Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Epidemiologia e Serviços de Saúde
versión impresa ISSN 1679-4974versión On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.29 no.3 Brasília jun. 2020 Epub 08-Jun-2020
http://dx.doi.org/10.5123/s1679-49742020000300016
Original article
Characterization of dengue cases confirmed using the database linkage technique: assessment of virus circulation in Belo Horizonte, Brazil, 2009-2014*
1Universidade Federal de Minas Gerais, Programa de Pós-Graduação em Ciências da Saúde, Belo Horizonte, MG, Brazil
2Fundação Hospitalar do Estado de Minas Gerais, Hospital João XXIII, Belo Horizonte, MG, Brazil
3Secretaria Municipal de Saúde de Belo Horizonte, Gerência de Zoonoses Norte, Belo Horizonte, MG, Brazil
4Secretaria de Estado de Saúde de Minas Gerais, Superintendência de Vigilância Epidemiológica, Belo Horizonte, MG, Brazil
5Universidade Federal de Minas Gerais, Departamento de Parasitologia, Belo Horizonte, MG, Brazil
Methods
this is a series study of cases of dengue infected by different virus serotypes, identified by virus isolation or RT-PCR; database linkage was performed between the Notifiable Health Conditions Information System (SINAN), the Brazilian National Health System Hospital Information System (SIH/SUS) and the Laboratory Environment Manager (GAL).
Results
91.1% of the records were linked (n=775); among the cases (n=851), 60.4% (n=514) were confirmed as DENV-1, 22.1% (n=188) DENV-4, 9.8% (n=83) as DENV-2, and 7.7% (n=66) as DENV-3; DENV-2 accounted for a higher percentage of severe cases (4.5%).
Key words: Dengue; Dengue Virus; Epidemiological Monitoring; Database
Introduction
Dengue is an acute febrile disease. Its clinical spectrum varies from asymptomatic and oligosymptomatic forms, to severe forms and even death.1 It is considered to be one of the most important arboviruses in the world.2 The dengue virus (DENV) is transmitted to humans by mosquitoes of the Aedes sp. genus and has four distinct serotypes: DENV-1, DENV-2, DENV-3 and DENV-4.1 All four serotypes have been in circulation in Brazil since 2010.1
The occurrence of dengue has been recorded in Brazil since 1846, while the first epidemic was recorded in 1981-1982 (Boa Vista, Roraima).1 The first cases in Belo Horizonte were recorded in 1996.3 Introduction of the four serotypes happened progressively,4 and between 2009 and 2014 all the serotypes circulated in the municipality at some time.5 In Belo Horizonte, virus identification is done by the Ezequiel Dias Foundation (FUNED) – the Minas Gerais state reference laboratory –, using virus isolation and reverse transcription polymerase chain reaction (RT-PCR) techniques. The laboratory results are input to the Laboratory Environment Manager (GAL).
In Brazil, all suspected dengue cases should be recorded on the Notifiable Health Conditions Information System (SINAN).1 Cases requiring hospitalization are recorded on the National Health System Hospital Information System (SIH/SUS).1 These systems do not have a unique identification field so that it is not possible to directly identify records of the same case on the different databases.
The database linkage technique has been used in Public Health analyses to detect and exclude duplicated records, as well as to integrate and aggregate information from different systems.6 In this way it is possible to identify and retrieve information held on one system but missing or incomplete on another,8 thus building a single and more complete database.6
Dengue epidemics place a significant burden on health services and the economy. Controlling the disease and reducing the density of its vector continue to be challenges. Severity of symptoms, as well as risk of epidemics, can depend on the serotype in circulation, so that it is important to assess and describe the dynamics of infections caused by DENV.
The objective of this study was to describe dengue virus circulation in Belo Horizonte between 2009 and 2014.
Methods
This is a dengue case series study.
Belo Horizonte is the capital of the state of Minas Gerais, located in the Southeast region of Brazil. In 2010, the municipality had a population of 2,375,151 inhabitants,10 divided into nine administrative regions (Barreiro, Centro Sul, Leste, Nordeste, Noroeste, Norte, Oeste, Pampulha and Venda Nova) coinciding with the territories of the Regional Health Directorates. The Regional Health Directorates are subdivided into 152 Health Center catchment areas.
The study included all dengue cases with positive DENV samples, analyzed by means of RT-PCR and/or virus isolation, between 2009 and 2014, in Belo Horizonte.
In order to identify the serotype – by-PCR and/or virus isolation –, blood samples were collected up to the fifth day following onset of symptoms and were sent to FUNED.
Serology testing and Non Structural 1 (NS1) antigen rapid testing were also assessed by the study as laboratory diagnosis methods. These methods do not allow serotype identification and were assessed as variables of interest but were not used as inclusion criteria. Serology tests are performed after the 6th day following the onset of symptoms and NSI testing is done up until the 5th day. In Belo Horizonte, the NS1 rapid test is used for sample screening for virus isolation. Positive samples and 10% of negative samples are sent to FUNED, up to a maximum limit of 90 samples a month.
The SINAN, SIH/SUS and GAL databases were linked. The three databases analyzed referred to the period between January 1st 2009 and December 31st 2014, comprising total records as follows: 263,995 (SINAN), 4,215 (SIH/SUS) and 4,392 (GAL).
The databases were prepared manually beforehand with regard to field standardization and coding. The same codes were applied for the ‘sex’ fields and the same data format was applied. The ‘race/skin color’, ‘methodology’ and ‘serotype’ fields were transformed into numerical characters.
The databases were imported into the MySQL Database Manager System (version 5.7) using the Front 5.2 tool. The databases used had some identifiers in common and, at this stage, a key was created with the ‘person’s name’, ‘mother’s name’ and ‘date of birth’ variables.
Considering the size of the databases used, the accuracy of linking techniques8and the use of manual revision to complement the strategy, the option was taken to use deterministic linkage (based on exact agreement).
