Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Epidemiologia e Serviços de Saúde
Print version ISSN 1679-4974On-line version ISSN 2237-9622
Epidemiol. Serv. Saúde vol.29 no.4 Brasília 2020 Epub July 23, 2020
http://dx.doi.org/10.5123/s1679-49742020000400003
Review article
International collaboration networks for the surveillance of congenital anomalies: a narrative review
1Ministério da Saúde, Secretaria de Vigilância em Saúde, Brasília, DF, Brazil
Objective
to identify international congenital anomaly surveillance collaboration networks, to list the programs that compose them and to compare their main characteristics.
Methods
this was a narrative literature review by means of a MEDLINE database search (via PubMed) and searches involving websites, reports and official documents.
Results
six international congenital anomaly surveillance collaboration networks were identified (ECLAMC, ICBDSR, EUROCAT, BINOCAR, SEAR-NBBD and ReLAMC), comprised of 98 programs present in 58 different countries on all continents, except Africa; the main characteristics regarding type of surveillance, coverage and location were discussed in a comparative manner.
Conclusion
international collaborative networks are important players for congenital anomaly surveillance, contributing to the understanding of the global epidemiological scenario of these conditions, in addition to acting both to strengthen individual existing programs and also to create surveillance initiatives in unassisted regions.
Key words: Congenital Abnormalities; Epidemiological Monitoring; Review; International Cooperation; Health Services; Epidemiology
Introduction
Congenital anomalies (CAs), congenital defects or congenital malformations are structural or functional changes that occur during life in the womb, detected during pregnancy, at childbirth or at some time later in life. According to the World Health Organization (OMS), every year some 300,000 newborn babies die from CAs globally. Apart from premature death, CAs can cause serious disabilities that have great impact on those affected, their families, health systems and society.1
Some congenital defects can be avoided through prevention measures, such as vaccination, dietary supplements and prenatal care; moreover, the burden associated with many of these anomalies can be reduced, especially when diagnosis occurs early. Geographic and temporal variations also exist and are important with regard to frequency of certain congenital defects, thus making knowledge about the epidemiological scenario of CAs valuable information for strategic planning of Public Health actions.1
Ever since the thalidomide tragedy in the 1960s, governments and institutions have invested efforts and resources in designing CAs surveillance programs with the primary objective of monitoring and investigating these changes, so as to prevent them or to reduce the impact of their consequences.3 CAs surveillance models can currently be classified into two main types: population-based, which investigate births with CAs among the population resident in a delimited geographic area; or hospital-based, which investigate births with CAs in selected hospitals, maternity hospitals or facilities and the coverage of which corresponds to births in those places. Within hospital-based surveillance, there is also sentinel surveillance, generally operating in specific places with the aim of quickly obtaining estimates of the occurrence of a pregnancy outcome. With regard to coverage, specific programs can promote CAs surveillance in a restricted area within a country (subnational coverage), in an entire country (national) or even encompass several countries (multinational).2
Also in the 1960s, it was perceived that organization of CAs surveillance programs into international collaboration networks could help in overcoming certain common issues reducing the frequency of some CAs cases; expansion of epidemiological findings to other broader areas; communication between programs; and standardization of definitions and methods for identifying CAs, making it possible to compare estimates of magnitude and evaluate prevention measures in different places.3
Currently there are both consolidated networks and recently created networks, with different characteristics, purposes and locations in diverse regions, so that the need exists to systematize this information.
The main objectives of this study were to identify international congenital anomaly surveillance collaboration networks around the world, to list the programs that comprise them and to compare their main characteristics.
Methods
This is a narrative literature review, comprised of two main stages: (i) review of specialized scientific literature by means of searching the MEDLINE database (via PubMed); and (ii) review of websites, online reports and official documents on the subject of congenital anomalies.
