Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Epidemiologia e Serviços de Saúde
versão impressa ISSN 1679-4974versão On-line ISSN 2237-9622
Epidemiol. Serv. Saúde vol.29 no.4 Brasília 2020 Epub 21-Ago-2020
http://dx.doi.org/10.5123/s1679-49742020000400020
Original article
Increase in Guillain-Barré syndrome hospitalizations in Brazil: an ecological study*
1Universidade de Brasília, Programa de Pós-Graduação em Medicina Tropical, Brasília, DF, Brazil
2Ministério da Saúde, Secretaria de Vigilância em Saúde, Brasília, DF, Brazil
Objective
to describe the demographic characteristics and the spatio-temporal dynamics of Guillain-Barré syndrome (GBS) hospitalizations in Brazil between 2008 and 2017.
Methods
this is an ecological study using data from the Hospital Information System of the Brazilian National Health System (SIH/SUS); GBS hospitalization rates were calculated and a control diagram was built; natural break ranges were used in the spatial analysis.
Results
15,512 GBS hospitalizations were recorded during the study period; between 2008-2014 there were 1,344 hospitalizations per year on average, in the following year (2015), 1,953 hospitalizations were registered, representing an increase of 45% in relation to the average of previous years; GBS hospitalizations reached an epidemic level in the Northeast region in 2015 and 2016.
Conclusion
GBS hospitalizations increased with effect from 2015, following the introduction of chikungunya virus and the rapid spread of Zika virus in Brazil.
Key words: Guillain-Barre Syndrome; Hospitalization; Epidemiology; Brazilian National Health System; Ecological Studies
Introduction
In 1916, Georges Guillain, J. A. Barré and A. Strohi described a syndrome characterized by acute paralysis and loss of reflexes among French army soldiers. On this occasion, occurrence of cerebrospinal fluid albuminocytologic dissociation was described for the first time. Strohi’s participation was limited to electrophysiology. Approximately four decades later, in honor of its investigators, the disease received the name of Guillain-Barré syndrome (GBS).1
GBS is a peripheral polyneuropathy, characterized by starting with acute bilateral and symmetric weakness of the limbs with reduced or absent myotactic reflexes. The disease is progressive and reaches its peak in between 12 hours and 28 days, followed by a plateau and subsequent improvement in survivors. GBS is generally triggered by an acute infectious process that occurs days or weeks before neurological symptoms.2
Several different prior infections have been identified in GBS patients. C jejuni is the most frequently identified prior infectious agent. Other infections associated with the syndrome are those caused by cytomegalovirus (CMV), Epstein-Barr virus, influenza A virus, Mycoplasma pneumoniae and Haemophilus influenzae.3 An emerging relationship between GBS and acute infections caused by arboviruses, including those cased by chikungunya virus and Zika virus, is being monitored and is a subject of great interest.5
Studies show that the disease is more frequent among males. The explanation for the difference between the sexes is unknown. There are no reports of the syndrome occurring seasonally.5
Around 25% of GBS cases develop respiratory insufficiency and many have signs of autonomic dysfunction in the acute phase of the disease. Approximately 5% of cases die and 20% suffer significant permanent disability.3
Diagnosis is generally clinical, although cerebrospinal fluid albuminocytologic dissociation and electrophysiological studies can help diagnosis and differentiate the demyelinating and axonal GBS subtypes.3 Specific treatment of GBS aims above all to accelerate the recovery process, reducing complications associated with the acute phase and reducing residual neurological deficits in the long term. Treatment management includes use of plasmapheresis and intravenous human immunoglobulin, both of which are available on the Brazilian National Health System (SUS).7
In many parts of the world GBS incidence is, however, unknown. Some evidence suggests that GBS epidemiology may be different in different regions. A study conducted in order to obtain estimates of GBS incidence in North America and Europe used publications dating from 1973 to 2009, and found a median of 1.1 case per 100,000 inhabitants/year.8
A study conducted in the Brazilian state of Rio Grande do Norte between 1994 and 2007, found incidence of 0.3 case per 100,000 inhab./year.9 In the state of São Paulo, another study conducted in a tertiary hospital found incidence of 0.6 case per 100,000 inhab./year.10
In 2015, the Brazilian Ministry of Health’s National Dengue Control Program recorded an increase in the number of SUS GBS hospitalizations, especially in the Northeastern states of Pernambuco and Bahia.11 This increase in cases was detected by the SUS Hospital Information System (SIH/SUS), the purpose of which is to record all care provided as a result of hospitalizations funded by the system, in both public and outsourced hospitals.