The INNER JOIN keyword was used between the databases, in the sequence GAL ↔ SINAN and GAL ↔ SIH/SUS, to link the databases and identify common records.11 In the case of unpaired names, considering that there were missing or badly filled in fields, and in the attempt to pair once more, the integrity of the key was reduced by removing variables in the following order: (i) Year of birth; (ii) Month of birth; (iii) Day of birth; (iv) Mother’s 3rd surname; (v) Person’s 3rd surname; (vi) Mother’s 2º surname; (vii) Person’s 2nd surname; (viii) Mother’s name; and (ix) Person’s name.
Unlinked records were reviewed manually, by one of the researchers (ACLR), in an attempt to achieve pairing.
Virus monitoring data were retrieved from GAL; hospitalization data were retrieved from SIH/SUS; while the remaining data (sociodemographic, final classification laboratory diagnosis, case closure, case progression, clinical manifestations and other data) were retrieved from SINAN.
The variables studied and their categories were:
sex (male; female);
age range (in years: ≤10; 11-20; 21-30; 31-40; 41-50; >50);
race/skin color (white; black; brown; yellow);
schooling (illiterate; incomplete/complete elementary education; incomplete/complete high school education; incomplete/complete higher education);
serotype (DENV-1; DENV-2; DENV-3; DENV-4);
virus identification technique (virus isolation; RT-PCR);
final classification (classic dengue; dengue with complications; dengue hemorrhagic fever; dengue; dengue with warning signs; dismissed case);
case closure criterion (laboratory; clinical/epidemiological);
case progression (cure; death from dengue; death from other causes);
serology and rapid NSI test result (reactive; non-reactive; inconclusive);
hospitalization (yes; no);
time between symptom onset and hospitalization; and
length of hospital stay in days (mean; standard deviation [SD]; median).
Summary measures (mean; median; SD) were used for quantitative variables. Pearson’s chi-square test (X2) was used to check for association of the ‘sex’ and ‘age range’ variables with the virus serotypes, taking a 5% significance level.
During the study period there was a change in the classification of dengue cases used by the Ministry of Health. To overcome this problem, the study population was divided into two periods: 2009-2013, cases were classified as classic dengue (CD), dengue with complications (DCC), dengue hemorrhagic fever (DHF) and dengue shock syndrome (DSS); 2014 onwards, cases were classified as dengue (D), dengue with warning signs (DwWS) and severe dengue (SD). In order to analyze the dynamics or virus circulation in Belo Horizonte, CD and D cases were grouped together as ‘classic dengue’ and DCC, DHF, DSS, DwWS and SD cases were grouped together as ‘non-classic dengue’.
The databases were stored and analyzed on SPSS™ version 20.
The Study was approved by the Federal University of Minas Gerais Research Ethics Committee (CEP/UFMG): Certification of Submission for Ethical Appraisal (CAAE) No. 62191116.1.0000.5149, on December 7th 2016. Nominal data were used only for database linkage and participant privacy was preserved.
Results
Between 2009 and 2014, 250,632 suspected dengue cases in Belo Horizonte were notified on SINAN. 163,947 cases were confirmed and the highest notifications were found in 2010 (51,755) and 2013 (96,121). In the same period, 4,376 blood samples were sent to FUNED for DENV identification.
A total of 851 cases confirmed by virus isolation and/or RT-PCR were included in the study, 796 (93.5%) of which were recorded on SINAN. All 47 cases recorded on SIH/SUS were also recorded on SINAN. The case selection flowchart is shown in Figure 1. Table 1 shows the main sociodemographic characteristics of these cases. It is noteworthy that 52.6% of cases were female; 5.8% of cases were 10 years old or under, 72.2% were between 11 and 50 years old and 19.6% were over 50 years old, so that mean case age was 34.8 years (standard deviation: 17.8 years).
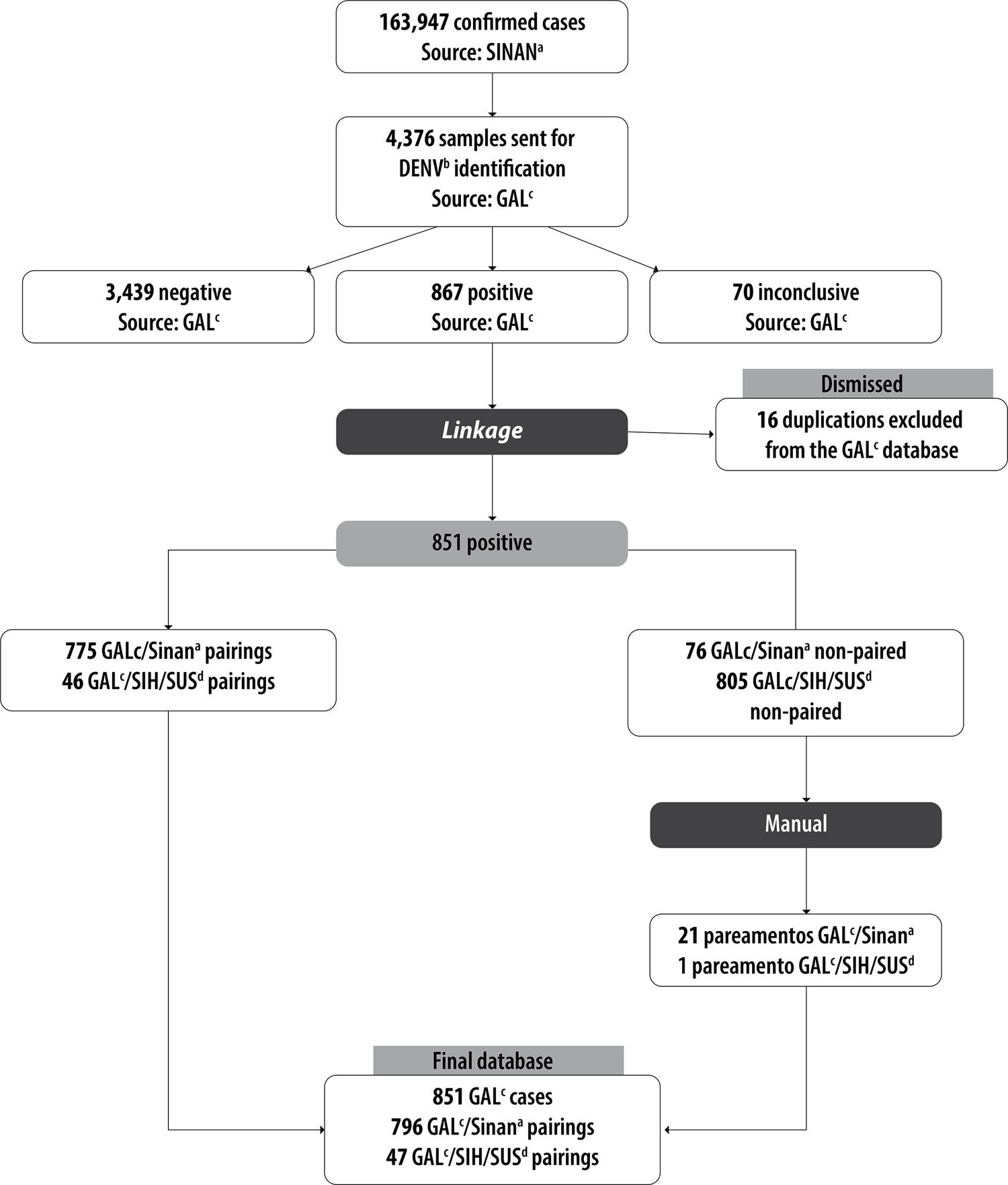
Figure 1 – Database linkage, Belo Horizonte, Minas Gerais, 2009-2014a) SINAN: Notifiable Health Conditions Information System.b) DENV: dengue virus.c) GAL: Laboratory Environment Manager.d) SIH/SUS: Brazilian National Health System Hospital Information System.
