.7 | 0.5 (0.2;2.4) |
| Local reaction | – | – | – | – | – | – | – | – | 1 | 100.0 | 1 | 1.7 | 0.5 (0.2;2.4) |
| Fatigue | – | – | 1 | 100.0 | – | – | – | – | – | – | 1 | 1.7 | 0.5 (0.2;2.4) |
| Headache | – | – | 1 | 100.0 | – | – | – | – | – | – | 1 | 1.7 | 0.5 (0.2;2.4) |
|
| |||||||||||||
| Total | 22 | 36.7 | 17 | 28.3 | 9 | 15.0 | 2 | 3.3 | 10 | 16.7 | 60 | 100.0 | 29.9 (23.8;36.5) |
a) The remaining adverse events under study were not reported by the women who were interviewed.
b) Tetanus, diphtheria and cellular pertussis adsorbed vaccine.
c) 95%CI: 95% confidence interval.
No rare, very rare or extremely rare adverse events were recorded. All 64 events were classified as being expected, as described on the vaccine package insert. No occurrence was classified as severe and the outcome of all occurrences was recovery without sequelae.
A third of the events (36.7%) occurred in up to 24 hours; the remainder occurred within 48 hours (28.3%), 72 hours (15.0%) and 96 hours (3.3%) following vaccination ( Table 2 ).
Discussion
The study found that Tdap vaccine is safe, given that only 24.5% of vaccinated women had some kind of adverse event. In addition, the greater part of the adverse events fell into the very common AEFI category. No unexpected adverse events following vaccination of the pregnant women were found. The adverse events identified were mentioned on the package insert of the product administered. It is noteworthy that no severe adverse events were found.
The findings are similar to those found in other studies.16 Muñoz et al. conducted a randomized double-blind trial with healthy pregnant women aged between 18 and 45 with low risk of obstetric complications. They were recruited in university and private clinics in three United States cities (Houston, Durham and Seattle). Their results show that there was no occurrence of severe adverse events associated with Tdap vaccine among the women or babies under observation; they did report local reactions in 26 pregnant women (78.8% [95%CI 61.1;91.0]) and 12 puerperal women (80.0% [95%CI 51.9;95.7]); systemic symptoms were also reported in 12 pregnant women (36.4% [95%CI 20.4;54.9]) and 11 puerperal women (73.3% [95%CI 44.9;92.2]) (p=0.03). The growth and development of their babies were similar for both groups of mothers. No cases of whooping cough were reported.16
Moro et al. reported on a study of data taken from the U.S. Vaccine Adverse Events Reporting System (VAERS), created in 1990 and jointly administered by the U.S. Centers for Diseases Control and Prevention (CDC) and the Food and Drug Administration (FDA), with the aim of identifying reports of AEFI among pregnant women who were given Tdap after recommendation of this vaccine had been included in the health service routine. The results revealed that in the 45 month period (from October 2011 to June 2015), no new or unexpected warning sign was identified as to the safety of the Tdap vaccine among the pregnant women who received it nor among their babies.17
Maertens et al. conducted a prospective controlled cohort study which monitored on healthy pregnant women and their health babies in five hospitals in the province of Antwerp, Belgium, between 2012 and 2014. The objective of their investigation was to measure (i) the influence of Tdap vaccine booster dose administered during pregnancy and (ii) duration of the presence of maternal antibodies in their infants. Although it was not an initial objective of the study, all the pregnant women were monitored by doctors as to the occurrence of systemic reactions for 30 minutes following vaccine administration. Possible adverse events of the vaccine were monitored during the 30 days following vaccination, especially occurrence of pain at the injection site, swelling, erythema and general symptoms, such as myalgia and fever, among others. The authors concluded that the adverse events identified did not differ from those described on the package insert of the product, and that the majority of the adverse event symptoms were mild and self-limited, progressing to recovery within 72 hours following vaccination.18