The MEDLINE database search was performed on January 10th 2020, using the following electronic search strategy:
((((((congenital abnormalities[MeSH Major Topic]) OR congenital abnormalities[MeSH Subheading]) OR birth defect[Title/Abstract])) OR microcephaly)) AND ((((Population Surveillance[MeSH Major Topic]) OR Public Health Surveillance[MeSH Major Topic]) OR Surveillance[Title/Abstract]) OR Epidemiological Monitoring[MeSH Major Topic])
Papers were included with reports of at least one international CAs surveillance collaboration network. No restriction was made as to language, year of publication or study design. Papers were excluded that (i) made no mention of congenital anomaly surveillance, (ii) did not have an abstract, (iii) addressed surveillance of just one specific defect or outcome, or (iv) that dealt with just one individual surveillance program that only had national or subnational coverage.
The electronic search resulted in 3024 records. After screening titles and abstracts and applying the eligibility criteria, 113 articles were selected for complete text reading (Figure 1). The final articles and surveillance networks selected from them were analyzed by two authors in an independent and randomized manner.
An illustrative thematic map, showing the geographic distribution of the congenital defects monitoring projects covered by the international networks found in this review was generated using Quantum GIS (QGIS) version 3.4.14. Cytoscape version 3.7.2 was used to generate an interaction network between those programs.
Results
Based on the articles considered to be eligible and on information derived from other sources, six international congenital anomaly surveillance collaboration networks were identified: ECLAMC, ICBDSR, EUROCAT, BINOCAR, SEAR-NBBD and ReLAMC. The meaning of these acronyms is shown in Table 1, as well as the main characteristics of each network.
Table 1 – Main characteristics of the international congenital anomaly surveillance collaboration networks
| Network | Description | Year created | Number of programs | Program coverage | Number of countries | Location |
|---|---|---|---|---|---|---|
| Estudio Colaborativo Latino Americano de Malformaciones Congénitas (ECLAMC) | Program for epidemiological and clinical investigation of congenital anomalies in Latin American hospitals | 1967 | – | – | 12 | Latin America |
| International Clearinghouse for Birth Defects Surveillance and Research (ICBDSR) | Voluntary non-profit organization, aiming to prevent birth defects around the world and reduce their consequences | 1974 | 45 | National: 18 Subnational: 26 Multinational: 1a | 30 (plus Latin America, via ECLAMC) | All continents except Africa |
| European Surveillance of Congenital Anomalies (EUROCAT) | European population-based registry for epidemiological surveillance of congenital anomalies | 1979 | 42b | National: 9 Subnational: 33 | 23 | Europe |
| British and Irish Network of Congenital Anomaly Researchers (BINOCAR) | Group of researchers working with congenital anomalies records for the population of the British Isles | 1985 | 13 | National: 2 Subnational: 11c | 4 | Europe (England, Wales, Scotland and Ireland) |
| Red Latinoamericana de Malformaciones Congénitas (ReLAMC) | National and subnational registry network (apart from ECLAMC) that conducts voluntary surveillance of congenital anomalies in Latin America | 2016 | 10 | National: 5 Subnational: 4 Multinational: 1a | 8 | Latin America |
| South-East Asia Region’s Newborn and Birth Defects Database (SEAR-NBBD) | Online system for collecting perinatal surveillance data with the aim of preventing and improving the health of children with congenital anomalies | 2014 | – | – | 9 | Asia (South-East Asia region) |
a) Includes ECLAMC.
b) This count includes 36 full members and 6 associate members that are part of EUROCAT.
c) With effect from 2015, records of congenital anomalies in England have been incorporated into its National Congenital Anomaly and Rare Disease Registration Service (NCARDRS).
Table 2 provides details of the CAs surveillance programs covered by the networks. We identified 98 programs present in 58 different countries that form part of one or more of the networks identified in this review. Twenty-two programs (22.4%) had national coverage, while only one program (1.0%) had multinational (coverage ECLAMC); the remaining 75 programs had subnational coverage. Among those with national coverage, the majority were located on the European continent (n=11, 50.0%), followed by Central America (n=4, 18.2%), Asia, South America and North America (n=2, 9.1% each) and Oceania (n=1, 4.5%). No records were found for the African continent in any of the networks identified (Figure 2).