Table 1 – Sociodemographic characteristics of dengue cases confirmed by virus isolation and RT-PCR,a Belo Horizonte, Minas Gerais, 2009-2014
| Characteristic | n = 851 | % |
|---|---|---|
| Sex | ||
| Female | 448 | 52.6 |
| Male | 403 | 47.4 |
| Age (continuous variable) | ||
| Mean ± standard deviation | 34.8 ± 17.8 | |
| Median (interquartile ranges) | 31.6 (21.6;47.1) | |
| Age (in years – categorical variable) | ||
| ≤10 | 49 | 5.8 |
| 11-20 | 155 | 18.2 |
| 21-30 | 197 | 23.1 |
| 31-40 | 151 | 17.7 |
| 41-50 | 112 | 13.2 |
| >50 | 167 | 19.6 |
| Unknown/blank | 20 | 2.4 |
| Race/skin color | ||
| White | 214 | 25.2 |
| Black | 52 | 6.1 |
| Brown | 190 | 22.3 |
| Yellow | 6 | 0.7 |
| Unknown/blank | 389 | 45.7 |
| Schooling | ||
| Illiterate | 80 | 9.4 |
| Incomplete elementary education | 113 | 13.3 |
| Complete elementary education | 45 | 5.3 |
| Incomplete high school education | 51 | 6.0 |
| Complete high school education | 103 | 12.2 |
| Incomplete higher education | 22 | 2.6 |
| Complete higher education | 24 | 2.8 |
| Unknown/blank | 413 | 48.5 |
| Virus monitoring – serotype | ||
| DENV-1b | 514 | 60.4 |
| DENV-2b | 83 | 9.8 |
| DENV-3b | 66 | 7.7 |
| DENV-4b | 188 | 22.1 |
| Virus identification technique | ||
| Virus isolation | 572 | 67.2 |
| RT-PCRa | 170 | 20.0 |
| Unknown/blank | 109 | 12.8 |
| Final classification | ||
| Classic dengue | 590 | 69.3 |
| Dengue with complications | 6 | 0.7 |
| Dengue hemorrhagic fever | 3 | 0.4 |
| Dengue | 128 | 15.0 |
| Dengue with warning signs | 1 | 0.1 |
| Dismissed | 49 | 5.8 |
| Unknown/blank | 74 | 8.7 |
| Closure criterion | ||
| Laboratory | 693 | 81.4 |
| Clinical/epidemiological | 85 | 10.0 |
| Unknown/blank | 73 | 8.6 |
| Case progression | ||
| Cure | 662 | 77.8 |
| Death from dengue | 6 | 0.7 |
| Death from other causes | 1 | 0.1 |
| Unknown/blank | 182 | 21.4 |
| Serology | ||
| Reactive | 302 | 35.5 |
| Non-reactive | 32 | 3.8 |
| Inconclusive | 1 | 0.1 |
| Unknown/blank | 516 | 60.6 |
| Rapid NS1c test | ||
| Reactive | 369 | 43.4 |
| Non-reactive | 17 | 2.0 |
| Unknown/blank | 465 | 54.6 |
| Hospitalization | ||
| Yes | 47 | 5.5 |
| No | 804 | 94.5 |
a) RT-PCR: reverse transcriptase reaction, followed by polymerase chain reaction.
b) Dengue virus serotypes: 1, 2, 3 and 4.
c) NS1 antigen test.
The DENV-1 serotype was identified in 514 cases (60.4%), DENV-4 in 188 (22.1%), DENV-2 in 83 (9.8%) and DENV-3 in 66 (7.7%). Of the total positive samples, 67.2% were confirmed by virus isolation. In 2009 and 2010, three serotypes circulated in Belo Horizonte: DENV-1, DENV-2 and DENV-3. In 2011, only the DENV-1 serotype was isolated. In the period 2012-2014, the DENV-1 and DENV-4 serotypes were isolated (Figure 2). In 2010 and 2013, which were epidemic years, circulation of three and two serotypes was identified, respectively.