In view of the growing records of GBS hospitalizations in Brazil, above all following the introduction of chikungunya virus and the rapid spread of Zika virus, it is justifiable to conduct studies that contribute to knowledge about the epidemiological scenario of the syndrome, as well as to informing decision making and public health policy planning, at a time when studies are being conducted to gain better understanding of arbovirus circulation and their complications.
The objective of this study was to describe the demographic characteristics and spatio-temporal dynamics of Guillain-Barré syndrome hospitalizations in Brazil between 2008 and 2017.
Methods
A mixed-type ecological study was conducted of SUS-funded GBS hospitalizations in the period 2008-2017, taking Brazil’s Federative Units (UFs) and regions as its units of analysis.
Brazil is divided into five geopolitical regions – North, Northeast, Midwest, Southeast and South –, all of which have different demographic, economic and social characteristics. There are 26 states and one Federal District. Brazil is South America’s largest country (8,515,767km2).12
The study used the SIH/SUS database. Resident populations, by region and UF, between 2008 and 2017 were estimated by the Brazilian Institute of Geography and Statistics (IBGE), based on the 2010 Demographic Census; estimated IBGE projections were used for the other years.
The study included all hospitalizations the main diagnosis of which was recorded as code G61.0 of the International Statistical Classification of Diseases and Related Health Problems – 10th revision (ICD-10).
A ten-year time series of GBS hospitalizations (2008-2017) was built. Frequencies and measures of central tendency and dispersion were used to describe the data. The demographic variables and variables relating to the disease were analyzed according to proportional contribution to total cases in each year of the time series, thus enabling possible changes over the period to be visualized. Likewise, proportional distribution was calculated for the UFs, so that it could be seen how much each one contributed to total GBS hospitalizations. The hospitalization rate was calculated for the 27 UFs, taking the number of people hospitalized due to GBS as the numerator and the population in the period as the denominator, multiplied by 100,000 inhabitants. Lethality was calculated by dividing the number of GBS deaths in Brazil each year by the total number of GBS hospitalizations in Brazil each year, multiplied by 100.
The number of inpatient days was used to calculate length of hospital stay. An inpatient day is when a patient stays for an indivisible period of up to 24 hours in a hospital institution. The start time and end time of the period considered in SIH/SUS calculations is midnight.13
The Cullen method was used for the control diagram, which was built with the aim of detecting epidemics in the GBS hospitalization time series. The control diagram encompasses two sets of information: a control graph, representative of the endemic channel; and a follow-up graph. The period observed was 2014-2017. Prior year data was used as the reference for building the endemic strip, which became the same for the three years.14 An expected monthly value was obtained for each month, by means of the GBS hospitalization incidence coefficient (average I.C. per month), based on preceding years. Monthly rate standard deviation (SD) was then calculated. The expected upper limit was calculated as follows: (average I.C. per month) + (standard deviation per month x 1.96). The same procedure was used for all the following months.
