Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Epidemiologia e Serviços de Saúde
versión impresa ISSN 1679-4974versión On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.29 no.3 Brasília jun. 2020 Epub 12-Mayo-2020
http://dx.doi.org/10.5123/s1679-49742020000300003
Original article
Temporal trends and spatial distribution of leprosy-related mortality in the state of Tocantins, Brazil, 2000-2015
1Fundação Escola de Saúde Pública de Palmas, Palmas, TO, Brazil
2Instituto Federal de Ciência, Educação e Tecnologia do Ceará, Caucaia, CE, Brazil
Objective
to describe temporal trends and spatial distribution patterns of leprosy-related deaths in the state of Tocantins, Brazil, 2000-2015.
Methods
this study was based on Mortality Information System (SIM) data; mortality coefficient temporal trend analysis was performed using joinpoint regression and descriptive spatial analysis, taking the state’s 139 municipalities as geographic units of analysis.
Results
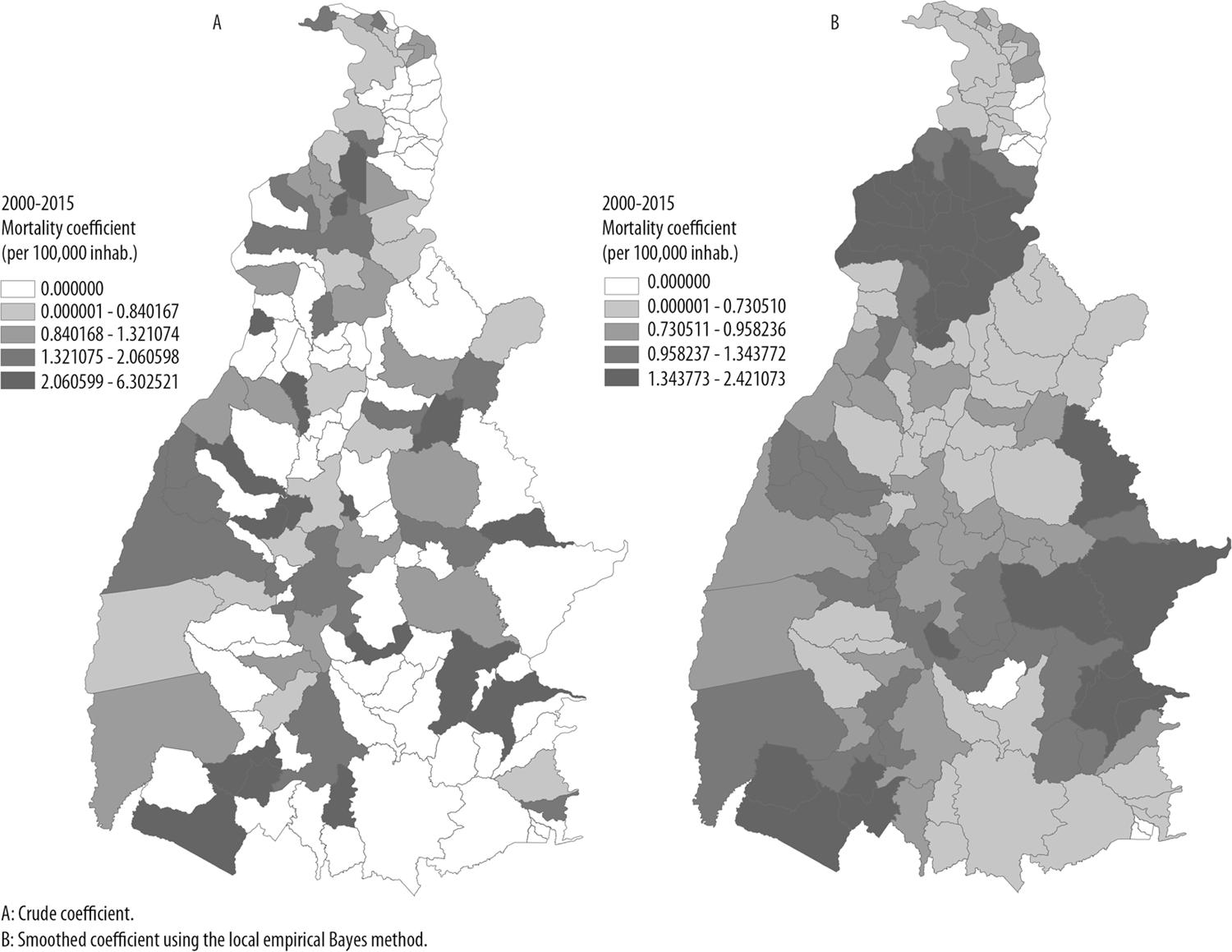
201 leprosy-related deaths were identified in Tocantins; average mortality coefficient was 0.7 deaths/100,000 inhabitants; mortality tended to increase significantly (5.4% Annual Percentage Change - 95% CI 2.0; 8.9); the average annual crude mortality coefficient ranged from 0.00 to 6.83 deaths/100,000 inhabitants, while the smoothed indicator ranged from 0.00 to 2.42 deaths/100,000 inhabitants.