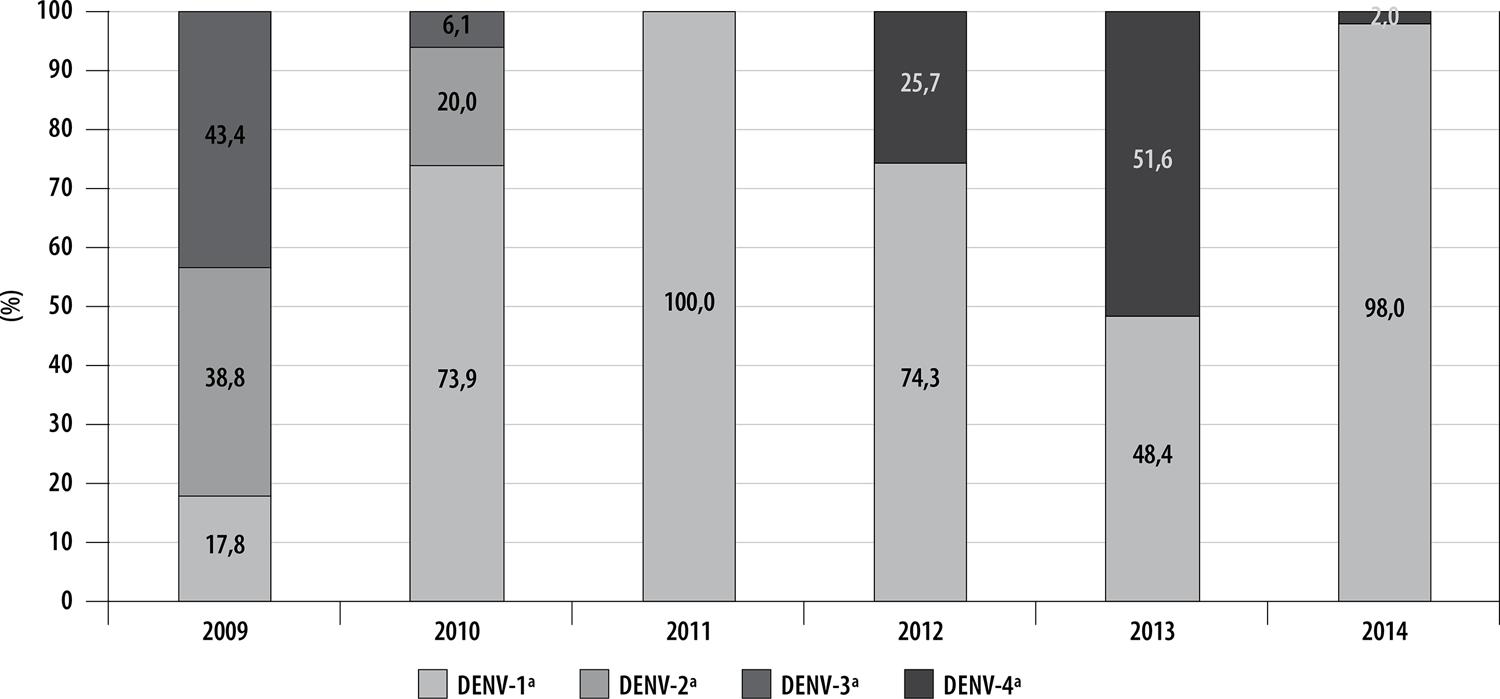
Figure 2 – Percentage of dengue virus serotypes by year of onset of symptoms, Belo Horizonte, Minas Gerais, 2009-2014a) Dengue virus serotypes – 1, 2, 3 and 4.
Among cases identified as having the DENV-1 and DENV-3 serotypes, the most frequent age range was 11-30 years (DENV-1, 44.9%; DENV-3, 43.8%). DENV-2 was identified more in people aged 11-30 (41;5%), while the DENV-4 serotype was identified more in the 31-50 age range (36.0%). There was no statistically significant difference in relation to ‘sex’ and ‘age range’ and circulating serotype (Table 2).
Table 2 – Classification and characterization by sex and age of confirmed cases by dengue virus serotype, Belo Horizonte, Minas Gerais, 2009-2014
| Classification and characteristics | Serotypes n (%) | Totalb | p-valuec | |||
|---|---|---|---|---|---|---|
| DENV-1a | DENV-2a | DENV-3a | DENV-4a | |||
| Case classification | ||||||
| Classic dengue | 442 (99.1) | 64 (95.5) | 52 (98.1) | 160 (98.8) | 718 (98.6) | – |
| ‘Non-classic’ dengue | 4 (0.9) | 3 (4.5) | 1 (1.9) | 2 (1.2) | 10 (1.4) | – |
| Sex | ||||||
| Female | 266 (51.8) | 46 (55.4) | 30 (45.5) | 106 (56.4) | 448 | 0.416 |
| Male | 248 (48.2) | 37 (44.6) | 36 (54.5) | 82 (43.6) | 403 | |
| Age range (in years) | ||||||
| ≤10 | 31 (6.2) | 4 (4.9) | 5 (7.8) | 9 (4.8) | 49 | 0.261 |
| 11-30 | 224 (44.9) | 34 (41.5) | 28 (43.8) | 66 (35.5) | ||
| 31-50 | 148 (29.7) | 24 (29.3) | 24 (37.5) | 67 (36.0) | 263 | |
| >50 | 96 (19.2) | 20 (24.4) | 7 (10.9) | 44 (23.7) | 167 | |
a) Dengue virus serotypes– 1, 2, 3 and 4.
b) Totals variable due to missing information for this variable in the information source.
c) Pearson’s chi-square test.
In 2009, the DENV-3 serotype was predominant in people under 10 years old, as well as those between 21-30 and 41-50 years old. DENV-2 was predominant in the 11-20 and over 50 age groups. In 2010, DENV-1 was the serotype with the highest percentage identified in all age ranges. In 2011, virus serotype was only identified in people who were 11-50 years old and only DENV-1 was isolated. In 2012, the serotype identified in the 10 years or under age range was DENV-4, while in the 11 years and over age ranges the predominant serotype was DENV-1. In 2013, the DENV-1 serotype was predominant among those under 30 years old, while the DENV-4 serotype was predominant among those over 31 years old. In the last year assessed, 2014, the predominant serotype in all age ranges was DENV-1 (Figure 3).