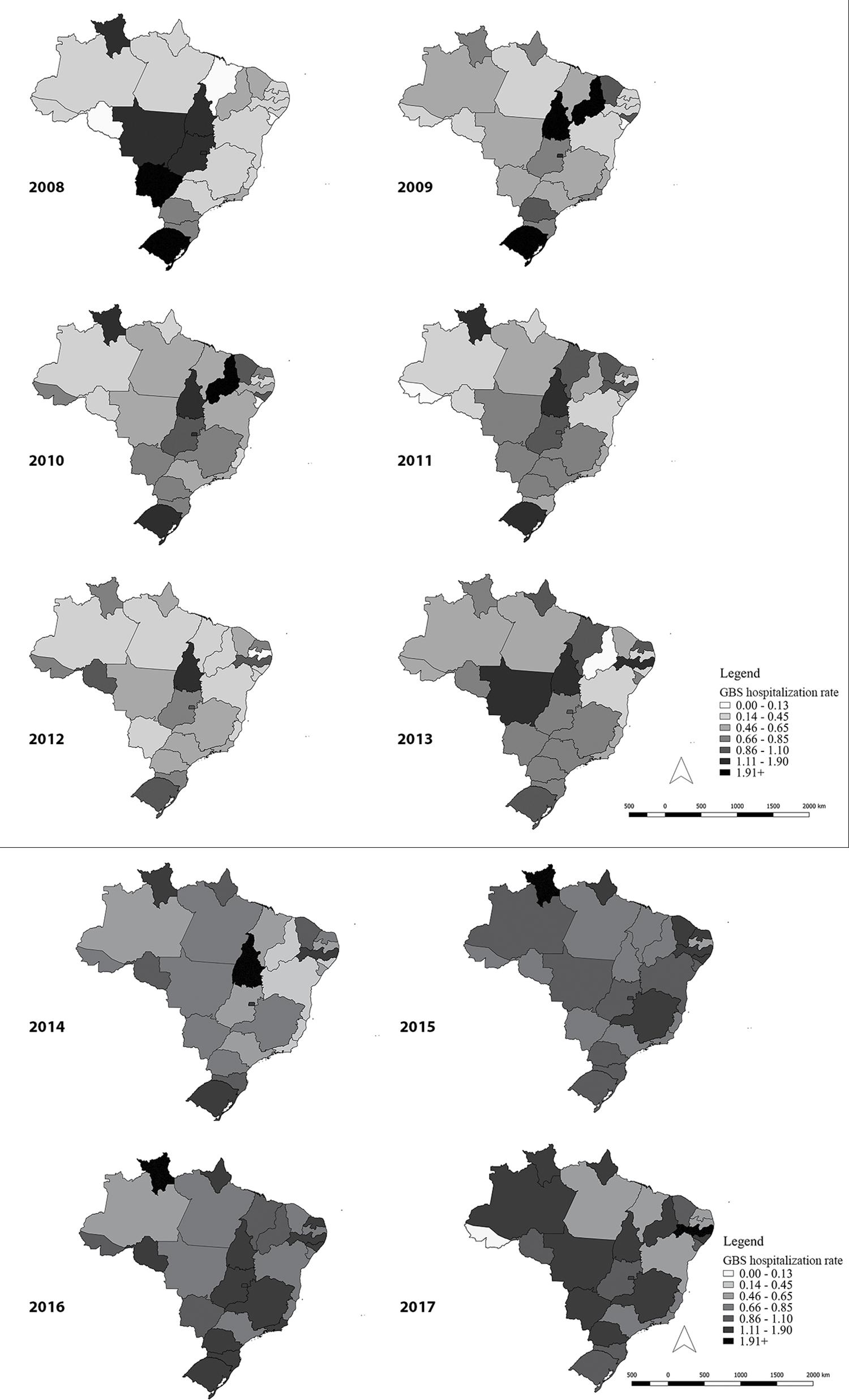
The natural break ranges of the data were used for graphical demonstration of incidence rate spatial distribution in the UFs, i.e.: 0.00-0.13; 0.14-0.45; 0.45-0.65; 0.66-0.85; 0.86-1.10; 1.11-1.90; 1.91 or more hospitalizations/100,000 inhab.
Epi Info™ 7.2.2, Tabwin™ 3.6 and Microsoft Office® 2010 were used to analyze the data. QGIS version 2.1.8 was used to prepare the maps.
Results
Between 2008 and 2017, 15,512 GBS hospitalizations were recorded in Brazil. Generally speaking, hospitalizations increased with effect from 2015 (Table 1). Between 2008 and 2014, hospitalizations were stable at an average rate of 1,344 per annum. In the following year (2015), 1,953 GBS hospitalizations were recorded, representing a 45% increase. 2016 stands out as the year with the highest number of hospitalizations: N=2,216 (Table 1).