Conclusion
leprosy mortality showed a rising trend and heterogeneous spatial distribution in Tocantins, with the need to strengthen leprosy surveillance and control measures.
Key words: Epidemiology; Leprosy; Spatial Analyses; Time Series Studies; Mortality
Introduction
Leprosy is caused by a bacterium known as Mycobacterium leprae and continues to be a neglected disease and a Public Health Problem in Brazil.1In 2017, there were around 211,000 new leprosy cases globally, 13.8% of which in the Americas; Brazil accounted for 92.3% of cases on the continent,3 with an overall detection coefficient of 12.9 new cases per 100,000 inhabitants.4 Several states in Brazil’s North and Midwest regions have heterogeneous and hyperendemic leprosy detection coefficients.4 The state of Tocantins has high leprosy incidence and the majority of its municipalities are considered to be hyperendemic.1
Between 2000 and 2011, leprosy was responsible for 7,732 deaths in Brazil, with Tocantins accounting for a considerable specific leprosy mortality burden. In Brazil as a whole, the mortality burden was greater among males, the elderly and black-skinned people. As to its spatial distribution, the mortality burden was greater in the endemic regions of the country’s North and Northeast regions.2 The fact of it being a chronic disease, with the potential for complications and adverse effects caused by prolonged medication, can lead to death, principally among neglected populations, although the disease’s lethality is considered to be low.2 In hyperendemic areas, lack of monitoring of leprosy mortality indicators points to negligence on the part of health services, especially epidemiological surveillance services, as well as researchers.2 Leprosy mortality is still a little studied subject. In particular, there are no studies aimed at gaining a better understanding of leprosy mortality in the state of Tocantins, despite it being a hyperendemic area. With the aim of filling this knowledge gap, the objective of this article was to analyze temporal trends and spatial distribution patterns of leprosy-related deaths in the state of Tocantins, between 2000 and 2015.
Methods
Descriptive time series analysis was performed based on data retrieved from the Health Ministry’s Mortality Information System (SIM). All deaths that occurred in the state of Tocantins between 2000 and 2015 were included when leprosy had been recorded on death certificates as the underlying cause or cause related to death (multiple causes of death).10 Leprosy-related causes of death were detected in accordance with the 10th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10): A30 Leprosy [Hansen’s disease]; A30.0 Indeterminate leprosy; A30.1 Tuberculoid leprosy; A30.2 Borderline tuberculoid leprosy; A30.3 Borderline leprosy; A30.4 Borderline lepromatous leprosy; A30.5 Lepromatous leprosy; A30.8 Other forms of leprosy; A30.9 Leprosy, unspecified; and B92 Sequelae of leprosy.11
Founded and integrated into Brazil’s Northern region in 1988, Tocantins was the country’s last state to be created. It is part of the Brazilian Amazon region and savanna is the predominant form of flora. Its territory of 277,622km2 is home to a population estimated as being 1,532,902 inhabitants in 2017. Comprised of 139 municipalities, Tocantins is divided into eight health regions created by the Bipartite Inter Health Managers Commission in 2012: namely, Amor Perfeito, Bico do Papagaio, Capim Dourado, Cantão, Ilha do Bananal, Cerrado Tocantins Araguaia, Sudeste and Médio Norte Araguaia.12 This division took into consideration the populations and demographic densities of the municipalities, with the aim of providing local essential actions and services.12 The rate of urbanization in Tocantins doubled between 1980 and 2020, rising from 40% to 80%. In the last twenty years there has been noticeable progress with the living conditions of the state’s population as demonstrated by its sociodemographic data.13
The data on mortality were retrieved from SIM. The source of these data are Death Certificates (DC) and they are public domain data available on the website of the Brazilian National Health System Information Technology Department (DATASUS).14 DCs are filled in based on a standardized document and this must be done by a doctor. The data were downloaded from SIM and processed, whereby deaths occurring in the state of Tocantins between 2000 and 2015 were selected. The information was checked for completeness and codes with different fields were standardized. Death records can have several causes on the same line and other lines had not been filled in. Because of this, new variables were generated for cause of death when more than one cause had been recorded on the same DC line. In this way we identified all deaths having leprosy as the underlying cause – or associated cause – of death notified on SIM.15