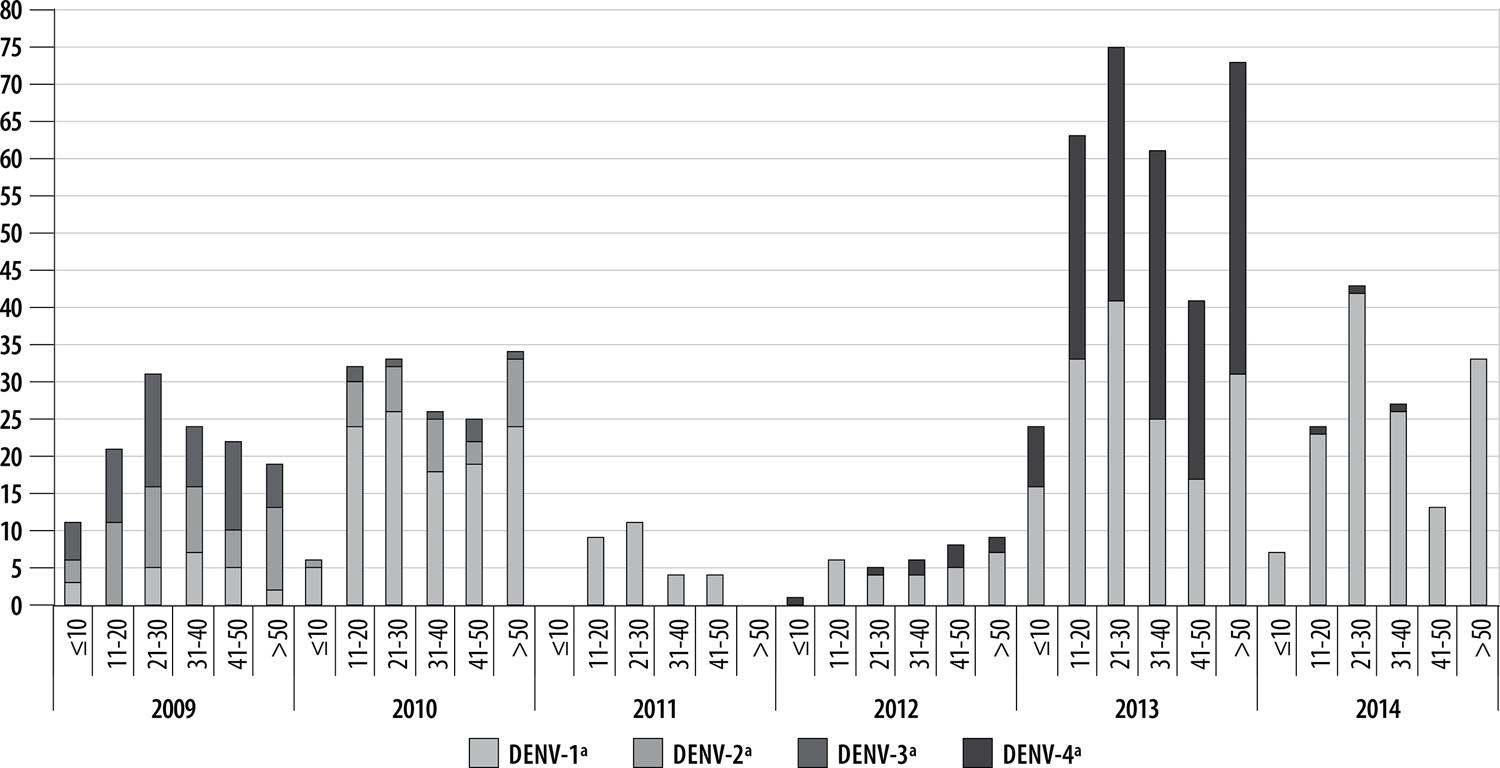
Figure 3 – Distribution of dengue virus serotypes by age range and year of onset of symptoms, Belo Horizonte, Minas Gerais, 2009-2014a) Dengue virus serotypes – 1, 2, 3 and 4.
Between 2009 and 2014, 718 cases (98.6%) were classified as classic dengue and 10 (1.4%) as dengue with complications, dengue hemorrhagic fever and dengue with warning signs, grouped together as non-classic dengue (Table 2). With regard to other laboratory diagnosis methods, 335 (39.4%) cases also had dengue serology tests (Table 1), whereby 302 (35.5%) had a reactive result, 32 (3.8%) were non-reactive and one (0.1%) was inconclusive. The rapid NS1 test was also performed for 386 cases and for 369 (43.4%) of them the test was reactive.
DENV-4 was the serotype identified for the highest percentage of rapid NS1 test (10.6%) and serology test (12.1%) negative cases. Serotypes DENV-2 and DENV-3 were not assessed for NS1 because in the years in which they circulated in Belo Horizonte, the rapid test was not in use.
Among cases for whom virus isolation and/or RT-PCR was performed, nine (1.1%) were classified as severe cases. Seven of them had hemorrhagic manifestations, two had hematuria and one had gastrointestinal bleeding.
With regard to the proportion of severe cases (non-classic) and classic cases, the highest percentage of severe cases (non-classic) was identified as having serotype DENV-2 (6.0%); while the highest percentage of classic dengue cases was identified as having the DENV-4 serotype (98.8%).
Of the total cases in the study, 47 (5.5%) were hospitalized and three (6.4%) needed care in an intensive therapy unit (ITU). Of the total hospitalized cases, 61.7% (n=29) were female and 66.0% (n=18) were between 11 and 50 years old. Mean time elapsing between onset of symptoms and hospitalization was 5 days (SD ±5), while the median was 4 days (interquartile ranges [IQ] 3;6). The mean number of hospital stay days was 3.8 (SD ±2.3), while the median was 3.0 days (IQ 2.0;5.0).
Among hospitalized cases, 61.7% (n=29) were identified as having DENV-1, 25.5% (N=12) as having DENV-4, 10.6% (n=5) as having DENV-2 and 2.1% (n=1) as having DENV-3. With regard to ITU use, the highest percentage was found for cases with DENV-4: 66.7% (n=2).
With regard to case closure, 693 (89.1%) were closed according to the laboratory criterion and 85 according to the clinical/epidemiological criterion (10.9%), despite the virus identification test result being positive. Of the total of cases for whom the ‘case progression’ field had been filled in, 662 (99.0%) progressed to cure, six (0.9%) died from dengue and one (0.1%) died from other causes. The highest absolute number of dengue deaths (n=4) occurred in people with the DENV-1 serotype. One case with the DENV-3 serotype died from dengue and another with the DENV-4 serotype died from other causes.