Table 1 – Distribution of Guillain-Barré syndrome hospitalizations (N=15,512) according to demographic data and related to the disease, Brazil, 2008-2017
| Year | Guillain-Barré syndrome hospitalizations | Sex | Age | Length of hospital stay, in days | Intensive therapy unit n (%) | Deaths lethality: n (%) | |
|---|---|---|---|---|---|---|---|
| Male: n (%) | Female: n (%) | Median (Q1-Q3) | Median (Q1-Q3) | ||||
| 2008 | 1,167 | 711 (60.9) | 456 (39.1) | 45 (28-59) | 6 (04-13) | 112 (9.6) | 33 (2.8) |
| 2009 | 1,350 | 796 (59.0) | 554 (41.0) | 42 (25-57) | 6 (04-12) | 133 (9.9) | 30 (2.2) |
| 2010 | 1,405 | 784 (55.8) | 621 (44.2) | 42 (25-57) | 7 (04-14) | 181 (12.9) | 33 (2.3) |
| 2011 | 1,364 | 833 (61.1) | 531 (33.9) | 41 (25-55) | 7 (04-14) | 189 (13.9) | 48 (3.5) |
| 2012 | 1,218 | 698 (57.3) | 520 (42.7) | 39 (21-55) | 8 (04-15) | 210 (17.2) | 46 (3.8) |
| 2013 | 1,449 | 832 (57.4) | 617 (42.6) | 39 (22-54) | 8 (04-16) | 254 (17.5) | 47 (3.2) |
| 2014 | 1,455 | 805 (55.3) | 650 (44.7) | 38 (20-54) | 8 (04-14) | 259 (17.8) | 38 (2.6) |
| 2015 | 1,953 | 1,111 (56.8) | 842 (43.2) | 40 (25-56) | 8 (04-14) | 380 (19.5) | 59 (3.0) |
| 2016 | 2,216 | 1,294 (58.4) | 922 (41.6) | 40 (25-56) | 8 (04-15) | 433 (19.5) | 97 (4.4) |
| 2017 | 1,935 | 1,141 (59.0) | 794 (41.0) | 39 (22-55) | 8 (04-14) | 369 (19.1) | 71 (3.7) |
Q1: 1st quartile.
Q3: 3rd quartile.
The male sex was predominant throughout the entire period: 58.0%. Mean age of hospitalized patients was around 40 years old; except in 2008 when mean age was 45 years.
A gradual increase in length of hospital stay in days was seen between 2008 and 2017. However, between 2012 and 2017, mean length of hospital stay remained steady at eight days. A gradual increase was also seen in the number of patients cared for in intensive therapy units (ITU): rising from 9.6% in 2008 to 19.5% in 2015. The number of deaths per year varied between 30 and 97, with 2016 standing out as the year with highest lethality (4.4%).
Table 2 shows the GBS hospitalization time series in the states and regions, and their respective line graphs. An increase in hospitalizations can be seen with effect from 2015, in comparison to the preceding year, in the Northeast, Southeast, Midwest and Southern regions, with increases of 78.3%, 35.0%, 18.5% and 7.7% respectively.
Table 2 – Distribution of Guillain-Barré syndrome hospitalizations (N=15,512) by region and Federative Unit of residence, Brazil, 2008-2017
The Northeastern states of Ceará and Bahia registered the largest number of GBS hospitalizations in 2015, in relation to the entire period studied, with 115 and 162 records, respectively. Although there was an increase in hospitalizations in the state of Pernambuco in 2015, the highest number of cases in that state was recorded in 2017: 189 hospitalizations.
In the Southeast region, the highest number of GBS hospitalizations in the study period (2008-2017) occurred in 2016, with 909 records. This behavior was also found in 2016 with relation to Minas Gerais, with 311 hospitalizations, Rio de Janeiro with 192, and São Paulo with 377.
Although it recorded the highest number of GBS hospitalizations in 2008 and 2009 (457 and 395 cases, respectively), the Southern region had a gradual increase following this period, in particular in 2016 with 348 hospitalizations.
In the Midwest region, GBS hospitalizations were seen to increase with effect from 2015, with a peak of 236 records in 2016, and the state of Goiás with an increase of 69.2% in 2016 compared to the year before.
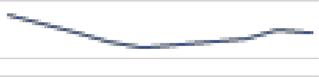
The Guillain-Barré syndrome hospitalization control diagram per region (Figure 1), showed epidemic peaks in the four years observed for the Northern region, in particular the first semester of 2016. The Northeast region showed a level above the endemic channel with effect from April 2015, with a peak in July of that year, and also in the first semester of 2016 and in April 2017. In the Southeast region, GBS hospitalizations occurred on an epidemic level between September 2015 and June 2016, and also in the first three months of 2017. In the Southern region, hospitalizations remained at an endemic level throughout the period. In the Midwest region GBS hospitalizations were at an epidemic level between December 2015 and April 2016.