The demographic information originated from the Brazilian Institute of Geography and Statistics (IBGE) in relation to the 2000 and 2010 demographic censuses, while the population estimates for each year from 2001 to 2009 and from 2011 to 2015 were retrieved from the DATASUS website.14
The variables available on SIM were described as follows:
sex (male; female);
age range (in years anos: <15; 15-29; 30-39; 40-49; 50-59; 60-69; ≥70);
race/skin color (yellow; white; indigenous; brown; black);
marital status (married; separated/divorced; single; widowed; other; unknown);
schooling (in years: none; 1-3; 4-7; 8-11; ≥12);
place of death (at home; hospital; public thoroughfare; other health establishments; others); and
health region of residence (place/health region);
causes of death (underlying and associated).
A ‘municipality size’ variable was created derived from the ‘health region of residence’ variable, categorized into: small I (<20,000 inhabitants); small II (20,000 - 49,000 inhab.); medium (50,000 - 99,000 inhab.); and large (over 100,000 inhab.). The municipalities were aggregated in the respective health regions created by the Bipartite Inter Health Managers Commission.12
The leprosy mortality coefficient was calculated by dividing the population of Tocantins by the number of leprosy deaths and multiplying the result by 100,000 for each year.
The temporal trends of the leprosy mortality coefficients for the study period were assessed using Joinpoint Trend Analysis Software version 4.1.0 (US National Cancer Institute, Bethesda, MD, USA). The state’s health regions formed the geographic units of analysis. This joinpoint regression statistical analysis technique was used with the aim of identifying, by means of a linear logarithmic scale, significant change in linear trend over the period 2000 to 2015.
The independent variable was the year of occurrence and the leprosy mortality indicators were the dependent variables. Years in which there were zero (0) death records per health region were replaced with the number 0.5 in order for it to be possible to perform the analysis in the linear regression model. Using the Poisson regression model with joinpoint it was possible, using a logarithmic scale, to model a sequence of trend lines and pinpoint their joinpoints. The Monte Carlo permutation method was used to test for significance.
Starting with the minimum number of joinpoints, where 0 joinpoint is a single straight line, the analysis progressed assessing one or more joinpoints to test the statistical significance of their inclusion in the model, until up to 1 (one) joinpoint was reached in this test. Joinpoints relating to slope changes with statistical significance were kept in the final model.
Annual percent change (APC) was calculated per period based on the regression line using a natural logarithmic scale of the mortality indicators, with their respective confidence intervals. In order to estimate APC, the weighted geometric average of the APCs was applied, with weighting equivalent to the length of each straight line stretch in the temporal hiatus.17 When a rising indicator trend with a minimum confidence interval value greater than zero was found, the increase was considered to be significant. Alternately, if a falling trend was found with a minimum confidence interval value less than zero, the trend was considered to be falling. In cases in which zero was inside the confidence interval, the indicator was considered to be stable.
The patterns of spatial distribution of leprosy-related mortality were then analyzed, taking the municipalities of residence of the deaths (n=139) as the geographic units of the study. The average leprosy mortality coefficients (per 100,000 inhab.) were calculated for the 16 year period selected by the researchers. The smoothed mortality coefficients (per 100,000 inhab.) were obtained using the local empirical Bayes method with the aim minimizing fluctuations due to unknown causes and ensuring better mortality coefficient balance in municipalities with smaller populations and uncommon cases.19 Deaths with records of residence in municipalities in other states or with unknown municipalities of residence were excluded.
TerraView version 4.1 (Instituto Nacional de Pesquisas Espaciais - INPE, São José dos Campos, SP, Brazil) and ArcGIS version 9.3 (Environmental Systems Research Institute - ESRI, Redlands, CA, United States) were used to process and organize the data, calculate the crude and smoothed indicators and build the thematic maps.