Discussion
Among dengue cases notified in Belo Horizonte between 2009 and 2014 and confirmed by virus isolation and/or RT-PCR, this study identified predominance of the DENV-1 serotype, which circulated throughout the entire period assessment, followed by DENV-4, DENV-2 and DENV-3.
The pattern of circulating dengue serotypes followed the national pattern,12 and the positivity of virus identification in Belo Horizonte was found to be below the national average. In Brazil as a whole , percentage positivity of virus identification in samples sent to reference laboratories was 36.8% in 2014,13 and 39.2% in 2015.14 In this study, among the samples tested, the highest percentage of seropositive results was 28.0% in 2013, an endemic year in which DENV-1 and DENV-4 circulated. A study conducted between 2010 and 2014 in Cuiabá, capital of the state of Mato Grosso, also found low percentage positivity and that 2012 was the year with the highest percentage of positive cases (23.1%).15
In this study, females were predominant for all serotypes. This is in agreement with the findings of Martins et al. in relation to Salvador, capital of the state of Bahia, for the period 2007-2014.16
By grouping cases together as classic cases and non-classic cases, we found that the percentage of cases classified as non-classic (DCC, DHF, DSS, DSCA and SD) is almost five times greater than that found on the municipal SINAN for the same period, when 0.3% of cases were non-classic. A study conducted in a public hospital in Rio de Janeiro, RJ, in 2008, during one of the biggest epidemics recorded in that municipality, found 6.7% non-classic cases.17
DENV-2 was the serotype with the highest percentage of severe cases in this study. Higher percentages of hospitalized cases were found for the DENV-4 and DENV-2 serotypes. These findings corroborate those of national study conducted by Burattini et al.,18 which showed a higher proportion of hospitalization related to DENV-2 serotype infection.
Distribution of hospitalizations by sex and age range was similar to cases reported in other places and periods. Between 1997 and 2002, in Belo Horizonte, a higher percentage of hospitalization of suspected dengue cases was reported among females (53.0%).19 The findings of our study corroborate those of that earlier study, according to which 60.4% of hospitalized cases were female. With regard to age range, between 1997 and 2002, in Belo Horizonte, the highest proportion of hospitalization due to dengue occurred in the 19-59 age group (65.0%),19 while in Goiânia, the capital of the state of Goiás, in the 2013 epidemic, the highest percentage of hospitalizations was among people who were 21-50 years old (45.8%) or older (27.1%)– the main age range hospitalized was the over 50s (37.5%).20
In a study about the state of Minas Gerais, Campos21 found that on average hospitalization took place after four days following onset of symptoms in children and the elderly, and after five days following onset of symptoms in young people and adults. Our findings are similar to those of Campos, according to whom average number of hospitalization days (two days)21was almost half that found in this study (3.8 days hospitalization).
It is noteworthy that in the years assessed, according to SIH/SUS data, overall percentage hospitalization due to dengue in Belo Horizonte was 2.2%, while our study found 5.5%. The fact of the rapid NS1 test, used for screening samples for virus identification, being available at emergency units and hospitals, usually dedicated to caring for more severe cases, may explain the higher percentage of hospitalization of virus isolation and/or RT-PCR positive cases.
The percentage of dengue deaths recorded on SINAN for the period and place studied was 0.02%. Our study found a percentage 4.5 times higher (0.9%), among those cases for which the ‘case progression’ variable had been filled in. This result may also be explained because of collection of samples for virus detection in severe cases.
With regard to laboratory diagnosis performed using other methods, of the total cases with positive virus isolation and/or RT-PCR for which NS1 was also performed, the rapid test result was positive for 95.5%. The highest percentage of negative NS1 tests corresponded to cases infected with the DENV-4 serotype. There are reports in the literature that differences in sensitivities between serotypes demonstrate that NS1 is less sensitive for detecting DENV-4 cases.22
In the period from 2009 to 2014, filling in the clinical manifestations field was not obligatory for classic dengue cases. It is supposed that the majority of classic cases had classical symptoms (fever starting suddenly, together with at least two of the following signs and symptoms: nausea/vomiting; exanthema; myalgia/arthralgia; headache/retro-orbital pain; petechiae/positive loop test; leukopenia)1. Moreover, in epidemic years, there is great difficulty in filling in notification forms with a large number of variables.24 As such, it was not possible to analyze associations between clinical manifestations and the different serotypes.
Among cases infected with the DENV-1 serotype, 1.2% had negative NS1, while among those infected with DENV-4, 10.6% had a negative NS1 test result. This finding emphasizes the importance of sending a percentage of NS1 negative samples for virus isolation. The municipality of Belo Horizonte currently sends 10.0% of negative samples for virus isolation, this being a percentage similar to that found when assessing the DENV-4 serotype.
The data linkage technique enabled the use of complementary information from different databases, optimizing working hours and showing itself to be viable from the operational point of view. Deterministic linkage has greater specificity; however, this technique can “dismiss” records with potential for pairing, because of inconsistencies or typing errors.25 In turn, probabilistic linkage increases the chance of pairing, but generates a larger number of pairs for manual reviewing, this being the main problem described in other papers.26 In view of its greater accuracy, use of deterministic linkage was defined for this study.
Standing out among the limitations of this study is the use of secondary data from databases with potential for losses, omission and/or incorrect records. The linkage technique allowed us to review the databases, assess information completeness and analyze the dynamics of virus circulation in Belo Horizonte. In addition, selection bias may occur in performing virus identification tests, given that the test used for sample screening is available in emergency units and hospitals that usually provide care to more severe cases. Moreover, the virus isolation technique is performed up until the fifth day after symptom onset, thus reducing the number of people with an adequate sample for this test, considering those who do not seek health care immediately. The way the sample is stored may also interfere with the possibility of performing this test.29
Correct and quality completion of notification forms is essential for studies related to health and is beneficial for decision making. Lack of basic information on clinical manifestations on notification forms makes more in-depth analysis of the characteristics of infections caused by different DEN-V serotypes impossible. Notwithstanding, as mentioned above, in epidemic years, filling in lengthy notification forms is practically impossible.24
The findings of this study emphasize the importance of good practices in relation to sample collection, storage and transportation, along with timely and adequate selection of cases in order to meet virus identification criteria. Implementation of these actions is feasible and they have positive impact on serotype identification and monitoring the dynamics of dengue.