Fortner et al. conducted a prospective observational cohort study in two United States cities with 599 women aged 18 to 45 who received Tdap vaccine: 374 pregnant women at between 20 and 33 weeks of pregnancy; and 225 non-pregnant vaccinated women. The results showed that severe local or systemic reactions or fever were rare (≤3% in both groups); and that pain at the vaccine administration site, classified as moderate to intense, was significantly greater in pregnant women (17.9%) when compared to non-pregnant women (11.1%). The frequency of other reactions classified as moderate to severe or severe was not significantly greater among pregnant women when compared to non-pregnant women. The study concluded that Tdap vaccine was tolerated well by both pregnant and non-pregnant women: pregnant women reported moderate to intense pain at the vaccine administration site more frequently than non-pregnant women, but they did not need medical assistance.19
In Brazil, shortly after the National Immunization Program implemented Tdap vaccination for pregnant women, Sancovski et al. conducted a single center retrospective descriptive observational study in São Bernardo do Campo, SP, in which the authors compared (i) data from the medical records of 1203 pregnant women who received Tdap as part of the maternal immunization program and their babies between May 2015 and February 2017 (exposed group), with (ii) data on 1259 unvaccinated women who gave birth between September 2012 and August 2014 (unexposed group). Their findings did not indicate the occurrence of increased risk of pregnancy-related adverse events, nor neonatal adverse events of interest, following maternal vaccination with Tdap. However, the authors stressed that the results of their study should be interpreted with caution in view of limitations – memory bias, difficulties with data collection and recording – inherent to retrospective observational studies.20
Another limitation of the study above that should be pointed out was its sample size, estimated to assess the efficacy of Tdap vaccine among vaccinated mothers and live born babies, which reduces the study’s scope for extrapolations.
An increase in the whooping cough incidence rate occurred in Brazil, followed by a decrease that coincides in time with the introduction of Tdap vaccination for pregnant women. In order to keep whooping cough incidence rates low, it is important to ensure trust in the vaccine’s safety both by health workers and women who use health services. According to Furuta et al., one of the main reasons for low Tdap vaccine coverage rates is concern – also on the part of doctors and pregnant women – about the safety of the vaccine when used during pregnancy. Certain measures are needed, such as disseminating information about vaccine safety and training health professionals to detect, notify and investigate adverse events so as to improve AEFI monitoring.21
These measures can have direct impact on reducing underreporting of adverse events following vaccination, which is a considerable challenge for the National Immunization Program. The results of this study confirm that it is safe for pregnant women to receive tetanus, diphtheria and acellular pertussis adsorbed vaccine (Tdap). The results also increase Tdap acceptance and strengthen the trust of the Brazilian National Health System (SUS) and the trust of SUS service users in the vaccination activities of the National Immunization Program.
REFERENCES
1. Ministério da Saúde (BR) . Secretaria de Vigilância em Saúde . Informe técnico para implantação da vacina adsorvida difteria, tétano e coqueluche (Pertussis Acelular) tipo adulto – dTpa [ Internet ]. Brasília : Ministério da Saúde ; 2014 [ citado 2018 dez 06 ]. Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2015/junho/26/Informe-T--cnico-dTpa-2014.pdf [ Links ]