Given that the study was based on public domain secondary data on mortality available for access at the DATASUS website, the database of which is anonymous and does not enable people to be identified, the study project did not need to be submitted for the appraisal of a Research Ethics Committee.
Results
Between 2000 and 2015, there were 93,021 deaths in the state of Tocantins. The average leprosy-related mortality coefficient was 0.7 deaths/100,000 inhab., whereby leprosy was identified in 201 deaths (0.2%), with 128 (63,7%) as associated cause of death and 73 (36.3%) as underlying cause of death (Table 1).
Table 1 – Characteristics of leprosy-related deaths (n=201) in the state of Tocantins, 2000-2015
| Variables | N | % |
|---|---|---|
| Clinical form/underlying cause (n=73) | ||
|
| ||
| Leprosy, unspecified (A30.9) | 41 | 20.4 |
| Sequelae of leprosy (B92) | 17 | 8.5 |
| Lepromatous leprosy (A30.5) | 5 | 2.5 |
| Indeterminate leprosy (A30.0) | 4 | 2.0 |
| Tuberculoid leprosy (A30.1) | 1 | 0.5 |
| Other forms of leprosy (A30.8) | 2 | 1.0 |
| Borderline leprosy (A30.3) | 3 | 1.5 |
|
| ||
| Sex | ||
|
| ||
| Male | 143 | 71.1 |
| Female | 58 | 28.9 |
|
| ||
| Age range (in years) | ||
|
| ||
| <15 | 2 | 1.0 |
| 15-29 | 10 | 5.0 |
| 30-39 | 16 | 8.0 |
| 40-49 | 26 | 12.9 |
| 50-59 | 35 | 17.4 |
| 60-69 | 29 | 14.4 |
| ≥70 | 83 | 41.3 |
|
| ||
| Race/skin color | ||
|
| ||
| White | 38 | 18.9 |
| Black | 30 | 14.9 |
| Yellow | 1 | 0.5 |
| Brown | 124 | 61.7 |
| Indigenous | 8 | 4.0 |
|
| ||
| Marital status | ||
|
| ||
| Single | 81 | 40.3 |
| Married | 71 | 35.3 |
| Widowed | 27 | 13.4 |
| Separated/divorced | 13 | 6.5 |
| Other | 5 | 2.5 |
| Unknown | 4 | 2.0 |
|
| ||
| Schooling (in completed years of study) | ||
|
| ||
| None | 84 | 41.7 |
| 1-3 | 56 | 27.9 |
| 4-7 | 27 | 13.4 |
| 8-11 | 13 | 6.5 |
| ≥12 | 2 | 1.0 |
| Unknown | 19 | 9.5 |
|
| ||
| Municipality size | ||
|
| ||
| Small I | 98 | 48.7 |
| Small II | 17 | 8.5 |
| Medium | 20 | 10.0 |
| Large | 66 | 32.8 |
|
| ||
| Place of occurrence | ||
|
| ||
| Hospital | 136 | 67.6 |
| Other health establishments | 1 | 0.5 |
| At home | 58 | 28.9 |
| Public thoroughfare | 1 | 0.5 |
| Other | 5 | 2.5 |
|
| ||
| Health region of residence | ||
|
| ||
| Capim Dourado | 35 | 17.4 |
| Sudeste | 12 | 6.0 |
| Ilha do Bananal | 28 | 13.9 |
| Cerrado Tocantins Araguaia | 25 | 12.4 |
| Médio Norte Araguaia | 51 | 25.3 |
| Amor Perfeito | 19 | 9.5 |
| Cantão | 16 | 8.0 |
| Bico do Papagaio | 15 | 7.5 |
A higher proportion of deaths was recorded in 2010 and 2013, being 10.0% in both years. The youngest leprosy death was 9 years old and the oldest was 98 years old. Average age at death was 61 years. Average death coefficients by age range were 0.03/100,000 inhab. in the <15 years age range, 0.17 between 15 and 29, 0.50 between 30 and 39, 1.17 between 40 and 49, 2.51 between 50 and 59 and 3.27 in those aged >70, with the latter being the highest. Deaths in those aged over 70 (41.3%) stood out, as did deaths in the 50-59 age range (17.4%). The majority of deaths were male (71.1%), with an average value of 1.28 deaths per 100,000 inhabitants over the entire period analyzed from 2000 to 2015. Deaths were more prevalent among people of brown race/skin color (61.7%), among those recorded as being single (40.3%) and among those who had never attended school (41.7%). 67.6% of deaths happened in hospital. Médio Norte Araguaia was the region of residence in which deaths were most predominant (25.3%) (Table 1).