We reiterate the importance of analyzing and describing characteristics of different DENV serotype infections. The recent history of dengue in Belo Horizonte and current epidemiological/entomological scenarios show greater susceptibility to the occurrence of new epidemics, highlighting that it is fundamental to gain more in-depth knowledge of the disease capable of aiding the formulation of more effective Public Health policies.
REFERENCES
1. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Coordenação-Geral de Desenvolvimento da Epidemiologia em Serviços. Guia de vigilância em saúde [Internet]. Brasília: Ministério da Saúde; 2016 [citado 2020 maio 19]. 773 p. Disponível em: https://www.saude.gov.br/images/pdf/2016/agosto/25/GVS-online.pdf [ Links ]
2. Tauil P. L. Prefácio. In: Valle D, Pimenta DN, Cunha RV, organizadores. Dengue: teorias e práticas. Rio de Janeiro: Editora Fiocruz; 2015. p. 11-13. [ Links ]
3. Secretaria Municipal de Saúde (Belo Horizonte). Balanço da dengue [Internet]. Belo Horizonte: Secretaria Municipal de Saúde; 2018 [citado 2018 jan 8]. Disponível em: http://www.pbh.gov.br/smsa/dengue/balanco_dengue.php [ Links ]
4. Pessanha JEM, Caiaffa WT, Kroon EG, Proietti FA. Dengue em três distritos sanitários de Belo Horizonte, Brasil: inquérito soroepidemiológico de base populacional, 2006 a 2007. Rev Panam Salud Publica [Internet]. 2010 [citado 2020 maio 19];27(4):252–8. Disponível em: https://scielosp.org/pdf/rpsp/2010.v27n4/252-258/pt [ Links ]
5. Fundação Ezequiel Dias – FUNED. Gal [Internet]. Belo Horizonte; FUNED; 2018 [citado 2018 jan 21]. Disponível Em: Http://Www.Funed.Mg.Gov.Br/Tag/Gal/ [ Links ]
6. Bartholomay P, Oliveira GP, Pinheiro RS, Vasconcelos AMN. Melhoria da qualidade das informações sobre tuberculose a partir do relacionamento entre bases de dados. Cad Saúde Pública [Internet]. 2014 nov [citado 2020 maio 19];30(11):2459-70. Disponível em: http://dx.doi.org/10.1590/0102-311X00116313 [ Links ]
7. Soeiro CMO, Miranda AE, Saraceni V, Santos MC, Talhari S, Ferreira LCL. Syphilis in pregnancy and congenital syphilis in Amazonas State, Brazil: an evaluation using database linkage. Cad Saúde Pública [Internet]. 2014 abr [citado 2020 maio 19];30(4):715-23. Disponível em: http://dx.doi.org/10.1590/0102-311X00156312 [ Links ]
8. Oliveira GP, Bierrenbach ALS, Camargo Júnior KR, Coeli CM, Pinheiro RS. Acurácia das técnicas de relacionamento probabilístico e determinístico: o caso da tuberculose. Rev Saúde Pública [Internet]. 2016 ago [citado 2020 maio 19];50:49. Disponível em: https://doi.org/10.1590/S1518-8787.2016050006327 [ Links ]
9. Rosselto EV, Luna EJA. Relacionamento entre bases de dados para vigilância da pandemia de influenza A(H1N1) pdm09, Brasil, 2009-2010. Cad Saúde Pública [Internet]. 2016 jul [citado 2020 maio 19];32(7):e00014115. Disponível em: https://doi.org/10.1590/0102-311X00014115 [ Links ]
10. Instituto Brasileiro de Geografia e Estatística - IBGE. Síntese: Belo Horizonte [Internet]. Rio de Janeiro: Instituto Brasileiro de Geografia e Estatística; 2017 [citado 2017 dez 17]. Disponível em: https://cidades.ibge.gov.br/brasil/mg/belo-horizonte/panorama [ Links ]
11. Plataforma Devmedia. SQL JOIN: entenda como funciona o retorno de dados. [Internet]. [S.l.]: Plataforma Devmedia; 2014 [citado 2020 fev 11]. Disponível em https://www.devmedia.com.br/sql-join-entenda-como-funciona-o-retorno-dos-dados/31006 [ Links ]
12. Teixeira MG, Siqueira Júnior JB, Ferreira GLC, Bricks L, Joint G. Epidemiological trends of dengue disease in Brazil (2000-2010): a systematic literature search and analysis. PLoS Negl Trop Dis [Internet]. 2013 Dec [cited 2020 May 19];19(7):e2520. Available from: https://doi.org/10.1371/journal.pntd.0002520 [ Links ]
13. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Dengue: monitoramento até a Semana Epidemiológica (SE) 32 de 2014. Bol epidemiol [Internet]. 2014 [citado 2018 mar 5];45(19):1-6. Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2014/setembro/01/Boletim-Dengue-SE32.pdf [ Links ]
14. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre de chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 48, 2015. Bol Epidemiol [Internet]. 2015 [citado 2018 mar 5];46(44):1-9. Disponível em: http://portalarquivos2.saude.gov.br/images/pdf/2016/janeiro/07/2015-svs-be-pncd-se48.pdf. Acesso em 05 de março de 2018 [ Links ]
15. Governo do Mato Grosso. Secretaria de Estado de Saúde. Diagnóstico laboratorial específico e nova classificação da dengue [Internet]. Cuiabá: Secretaria de Estado de Saúde ; 2015 [citado 2019 mar 5]. Disponível em: http://www.telessaude.mt.gov.br/Arquivo/Download/2075 [ Links ]
16. Martins MMF, Almeida AMFL, Fernandes NDR, Silva LS, Lima TB, Orrico AS, et al. Análise dos aspectos epidemiológicos da dengue na microrregião de saúde de Salvador, Bahia, no período de 2007 a 2014. Rev Espaço Saúde [Internet]. 2015 out-dez [citado 2020 maio 19];16(4):64-73. Disponível em: http://www.uel.br/revistas/uel/index.php/espacoparasaude/article/download/23300/9 [ Links ]