Figure 1 – Guillain-Barré syndrome hospitalization rate control diagram by region of residence, Brazil, 2014-2017
The spatio-temporal analysis showed a gradual increase in GBS hospitalizations with effect from 2015, with 28.4 hospitalizations recorded per 100,000 inhab. In 2015, the states of Roraima and Amapá stood out in the Northern region and the states of Rio Grande do Norte, Pernambuco and Alagoas stood out in the Northeast region (Figure 2). There was a shift in 2016 to the Midwest and Southeast regions.
Discussion
There was an increase in the number of Guillain-Barré syndrome hospitalizations in Brazil with effect from 2015. GBS hospitalizations occurred on an epidemic level in the Northeast region in the first semesters of 2015 and 2016 and with less intensity in the first semester of 2017. A similar situation was found in the Southeast, Midwest and Northern regions.
Among GBS hospitalizations in the period studied, there was a predominance of male cases, length of hospital stay of around a week and a lower percentage submitted to intensive care. The need for ITU monitoring is one of the indicators of severity, and the proportion found in this study was similar to that found in other studies.2
The lethality rate found for Brazil as a whole over the period was similar to or even below that found in North America and Europe.8 It is possible that timely use of intravenous human immunoglobulin in Brazil available on the SUS was reflected in prognosis.7
Zika virus was identified for the first time in Brazil in April 2015, in the Northeast region, where the initial cases of the epidemic occurred. In the following year, the epidemic shifted to the Southeast, Midwest and Southern regions.15 The spatio-temporal analysis of GBS hospitalizations demonstrated an increase in the incidence rate with effect from 2015; and clustering of the syndrome in the states of the Northeast region, with later shifts to other regions, coinciding with the epidemiological situation of acute Zika virus disease.
Although GBS is not seasonal, an increase in hospitalizations on an epidemic level was frequently found in the first semester of the year. It should be noted that this is the seasonal period of arbovirus infections.16 Epidemiological investigations carried out by the Training Program on Epidemiology Applied to SUS Services (EpiSUS-Avançado/SVS/MS) in the state of Bahia, identifier a greater number of GBS cases after the introduction of Zika virus, demonstrating temporal association: the majority of patients interviewed reported symptoms suggestive of acute Zika virus fever six weeks before the onset of the neurological disease.15
Some studies have reported increases in GBS cases associated with Zika virus, such as in French Polynesia, where cases presented IgM or IgG markers and Zika virus neutralizing antibodies.18 A study conducted in Puerto Rico timely identified GBS cases associated with Zika virus infection confirmed by RT-PCR.19
GBS has also been found to be associated with chikungunya fever. Another study conducted in French Polynesia between 2014 and 2015, found an increase in the syndrome following a chikungunya outbreak.20 GBS cases following chikungunya infection were reported on Reunion Island, where syndrome incidence increase was estimated at 22%.21 A study conducted in Martinique and in Guadeloupe in 2014 indicated increased incidence during the outbreak period.22
The data used in this study were aggregated. This therefore does not mean that the same findings may have happened on the individual level. The information was obtained from the population group as a whole. This study used secondary data, and the GBS hospitalization rate was calculated using SIH/SUS data, an information system which in a nutshell has the purpose of compiling records for payment for health services provided to SUS patients during hospitalization. In other words, the study did not include private sector or supplementary sector hospitalizations. Nor were possible duplications excluded, given that the study used a non-nominal database.
This study describes a panorama of GBS hospitalizations in Brazil, in the country’s different regions, over a ten-year period. A notable increase in Guillain-Barré syndrome hospitalizations occurred with effect from 2015, coinciding with the introduction of chikungunya virus and with the identification – and rapid spread – of Zika virus. We recommend that Public Health authorities take into consideration and put on the agenda surveillance of neuroinvasive diseases caused by arboviruses, given the magnitude of the syndrome, the potential for transmission by its vector and, consequently, dissemination of the virus and its irreparable consequences for the population such as death, sequelae and acquired disabilities.