The temporal trend analysis demonstrated that in the period 2000-2015 the crude leprosy-related mortality coefficient rose significantly for the state as a whole (APC=5.4 – 95%CI 2,0;8,9), as well as in the Sudeste (APC=9.7 – 95%CI 3.1;16.8) and Capim Dourado (APC=10.1 – 95%CI 4.7;15.8) health regions. The indicator was stable for the remaining health regions (Table 2).
Table 2 – Temporal trend of crude mortality coefficients (per 100,000 inhab.) related to Leprosy according to joinpoint regression in the state of Tocantins and its health regions, 2000-2015
| Health regions (state of Tocantins) | APCa | 95%CIb |
|---|---|---|
| Tocantins | 5.4c | 2.0;8.9 |
|
| ||
| Cantão | -2.7 | -9.5;4.5 |
| Bico do Papagaio | 1.3 | -6.4;9.7 |
| Médio Norte Araguaia | -1.6 | -9.5;6.9 |
| Amor Perfeito | 7.7 | -3.1;19.7 |
| Cerrado Tocantins Araguaia | 4.1 | -3.2;11.9 |
| Ilha do Bananal | 5.9 | -0.4;12.6 |
| Sudeste | 9.7c | 3.1;16.8 |
| Capim Dourado | 10.1c | 4.7;15.8 |
a) APC: annual percentage change.
b) IC95%: intervalo de confiança de 95%.
c) Significativamente diferente de 0 (p<0,05).
In the period 2000-2015, 48.9% of municipalities in the state of Tocantins registered at least one leprosy-related death (Figure 1A). Corrected indicators with less fluctuation were achieved using the local empirical Bayes method. The crude average annual mortality coefficient varied between 0.00 and 6.30 deaths/100,000 inhab., while the smoothed indicator was 0.00 to 2.42 death/100,000 inhab. (Figures 1A and 1B). 30.9% of the municipalities were found to have high leprosy-related mortality coefficients in all the state’s health regions, with higher concentration in the Norte Araguaia and Médio Araguaia health regions.
Discussion
This study has enabled a comprehensive understanding of the epidemiological patterns of leprosy mortality in the state of Tocantins. The results reveal a scenario of relevant occurrence of mortality due to leprosy, a neglected disease considered to be of low lethality. The study found greater magnitude of deaths among males, as well as among people in older age groups, people of brown race/skin color and those with no or little schooling. The Sudeste and Capim Dourado health regions had significant increase in cases of death due to leprosy. Spatial and temporal identification of risk areas enabled municipalities needing priority actions to be identified among health regions where mortality was high.
Low lethality, long latency period, attention given to underlying cause of death and underrecording can contribute to the disease being dealt with as an acute condition and favor increase in deaths attributed to it, given the lack of leprosy being managed as a chronic condition.9 In the studies we consulted, systemic changes in the body are most related to death, possibly because of drug reactions, adverse effects of drugs and higher level of infiltration owing to high bacillary load, apart from other infections.6It should be emphasized that the lethal potential of the disease is associated with complications arising from episodes of drug reactions, and can be related to kidney failure and sepsis.21
It is a fact that high leprosy mortality among elderly people in Tocantins relates to the chronic aspects of the disease, a possible relationship with lower response capacity to polychemotherapy and greater vulnerability of this age group, associated with the severity of the disease and preexisting comorbidities.9 Attention is drawn to the magnitude of years of potential life lost due to death from leprosy: almost half the deaths occurred in people of economically active age, indicating an important social and economic impact.22 Despite most deaths occurring in older age groups, child deaths were also recorded. Leprosy related deaths among children are not expected, in view of the pattern of the disease’s development and chronic nature. These findings point to a relationship with the pattern of hyperendemicity of this age range in the state of Tocantins and, consequently, active transmission of the disease and weakness of control programs with regard to early detection of bacilliferous sources. It is a relationship that also involves adult migrants and men in particular.1
Vulnerability to becoming ill predominant among the male population3 was not only expected but also found in this study. The reasons explaining this phenomenon are linked to (i) cultural issues, (ii) greater exposure to risk environments, (iii) low use of medical services and (iv) health service shortcomings in adopting specific strategies to reach this population.25 What was already known and was confirmed by this analysis is that the greater proportion of cases of advanced forms of the disease occurs in the male population, favoring severe complications and greater likelihood of mortality.2
Another aspect that marked vulnerability to death from leprosy was its predominance in people of brown race/skin color. This finding may be due to the relationship between social inequality and living and health conditions in this group, corroborating results of other studies.2 The fact that almost 70% of those who died had less than three years of schooling is evidence and reaffirms the neglected nature of the disease among populations in situations of social vulnerability.8 A higher proportion of deaths was also found in small towns with less than 20,000 inhabitants. Similarly, a recent study also conducted in Tocantins found that lack of survival conditions was a decisive factor for leprosy transmission and occurrence, especially in municipalities with less than 50,000 inhabitants.24 On the other hand, it cannot be ruled out that the higher proportion of leprosy deaths recorded in the Amor Perfeito and Médio Norte Araguaia health regions may have been influenced by better diagnosis and clinical management capacity.