2. Switzer C , D’Heilly C , Macina D . Immunological and clinical benefits of maternal immunization against pertussis: a systematic review . Infect Dis Ther [ Internet ]. 2019 Dec [ cited 2020 Apr 6 ]; 8 ( 4 ): 499 - 541 . Available from: https://doi.org/10.1007/s40121-019-00264-7 [ Links ]
3. World Health Organization . Pertussis vaccines: WHO position paper – August 2015 . Wkly Epidemiol Rec [ Internet ]. 2015 Aug [ cited 2018 Dec 12 ]; 90 ( 34 ): 433 - 58 . Available from: https://www.who.int/wer/2015/wer9035.pdf?ua=1 [ Links ]
4. Ministério da Saúde (BR) . Calendário nacional de saúde [ Internet ]. Brasília : Ministério da Saúde ; 2019 [ citado 2019 dez 10 ]. Disponível em: http://saude.gov.br/saude-de-a-z/vacinacao/calendario-vacinacao [ Links ]
5. Ministério da Saúde (BR) . Secretaria de Vigilância em Saúde . Departamento de Vigilância das Doenças Transmissíveis . Coqueluche no Brasil: análise da situação epidemiológica de 2010 a 2014 . Bol Epidemiol [ Internet ]. 2015 [ citado 2019 abr 12 ]; 46 ( 39 ): 1 - 8 . Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2018/marco/29/Boletim-epidemiologico-de-2010-a-2014.pdf [ Links ]
6. Fernandes EG , Sartori AMC , Soárez PC , Carvalhanas TRMP , Rodrigues M , Novaes HMD . Challenges of interpreting epidemiologic surveillance pertussis data with changing diagnostic and immunization practices: the case of the state of São Paulo, Brazil . BMC Infect Dis [ Internet ]. 2018 Mar [ cited 2019 Apr 16 ]; 18 ( 1 ): 126 . Available from: https://doi.org/10.1186/s12879-018-3004-1 [ Links ]
7. Donegan K , King B , Bryan P . Safety of pertussis vaccination in pregnant women in UK: observational study . BMJ [ Internet ]. 2014 Jul [ cited 2018 Dec 06 ]; 349 : g4219 . Available from: https:doi.org/10.1136/bmj.g4219 [ Links ]
8. Liang JL , Tiwari T , Moro P , Messonnier NE , Reingold A , Sawyer M , et al . Prevention of pertussis, tetanus, and diphtheria with vaccines in the United States: recommendations of the Advisory Committee on Immunization Practices (ACIP) . MMWR Recomm Rep [ Internet ]. 2018 Apr [ cited 2019 Mar 20 ]; 67 ( 2 ): 1 - 44 . Available from: https://doi.org/10.15585/mmwr.rr6702a1 [ Links ]
9. Ministério da Saúde (BR) . Secretaria de Vigilância em Saúde . Departamento de Vigilância das Doenças Transmissíveis . CGPNI/GT-INTEC/FCP, SFP e RCM. Nota informativa sobre mudanças no calendário nacional de vacinação para o ano de 2017 [ Internet ]. Brasília : Ministério da Saúde ; 2016 [ citado 2018 abr 16 ]. Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2016/dezembro/28/Nota-Informativa-384-Calendario-Nacional-de-Vacinacao-2017.pdf [ Links ]
10. Fernandes EG , Sato APS , Vaz-de-Lima LRA , Rodrigues M , Leite D , Brito CA , et al . The effectiveness of maternal pertussis vaccination in protecting newborn infants in Brazil: A case-control study . Vaccine [ Internet ]. 2019 Aug [ cited 2019 Apr 11 ]. Available from: https://doi.org/10.1016/j.vaccine.2019.03.049 [ Links ]
11. Vaz-de-Lima LRA , Sato HK , Fernandes EG , Sato APS , Pawloski LC , Tondella ML , Maternal Pertussis Vaccine Working Group , et al . Association between the timing of maternal vaccination and newborns’ anti-pertussis toxin antibody levels . Vaccine [ Internet ]. 2019 May [ cited 2020 Apr 6 ]; 37 ( 36 ): 5474 - 80 . Available from: https:doi.org/10.1016/j.vaccine.2019.04.079 [ Links ]
12. Instituto Brasileiro de Geografia e Estatística . Cidade: São Paulo [ Internet ]. Rio de Janeiro : Instituto Brasileiro de Geografia e Estatística ; 2017 [ citado 2020 jan 30 ]. Disponível em: https://cidades.ibge.gov.br/brasil/sp/sao-paulo/panorama [ Links ]
13. Instituto Brasileiro de Geografia e Estatística . Cidades: índice de desenvolvimento humano de São Paulo [ Internet ]. Rio de Janeiro : Instituto Brasileiro de Geografia e Estatística ; 2017 [ citado 2020 jan 30 ]]. Disponível em: https://cidades.ibge.gov.br/brasil/sp/sao-paulo/pesquisa/37/30255 [ Links ]