REFERENCES
1. Freitas MRG, Vidal CM, Orsini M. Guillain-Barré syndrome: celebrating a century. Arq Neuro-Psiquiatr [Internet]. 2017 Aug [cited 2020 Jun 2];75(8):600-3. Available from: https://doi.org/10.1590/0004-282x20170093 [ Links ]
2. Van den Berg B, Walgaard C, Drenthen J, Fokke C, Jacobs BC, Van Doorn PA. Guillain-Barre syndrome: pathogenesis, diagnosis, treatment and prognosis. Nat Rev Neurol [Internet]. 2014 Jul [cited 2020 Jun 2];10(8):469-82. Available from: https://doi.org/10.1038/nrneurol.2014.121 [ Links ]
3. Sejvar JJ, Kohl KS, Gidudu J, Amato A, Bakshi N, Baxter R, et al. Guillain-Barré syndrome and Fisher syndrome: case definitions and guidelines for collection, analysis, and presentation of immunization safety data. Vaccine [Internet]. 2011 Jan [cited 2020 Jun 2];29(3):599-612. Available from: https://doi.org/10.1016/j.vaccine.2010.06.003 [ Links ]
4. Souza CO, Vieira MACS, Batista FMA, Eulálio KD, Neves JMM, Sá LC, et al. Marcadores sorológicos da infecção recente por Campylobacter jejuni em pacientes com síndrome de Guillain-Barré no estado do Piauí, Brasil, 2014-2016. Am J Trop Med Hyg [Internet]. 2018 fev [citado 2020 jun 2];98(2):586-8. Disponível em: https://doi.org/10.4269/ajtmh.17-0666 [ Links ]
5. Willison HJ, Jacobs BC, van Doorn PA. Guillain-Barre syndrome. Lancet [Internet]. 2016 Feb [cited 2020 Jun 2];388(10045):P717-27. Available from: https://doi.org/10.1016/S0140-6736(16)00339-1 [ Links ]
6. Blazquez AB, Saiz JC. Neurological manifestations of Zika virus infection. World J Virol [Internet]. 2016 Nov [cited 2020 Jun 2];5(4):135-43. Available from: https://dx.doi.org/10.5501%2Fwjv.v5.i4.135 [ Links ]
7. Ministério da Saúde (BR). Portaria MS/SAS nº 1171, de 19 de novembro de 2015. Aprova o protocolo clínico e diretrizes terapêuticas síndrome de Guillain-Barré [Internet]. Diário Oficial da União; Brasília (DF); 2015 jov 19 [citado 2020 jun 2];Seção 1:83. Disponível em: https://bvsms.saude.gov.br/bvs/saudelegis/sas/2015/prt1171_19_11_2015.html [ Links ]
8. Sejvar JJ, Baughman AL, Wise M, Morgan OW. Population incidence of Guillain-Barré syndrome: a systematic review and meta-analysis. Neuroepidemiology [Internet]. 2011 [cited 2020 Jun 2];36(2):123-33. Available from: https://doi.org/10.1159/000324710 [ Links ]
9. Dourado ME, Félix RH, Silva WK, Queiroz JW, Jeronimo SM. Clinical characteristics of Guillain-Barré syndrome in a tropical country: a Brazilian experience. Acta Neurol Scand [Internet]. 2012 Jan [cited 2020 Jun 2];125(1):47-53. Available from: https://doi.org/10.1111/j.1600-0404.2011.01503.x [ Links ]
10. Rocha MSG, Brucki SMD, Carvalho AAS, Lima UWP. Epidemiologic features of Guillain-Barré syndrome in São Paulo, Brazil. Arq Neuro-Psiquiatr [Internet]. 2004 Mar [cited 2020 Jun 2];62(1):33-7. Available from: https://doi.org/10.1590/S0004-282X2004000100006 [ Links ]
11. Leite PL. Síndrome de Guillan-Barré e manifestações neurológicas relacionadas a doença exantemática anterior. Apresentação realizada para o Comitê Técnico Assessor do Programa Nacional de Controle da Dengue do Ministério da Saúde, Brasília. (CTA-PNCD/MS). 2015 set 30. [ Links ]
12. Instituto Brasileiro de Geografia e Estatística - IBGE. Estimativas de população para 1º de julho de 2015 [Internet]. Rio de Janeiro: Instituto Brasileiro de Geografia e Estatística; 2016 [citado 2020 jun 2]. Disponível em: http://www.ibge.gov.br/home/estatistica/populacao/estimativa2015/estimativa_tcu.shtm [ Links ]