With regard to temporal trend, a pattern of significant increase in the leprosy mortality coefficient was found over the 16 year period, both for the state as a whole and for the Sudeste and Capim Dourado health regions, while this coefficient was stable in the remaining health regions. Coincidentally, the indicators of significant increase in deaths found for the state as a whole were also stable with regard to the detection coefficient for grade 2 physical disability and also with regard to the detection coefficient for people under 15 years old, similarly to results of previous time series studies conducted in Tocantins.27
The crude spatial analysis indicators found heterogeneity among the high leprosy mortality coefficients in the state’s municipalities. The clusters found by smoothing the leprosy death indicators corroborate the leprosy incidence coefficient clusters found by recent studies.1 An interesting finding was the presence of municipalities with high and low smoothed coefficients within the same health region, even though they had the same environmental and sociodemographic characteristics in common, and even though they had the same access to regionalized health services. This finding may be explained by better or worse health service operational capacity at each location.13 Spatial and temporal identification of risk or hyperendemic areas enabled identification of priority municipalities in health regions with high mortality rates. This can serve to scientifically inform the organization and planning of actions to improve health care.
This study may have limitations due to its analyzing secondary databases retrieved from the SIM health information system derived from Death Certificate information. Data was collected on multiple causes of death where mention was made of leprosy in any field, rather than only leprosy as the underlying cause, in order to ensure a more reliable estimate of the true severity of leprosy-related mortality.
Spatial trend and distribution of leprosy mortality was heterogeneous among the state’s different regions, and they enabled selection of priority municipalities for care in relation to infection. Leprosy is a neglected cause of death, given that control measures can be strengthened and treatment can be provided. The state needs to make efforts to achieve acceptable indicators. This is possible through the implementation of sustainable control measures, with appropriate case management and systematic monitoring of leprosy-related complications, such as severe reactions and adverse effects of polychemotherapy.