Table 2 – Congenital anomaly monitoring programs covered by the international networks
| Country | International Network | Program | Coverage | Surveillance type |
|---|---|---|---|---|
| Argentina | ICBDSR and ReLAMC | National Network of Congenital Anomalies of Argentina (RENAC) | National | Hospital-based |
| ECLAMC | – | Subnational | Hospital-based | |
| Australia | ICBDSR | Western Australian Register of Developmental Anomalies (WARDA) | Subnational (Western Australia) | Population-based |
| Austria | EUROCAT | Styrian Malformation Registry | Subnational (Styria) | Population-based |
| Bangladesh | SEAR-NBBD | – | Subnational (20 hospitals) | Hospital-based |
| Belgium | EUROCAT | – | Subnational (Antwerp) | Population-based |
| EUROCAT | – | Subnational (Hainaut-Namur) | Population-based | |
| Bhutan | SEAR-NBBD | – | Subnational (7 hospitals) | Hospital-based |
| Bolivia | ECLAMC | – | Subnational | Hospital-based |
| Brazil | ECLAMC | – | Subnational | Hospital-based |
| Bulgaria | EUROCAT | – | Subnational (Pleven) | Hospital-based |
| Canada | ICBDSR | Alberta Congenital Anomalies Surveillance System (ACASS) | Subnational (Alberta) | Population-based |
| ICBDSR | Canadian Congenital Anomalies Surveillance Network (CCASS) | National | Population-based | |
| Chile | ICBDSR and ReLAMC | Regional Register Congenital Malformational Maule Health Service (RRMC-SSM) | Subnational (Maule) | Hospital-based |
| ECLAMC | – | Subnational | Hospital-based | |
| Colombia | ICBDSR and ReLAMC | Congenital Malformations Surveillance Programme (CMSP) | Subnational (Bogota) | Hospital-based |
| ICBDSR and ReLAMC | Congenital Birth Defects Surveillance Programme (CBDSP) | Subnational (Cali) | Hospital-based | |
| ECLAMC | – | Subnational | Hospital-based | |
| Costa Rica | ECLAMC | – | Subnational | Hospital-based |
| ICBDSR and ReLAMC | Costa Rican Birth Defects Register Center (CREC) | National | Population-based | |
| Croatia | EUROCAT | – | Subnational (Zagreb) | Population-based |
| Cuba | ICBDSR | Cuban Register of Congenital Malformation (RECUMAC) | National | Hospital-based |
| Czech Republic | ICBDSR and EUROCAT | Czech Republic National Registry of Congenital Anomalies | National | Population-based |
| Denmark | EUROCAT | – | Subnational (Odense) | Population-based |
| Dominican Republic | ECLAMC | – | Subnational | Hospital-based |
| Ecuador | ECLAMC | – | Subnational | Hospital-based |
| England | ICBDSR and BINOCAR | National Congenital Anomaly and Rare Disease Registration Service (NCARDRS) | National | – |
| EUROCAT and BINOCAR | East Midlands and South Yorkshire Congenital Anomalies Register (EM&SY CAR) | Subnational (East Midlands & South Yorkshire) | Population-based | |
| EUROCAT and BINOCAR | Northern Congenital Abnormality Survey (NorCAS) | Subnational (Northern England) | Population-based | |
| EUROCAT and BINOCAR | South West Congenital Anomaly Register (SWCAR) | Subnational (Southeast England) | Population-based | |
| EUROCAT and BINOCAR | The Congenital Anomaly Register of Oxfordshire, Berkshire and Buckinghamshire (CAROBB) | Subnational (Thames Valley) | Population-based | |
| EUROCAT and BINOCAR | The Wessex Antenatally Detected Anomalies Register (WANDA) | Subnational (Wessex) | Population-based | |
| BINOCAR | West Midlands Congenital Anomaly Register (WMCAR) | Subnational (West Midlands) | Population-based | |
| BINOCAR | Yorkshire and the Humber Congenital Anomalies Register (YHCAR) | Subnational (Yorkshire & Humber) | Population-based | |
| Finland | ICBDSR and EUROCAT | Finnish Register of Congenital Malformations | National | Population-based |