14. Secretaria de Estado da Saúde do Estado de São Paulo . Instituto Butantan . Vacina adsorvida difteria, tétano e pertussis (acelular)/dTpa [ Internet ]. São Paulo : Instituto Butantan ; [ 2018 ] [ citado 2020 Abr 6 ]. ( Texto de bula para profissionais de saúde. Aprovada pela Anvisa em 26/09/2018 ). Disponível em: http://butantan.gov.br/assets/pdf/soros_vacinas/vacinas/Bula-Vacina-Adsorvida-Difteria-Tetano-e-Pertussis-Acelular-Instituto-Butantan-Paciente-Consulta-Remedios.pdf [ Links ]
15. Ministério da Saúde (BR) . Secretaria de Vigilância em Saúde . Departamento de Vigilância das Doenças Transmissíveis . Manual de vigilância epidemiológica de eventos adversos pós-vacinação [ Internet ]. 3 . ed. Brasília : Ministério da Saúde ; 2014 [ citado 2020 abr 6 ]. 250 p. Disponível em: https://bvsms.saude.gov.br/bvs/publicacoes/manual_vigilancia_epidemiologica_eventos_adversos_pos_vacinacao.pdf [ Links ]
16. Muñoz FM , Bond NH , Maccato M , Pinell P , Hammill HA , Swamy GK , et al . Safety and immunogenicity of tetanus diphtheria and acellular pertussis (Tdap) immunization during pregnancy in mothers and infants: a randomized clinical trial . JAMA [ Internet ]. 2014 May [ cited 2018 Dec 06 ]; 311 ( 17 ): 1760 - 9 . Available from: https://doi.org/10.1001/jama.2014.3633 [ Links ]
17. Moro PL , Perez-Vilar S , Lewis P , Bryant-Genevier M , Kamiya H , Cano M . Safety surveillance of diphtheria and tetanus toxoids and acellular pertussis (DTaP) vaccines . Pediatrics [ Internet ]. 2018 Jul [ cited 2018 Jun 08 ]; 142 ( 1 ): e20174171 . Available from: https://doi.org/10.1542/peds.2017-4171 [ Links ]
18. Maertens K , Caboré RN , Huygen K , Hens N , Van Damme P , Leuridan E . Pertussis vaccination during pregnancy in Belgium: results of a prospective controlled cohort study . Vaccine [ Internet ]. 2016 Jan [ cited 2019 Mar 19 ]; 34 ( 1 ): 142 - 50 . Available from: https://doi.org/10.1016/j.vaccine.2015.10.100 [ Links ]
19. Fortner KB , Swamy GK , Broder KR , Jimenez-Truque N , Zhu Y , Moro PL , et al . Reactogenicity and immunogenicity of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine (Tdap) in pregnant and nonpregnant women . Vaccine [ Internet ]. 2018 Oct [ cited 2018 Dec 06 ]; 36 ( 42 ): 6354 - 6360 . Available from: https://doi.org/10.1016/j.vaccine.2018.07.012 [ Links ]
20. Sancovski M , Mesaros N , Feng Y , Ceregido MA , Luyts D , Barros E . Safety of reduced antigen content diphtheria-tetanus-acellular pertussis vaccine when administered during pregnancy as part of the maternal immunization program in Brazil: a single center, observational, retrospective, cohort study . Hum Vaccin Immunother [ Internet ]. 2019 Jun [ cited 2020 Apr 6 ]; 15 ( 12 ): 2873 - 81 . Available from: https://doi.org/10.1080/21645515.2019.1627161 [ Links ]
21. Furuta M , Sin J , Ng ESW , Wang K . Efficacy and safety of pertussis vaccination for pregnant women - a systematic review of randomised controlled trials and observational studies . BMC Pregnancy Childbirth [ Internet ]. 2017 Nov [ cited 2020 Apr 6 ]; 17 ( 1 ): 390 . Available from: https://doi.org/10.1186/s12884-017-1559-2 [ Links ]
Associate Editor: Doroteia Aparecida Höfelmann – orcid.org/0000-0003-1046-3319
*This study received support from the following research institutions and projects: Special Health Fund for Mass Immunization and Disease Control (FESIMA); Adolfo Lutz Institute (IAL)/São Paulo State Health Department Disease Control Coordination Body (CCD/SES); São Paulo State Research Support Fund (FAPESP), Process No. 16157-1/2015); and Latin American Pertussis Project (LAPP). The researchers also received support via a cooperation project between the United States Centers for Disease Control and Prevention (CDC), Pan American Health Organization (PAHO-WHO), Sabin Vaccine Institute and Health Ministries of the countries chosen to strengthen whooping cough surveillance in Latin America.
Received: October 07, 2019; Accepted: March 24, 2020











 texto em
texto em 