REFERENCES
1. Monteiro LD, Martins-Melo FR, Brito AL, Alencar CH, Heukelbach J. Padrões espaciais da hanseníase em um estado hiperendêmico no Norte do Brasil, 2001-2012. Rev Saúde Pública [Internet]. 2015 [citado 2020 fev 28];49:84. Disponível em: https://doi.org/10.1590/S0034-8910.2015049005866 [ Links ]
2. Martins-Melo FR, Assunção Ramos AV, Ramos Junior AN, Alencar CH, Montenegro Junior RM, Heukelbach J. Leprosy-related mortality in Brazil: a neglected condition of a neglected disease. Trans R Soc Trop Med Hyg [Internet]. 2015 Sep [cited 2020 Feb 18];109(1):643-52. Available from: https://doi.org/10.1093/trstmh/trv069 [ Links ]
3. World Health Organization. Global leprosy update, 2017: reducing the disease burden due to leprosy. Wkly Epidemiol Rec [Internet]. 2018 Aug [cited 2020 Feb 28];93(35):445-56. Available from: https://apps.who.int/iris/handle/10665/274290 [ Links ]
4. Ministério da Saúde (BR). DATASUS. Hanseníase – indicadores operacionais e epidemiológicos [Internet]. Brasília: Ministério da Saúde; 201- [citado 2019 abr 2]. Disponível em: http://tabnet.datasus.gov.br/cgi/sinannetbd/hanseniase/hans_indicadores.htm [ Links ]
5. Alencar CH, Ramos Junior AN, Santos ES, Richter J, Heukelbach J. Clusters of leprosy transmission and of late diagnosis in a highly endemic area in Brazil: focus on different spatial analysis approaches. Trop Med Int Health [Internet]. 2012 Apr [cited 2020 Feb 28];17(4):518-25. Available from: https://doi.org/10.1111/j.1365-3156.2011.02945.x [ Links ]
6. Lombardi C. Aspectos epidemiológicos da mortalidade entre doentes de hanseníase no estado de São Paulo. Rev Saúde Pública [Internet]. 1984 abr [citado 2020 fev 28];18(2):71-107. Disponível em: https://doi.org/10.1590/S0034-89101984000200003 [ Links ]
7. Opromolla DVA (ed.). Noções de hansenologia. Bauru: Centro de Estudos Dr. Reynaldo Quagliato; 2000. [ Links ]
8. Rocha MCN, Lima RB, Stevens A, Gutierrez MMU, Garcia LP. Óbitos registrados com causa básica hanseníase no Brasil: uso do relacionamento de bases de dados para melhoria da informação. Ciênc Saúde Coletiva [Internet]. 2015 abr [citado 2020 fev 28];20(4):1017-26. Disponível em: https://doi.org/10.1590/1413-81232015204.20392014 [ Links ]
9. Ramos ARS, Ferreira SMB, Ignotti E. Óbitos por hanseníase como causa básica em residentes no estado de Mato Grosso, Brasil, no período de 2000 a 2007. Epidemiol Serv Saúde [Internet]. 2013 abr-jun [citado 2020 fev 28];22(2):273-84. Disponível em: http://dx.doi.org/10.5123/S1679-49742013000200009 [ Links ]
10. Australian Institute of Health and Welfare. Multiple causes of death in Australia: an analysis of all natural and selected chronic disease causes of death 1997–2007 [Internet]. Canberra: AIHW; 2012 [cited 2018 Jan 17]. Available from: http://www.aihw.gov.au/publication-detail/?id=10737422603 [ Links ]
11. World Health Organization. Statistical classification of diseases and related health problems (ICD): 10th revision [Internet]. Geneva: World Health Organization; 2010 [cited 2018 Jan 17]. Available from: http://apps.who.int/classifications/apps/icd/icd10online [ Links ]
12. Governo do Estado do Tocantins. Secretaria de Estado de Saúde. Comissão Intergestores Bipartite. Resolução CIB n 161/2012 de 29 de agosto de 2012. Dispõe sobre a conformação das novas regiões de saúde do Estado do Tocantins e as ações e serviços mínimos a serem ofertados nesses territórios [Internet]. Palmas: Governo do Estado do Tocantins; 2012 [citado 2020 fev 28]. Disponível em: https://central3.to.gov.br/arquivo/244723/ [ Links ]
13. Instituto Brasileiro de Geografia e Estatística. Estados@: Tocantins [Internet]. Rio de Janeiro: Instituto Brasileiro de Geografia e Estatística; 2015 [citado 2017 ago 28]. Disponível em: http://ibge.gov.br/estadosat/perfil.php?sigla=to [ Links ]
14. Ministério da Saúde (BR). Departamento de Informática do Sistema Único de Saúde. Sistema de Informação sobre Mortalidade (SIM) [Internet]. Brasília: Ministério da Saúde; 2017 [citado 2017 nov 10]. Disponível em: http://tabnet.datasus.gov.br/cgi/sim/dados/cid10_indice.htm [ Links ]