| France | EUROCAT | Centre d’Etude des Malformations Congénitales en Auvergne | Subnational (Auvergne) | Population-based |
| ICBDSR and EUROCAT | Registre des Malformations Congénitales de Bretagne | Subnational (Brittany region) | Population-based | |
| EUROCAT | Registre des Malformations des Antilles | Subnational (French West Indies) | – | |
| France | EUROCAT | Registre des Malformations Congénitales de la Reunion | Subnational (Reunion Island) | Population-based |
| ICBDSR and EUROCAT | Paris Registry of Congenital Malformations | Subnational (Paris) | Population-based | |
| ICBDSR and EUROCAT | REMERA | Subnational (Rhône-Alpes) | Population-based | |
| Germany | EUROCAT | Birth Registry Mainz Model | Subnational (Mainz) | Population-based |
| ICBDSR and EUROCAT | Malformation Monitoring Saxony-Anhalt | Subnational (Sax-Anh) | Population-based | |
| Hungary | ICBDSR and EUROCAT | Hungarian Congenital Abnormality Registry (HCAR) | National | Population-based |
| India | SEAR-NBBD | – | Subnational (76 hospitals) | Hospital-based |
| ICBDSR | Birth Defects Registry of India (BDRI) | Subnational (Chennai) | Hospital-based | |
| Indonesia | SEAR-NBBD | – | Subnational (34 hospitals) | Hospital-based |
| Iran | ICBDSR | Tabriz Registry of Congenital Anomalies (TROCA) | Subnational (Tabriz) | Hospital-based |
| Ireland | EUROCAT and BINOCAR | Cork and Kerry Congenital Anomaly Register | Subnational (Cork and Kerry) | Population-based |
| EUROCAT and BINOCAR | South East Congenital Anomaly Register | Subnational (Southeast Ireland) | Population-based | |
| EUROCAT and BINOCAR | Dublin Congenital Anomaly Register | Subnational (Dublin) | Population-based | |
| Israel | ICBDSR | Israel Birth Defects Surveillance Program (IBDSP) | Subnational (Central District of Israel) | Hospital-based |
| Italy | EUROCAT | Congenital Anomalies Registry for the Metropolitan Area of Milan | Subnational (Metropolitan Area of Milan) | Population-based |
| ICBDSR | Birth Defects Registry of Campania (BDRCam) | Subnational (Campania) | Population-based | |
| ICBDSR and EUROCAT | Emilia-Romagna Registry of Congenital Malformations (IMER) | Subnational (Emilia-Romagna) | Population-based | |
| ICBDSR | Register of Congenital Anomalies of Veneto Region | Subnational (Veneto) | – | |
| ICBDSR and EUROCAT | Tuscany Registry of Congenital Defects (RTDC) | Subnational (Tuscany) | Population-based | |
| ICBDSR | Congenital Malformation Registry of Northern Lombardy (RMCL) | Subnational (Lombardy) | Population-based | |
| Japan | ICBDSR | Japan Association of Obstetricians and Gynaecologists (JAOG) | National | Hospital-based |
| Latin America | ICBDSR and ReLAMC | Latin American Collaborative Study of Congenital Malformations (ECLAMC) | Multinational | Hospital-based |
| Maldives | SEAR-NBBD | – | Subnational (12 hospitals) | Hospital-based |
| Malta | ICBDSR and EUROCAT | Malta Congenital Anomalies Registry (MCAR) | National | Population-based |
| Mexico | ICBDSR | Mexican Registry and Epidemiological Surveillance of External Congenital Malformations (RYVEMCE) | National | Hospital-based |
| ICBDSR | Birth Defects Prevention and Surveillance Programme (BDPSP) | Subnational (Nuevo León) | Population-based | |
| ICBDSR and ReLAMC | Registro de Defectos Congénitos Hospital Universitario UANL (ReDeCon HU) | Subnational (Nuevo León) | – | |
| Myanmar | SEAR-NBBD | – | Subnational (14 hospitals) | Hospital-based |
| Nepal | SEAR-NBBD | – | Subnational (5 hospitals) | Hospital-based |
| Netherlands | ICBDSR and EUROCAT | EUROCAT Registration Northern Netherlands | Subnational (North Holland) | Population-based |