13. Ministério da Saúde (BR). Secretaria de Atenção à Saúde. Departamento de Regulação, Avaliação e Controle. Coordenação Geral de Sistemas de Informação. SIH – Sistema de Informação Hospitalar do SUS: manual técnico operacional do sistema [Internet]. Brasília: Ministério da Saúde; 2017 [citado 2020 jun 2]. 103 p. Disponível em: http://www.saude.sp.gov.br/resources/ses/perfil/gestor/homepage/auditoria/manuais/manual_sih_janeiro_2017.pdf [ Links ]
14. Rouquayrol MZ, Gurgel M. Rouquaryol: epidemiologia & saúde. 8. ed. Rio de Janeiro: Medbook; 2018. 752 p. [ Links ]
15. Malta JM, Vargas A, Leite PL, Percio J, Coelho GE, Ferraro AHA, et al. Síndrome de Guillain-Barré e outras manifestações neurológicas possivelmente relacionadas à infecção pelo ZIKV em municípios da Bahia, 2015. Epidemiol Serv Saúde [Internet]. 2017 ajn-mar [citado 2020 jun 2];26(1):9-18. Disponível em: http://dx.doi.org/10.5123/s1679-49742017000100002 [ Links ]
16. Braga IA, Valle D. Aedes aegypti: histórico do controle no Brasil. Epidemiol Serv Saúde [Internet]. 2007 jun [citado 2020 jun 2];16(2):113-8. Disponível em: http://dx.doi.org/10.5123/S1679-49742007000200006 [ Links ]
17. Styczynski AR, Malta JMAS, Krow-Lucal ER, Percio J, Nóbrega ME, Vargas A, et al. Increased rates of Guillain-Barré syndrome associated with Zika virus outbreak in the Salvador metropolitan area, Brazil. PLoS Negl Trop Dis [Internet]. 2017 Aug [cited 2020 Jun 2];11(8):e0005869. Available from: https://doi.org/10.1371/journal.pntd.0005869 [ Links ]
18. Cao-Lormeau VM, Blake A, Mons S, Lastere S, Roche C, Vanhomwegen J, et al. Guillain-Barre syndrome outbreak associated with Zika virus infection in French Polynesia: a case-control study. Lancet [Internet]. 2016 Apr [cited 2020 jun 2];387(10027);1531-9. Available from: https://doi.org/10.1016/s0140-6736(16)00562-6 [ Links ]
19. Dirlikov E, Major CG, Mayshack M, Medina N, Matos D, Ryff KR, et al. Guillain-Barre syndrome during ongoing Zika virus transmission—Puerto Rico, January 1-July 31, 2016. MMWR Morb Mortal Wkly Rep [Internet]. 2016 Sep [cited 2020 Jun 2];65(34):910-4. Available from: https://doi.org/10.15585/mmwr.mm6534e1 [ Links ]
20. Oehler E, Fournier E, Leparc-Goffart I, Larre P, Cubizolle S, Sookhareea C, et al. Aumento dos casos de síndrome de Guillain-Barré durante um surto de chikungunya, Polinésia Francesa, 2014 a 2015. Euro Surveill [Internet]. 2015 dez [citado 2020 jun 2];20(48):30079. Disponível em: https://doi.org/10.2807/1560-7917.ES.2015.20.48.30079 [ Links ]
21. Lebrun G, Chadda K, Reboux AH, Martinet O, Gauzere BA. Síndrome de Guillain-Barre após infecção por chikungunya. Emerg Infect Dis [Internet]. 2009 mar [citado 2020 jun 2];15(3):495-6. Disponível em: https://dx.doi.org/10.3201/eid1503.071482 [ Links ]
22. Balavoine S, Pircher M, Hoen B, Herrmann-Stock C, Najioullah F, Madeux B, et al. Síndrome de Guillain-Barre e chikungunya: descrição de todos os casos diagnosticados durante o surto de 2014 nas Antilhas Francesas. Am J Trop Med Hyg [Internet]. 2017 ago [citado 2020 jun 2];97(2):356-60. Disponível em: https://doi.org/10.4269/ajtmh.15-0753 [ Links ]
Received: April 09, 2020; Accepted: May 13, 2020











 texto em
texto em