15. Martins-Melo FR, Alencar CH, Ramos Junior AN, Heukelbach J. Epidemiology of mortality related to Chagas’ disease in Brazil, 1999-2007. PLoS Negl Trop Dis [Internet]. 2012 Feb [cited 2020 Feb 28];6(2):e1508. Available from: http://doi.org/10.1371/journal.pntd.0001508 [ Links ]
16. Martins-Melo FR, Ramos Junior AN, Alencar CH, Lange W, Heukelbach J. Mortality of Chagas’ disease in Brazil: spatial patterns and definition of high-risk areas. Trop Med Int Health [Internet]. 2012 Sep [cited 2020 Feb 28];17(9):1066-75. Available from: https://doi.org/10.1111/j.1365-3156.2012.03043.x [ Links ]
17. Kim HJ, Fay MP, Feuer EJ, Midthune DN. Permutation tests for joinpoint regression with application to cancer rates. Stat Med. 2000 Feb;19(3);335-51. [ Links ]
18. Clegg LX, Hankey BF, Tiwari R, Feuer EJ, Edwards BK. Estimating average annual per cent change in the in-trend analysis. Stat Med [Internet]. 2009 Dec [cited 2020 Feb 28];28(29):3670-82. Available from: https://doi.org/10.1002/sim.3733 [ Links ]
19. Assunção RM, Barreto SM, Guerra HL, Sakurai E. Maps of epidemiological rates: a Bayesian approach. Cad Saúde Pública [Internet]. 1998 out [citado 2020 fev 28];14(4):713-23. Disponível em: https://doi.org/10.1590/S0102-311X1998000400013 [ Links ]
20. Ramos AVA. Mortalidade relacionada à hanseníase e sua associação com Diabetes mellitus e hipertensão arterial sistêmica na população brasileira de 1999 a 2007 [dissertação]. Mestrado em Saúde Pública. Fortaleza: Universidade Federal do Ceará; 2011. Disponível em: http://www.repositorio.ufc.br/handle/riufc/6510 [ Links ]
21. Fleury RN, Araújo MG. Manifestações sistêmicas. In: Dermatologia tropical: hanseníase. 4.ed. Manaus: Tropical; 2006. p. 95-100. [ Links ]
22. Ramos ACV, Yamamura M, Arroyo LH, Popolin MP, Chiaravalloti Neto F, Palha PF, et al. Spatial clustering and local risk of leprosy in São Paulo, Brazil. PLoS Negl Trop Dis [Internet]. 2017 Feb [cited 2020 Feb 28];11(2):e0005381. Available from: https://doi.org/10.1371/journal.pntd.0005381 [ Links ]
23. Freitas BHBM, Xavier DR, Cortela DCB, Ferreira SMB. Tendência da hanseníase em menores de 15 anos em Mato Grosso (Brasil), 2001-2013. Rev Saúde Pública [Internet]. 2017 ago [citado 2020 fev 28];51:28. Disponível em: http://doi.org/10.1590/S1518-8787.2017051006884 [ Links ]
24. Monteiro LD, Mota RMS, Martins-Melo FR, Alencar CH, Heukelbach J. Determinantes sociais da hanseníase em um estado hiperendêmico da região Norte do Brasil. Rev Saúde Pública [Internet]. 2017 [citado 2020 fev 28];51:70. Disponível em: 1https://doi.org/10.1590/S1518-8787.2017051006655 [ Links ]
25. Melão S, Blanco LFO, Mounzer N, Veronezi CCD, Simoes PWTA. Perfil epidemiológico dos pacientes com hanseníase no extremo sul de Santa Catarina, no período de 2001 a 2007. Rev Soc Bras Med Trop [Internet]. 2011 fev [citado 2020 fev 28];44(1):79-84. Disponível em: https://doi.org/10.1590/S0037-86822011000100018 [ Links ]
26. Teixeira MAG, Silveira VM, França ERB. Características epidemiológicas e clínicas das reações hansênicas em indivíduos paucibacilares e multibacilares, atendidos em dois centros de referência para hanseníase, na Cidade de Recife, estado de Pernambuco. Rev Soc Bras Med Trop [Internet]. 2010 jun [citado 2020 fev 28];43(3):287-92. Disponível em: https://doi.org/10.1590/S0037-86822010000300015 [ Links ]
27. Monteiro LD, Martins-Melo FR, Brito AL, Lima MS, Alencar CH, Heukelbach J. Tendências da hanseníase no Tocantins, um estado hiperendêmico do Norte do Brasil, 2001-2012. Cad Saúde Pública [Internet]. 2015 maio [citado 2020 fev 28];31(5):971-80. Disponível em: https://doi.org/10.1590/0102-311X00075314 [ Links ]
Associate editor: Suele Manjourany Duro - orcid.org/0000-0001-5730-0811
Received: September 25, 2018; Accepted: February 09, 2020










 texto en
texto en