17. Escosteguy CC, Pereira AGL, Medronho RA, Rodrigues CS, Chagas KKF. Diferenças, segundo faixa etária, do perfil clínico-epidemiológico dos casos de dengue grave atendidos no Hospital Federal dos Servidores do Estado, Rio de Janeiro-RJ, Brasil, durante a epidemia de 2008. Epidemiol Serv Saúde [Internet]. 2013 mar [citado 2020 maio 19];22(1):67-76. Disponível em: https://doi.org/10.5123/S1679-49742013000100007 [ Links ]
18. Burattini MN, Lopez LF, Coutinho FAB, Siqueira Júnior JB, Homsani S, Sarti E, et al. Age and regional differences in clinical presentation and risk of hospitalization for dengue in Brazil, 2000–2014. Clinics [Internet]. 2016 ago [citado 2020 maio 19];71(8):455-63. Disponível em: http://dx.doi.org/10.6061/clinics/2016(08)08 [ Links ]
19. Duarte HH, França EB. Data quality of dengue epidemiological surveillance in Belo Horizonte, Southeastern Brazil. Rev Saúde Pública [Internet]. 2006 fev [citado 2020 maio 19];40(1):134-42. Disponível em: http://dx.doi.org/10.1590/S0034-89102006000100021 [ Links ]
20. Nascimento LB, Oliveira PS, Magalhães DP, França DDS, Magalhães ALA, Silva JB, et al. Caracterização dos casos suspeitos de dengue internados na capital do estado de Goiás em 2013: período de grande epidemia. Epidemiol Serv Saúde [Internet]. 2015 jul-set [citado 2020 maio 19];24(3):475-484. Disponível em: http://dx.doi.org/10.5123/S1679-49742015000300013 [ Links ]
21. Campos KB. Características clínico-epidemiológicas dos casos graves e óbitos por dengue, minas gerais, 2008 a 2010. Belo Horizonte, 2012 [dissertação]. Belo Horizonte: Universidade Federal de Minas Gerais; 2012. Disponível em: https://repositorio.ufmg.br/handle/1843/BUOS-8ZKMJZ [ Links ]
22. Bessoff K, Delorey M, Sun W, Hunsperger E. Comparison of two commercially available dengue virus (denv) ns1 capture enzyme-linked immunosorbent assays using a single clinical sample for diagnosis of acute denv infection. Clin Vaccine Immunol [Internet]. 2008 Oct [cited 2020 May 19];15(10):1513-8. Available from: https://doi.org/10.1128/cvi.00140-08 [ Links ]
23. Mcbride WJH. Evaluation of dengue ns1 test kits for the diagnosis of dengue fever. Diagn Microbiol Infect Dis [Internet]. 2009 May [cited 2020 May 19];64(1):31-6. Available from: https://doi.org/10.1016/j.diagmicrobio.2009.01.002 [ Links ]
24. Barbosa JK, Barrado JCS, Zara ALSA, Siqueira Júnior JB. Avaliação da qualidade dos dados, valor preditivo positivo, oportunidade e representatividade do sistema de vigilância epidemiológica da dengue no Brasil, 2005 a 2009. Epidemiol Serv Saúde [Internet]. 2015 jan-mar [citado 2020 maio 19];24(1):49-58. Disponível em: https://doi.org/10.5123/S1679-49742015000100006 [ Links ]
25. Sayers A, ben-Shlomo Y, Blom AW, Steele F. Probabilistic record linkage. Int J Epidemiol [Internet]. 2015 Jun [cited 2020 May 19];45(3):954-64. Available from: https://doi.org/10.1093/ije/dyv322 [ Links ]
26. Capuani L, Bierrenbach AL, Abreu F, Takecian PL, Ferreira JE, Sabino EC. Accuracy of a probabilistic record-linkage methodology used to track blood donors in the Mortality Information System database. Cad Saúde Pública [Internet]. 2014 Aug [cited 2020 May 19];30(8):1623-32. Available from: https://doi.org/10.1590/0102-311X00024914 [ Links ]
27. Camargo Júnior KR, Coeli CM. RecLink 3: nova versão do programa que implementa a técnica de associação probabilística de registros (probabilistic record linkage). Cad Saúde Coletiva. 2006 abr-jun;14(2):399-404. [ Links ]
28. Coeli CM, PinheiroRS, Camargo Júnior KR. Conquistas e desafios para o emprego das técnicas de record linkage na pesquisa e avaliação em saúde no Brasil. Epidemiol Serv Saúde [Internet]. 2015 out-dez [citado 2020 maio 19];24(4):795-802. Disponível em: http://dx.doi.org/10.5123/S1679-49742015000400023 [ Links ]
29. Fundação Ezequiel Dias - FUNED. Manual de coleta, acondicionamento e transporte de material biológico para exames laboratoriais [Internet]. Belo Horizonte: Fundação Ezequiel Dias; 2018 [citado 2020 maio 19]. 161 p. Disponível em: http://www.funed.mg.gov.br/wp-content/uploads/2018/10/manual_coleta_amostras_biologicas_Funed.pdf [ Links ]
Associate Editor: Luciana Guerra Gallo - orcid.org/0000-0001-8344-9951
*Article derived from the dissertation entitled ‘Database linkage and descriptive analysis of dengue cases confirmed by virus isolation and polymerase chain reaction in the municipality of Belo Horizonte, 2009-2014’, defended by Ana Carolina Lemos Rabelo at the Health Sciences Postgraduate Program, Infectious Diseases and Tropical Medicine Division, Federal University of Minas Gerais (UFMG), in 2018.
Received: December 10, 2019; Accepted: April 24, 2020










 texto en
texto en