| New Zealand | ICBDSR | New Zealand Birth Defects Monitoring Programme | National | Population-based |
| Nicaragua | ReLAMC | Sistema de Vigilancia de Defectos Congénitos (SVDC UNAN) | National | – |
| Norway | ICBDSR and EUROCAT | Medical Birth Registry of Norway (MBRN) | National | Population-based |
| Panama | ReLAMC | Programa Nacional de Malformaciones Congénitas de Panama (PNMC) | National | – |
| Paraguay | ReLAMC | Programa Nacional de Prevención de Defectos Congénitos del Ministerio de Salud Pública del Paraguay (PNPDC) | National | – |
| ECLAMC | – | Subnational | Hospital-based | |
| Peru | ECLAMC | – | Subnational | Hospital-based |
| Poland | EUROCAT | – | Subnational (Wielkopolska) | Population-based |
| EUROCAT | Polish Registry of Congenital Malformations (PRCM) | National | Population-based | |
| Portugal | EUROCAT | – | Subnational (Southern Portugal) | Population-based |
| Saudi Arabia | ICBDSR | Medical Service Department- Birth Defect Registry (MSD-BDR) | National | Hospital-based |
| Scotland | BINOCAR | Glasgow Register of Congenital Anomalies (Glasgow) | Subnational (Glasgow) | Population-based |
| Slovakia | ICBDSR | Programme of Slovak Teratological Information Center (STIC) | National | Population-based |
| Spain | ICBDSR and EUROCAT | Spanish Collaborative Study of Congenital Malformations (ECEMC) | National | Hospital-based |
| EUROCAT | Registro Anomalías Congénitas CAPV (RACAV) | Subnational (Basque Country) | Population-based | |
| EUROCAT | Congenital Anomalies Registry of Navarra (RACEHNA) | Subnational (Navarra) | Population-based | |
| EUROCAT | Congenital Anomalies Registry of Comunitat Valenciana | Subnational (Valencia Region) | Population-based | |
| Sri Lanka | SEAR-NBBD | – | Subnational (81 hospitals) | Hospital-based |
| Sweden | ICBDSR and EUROCAT | Swedish Register of Congenital Malformations | National | Population-based |
| Switzerland | EUROCAT | Registry of Vaud (Switzerland) | Subnational (Vaud) | Population-based |
| Thailand | SEAR-NBBD | – | Subnational (929 hospitals) | Hospital-based |
| Ukraine | ICBDSR and EUROCAT | OMNI-Net Ukraine Birth Defects Program (OMNI-Net) | Subnational (Northeast Ukraine) | Population-based |
| Uruguay | ECLAMC | – | Subnational | Hospital-based |
| USA | ICBDSR | Arkansas Reproductive Health Monitoring System (ARHMS) | Subnational (Arkansas) | Population-based |
| ICBDSR | Metropolitan Atlanta Congenital Defects Program (MACDP) | Subnational (Atlanta) | Population-based | |
| ICBDSR | Iowa Registry for Congenital and Inherited Disorders (IRCID) | Subnational (Iowa) | Population-based | |
| ICBDSR | Texas Birth Defects Epidemiology and Surveillance Branch (BDES) | Subnational (Texas) | Population-based | |
| ICBDSR | Utah Birth Defects Network (UBDN) | Subnational (Utah) | Population-based | |
| Venezuela | ECLAMC | – | Subnational | Hospital-based |
| Wales | ICBDSR, EUROCAT and BINOCAR | Congenital Anomaly Register and Information Service (CARIS) | National | Population-based |
Legend:
BINOCAR: British and Irish Network of Congenital Anomaly Researchers.
ECLAMC: Estudio Colaborativo Latino Americano de Malformaciones Congénitas.
EUROCAT: European Surveillance of Congenital Anomalies.
ICBDSR: International Clearinghouse for Birth Defects Surveillance and Research.
ReLAMC: Red Latinoamericana de Malformaciones Congénitas.
SEAR-NBBD: South-East Asia Region’s - Newborn and Birth Defects Database.
Notes:
With regard to the ECLAMC and SEAR-NBBD hospital networks, each participating country was counted as one surveillance program.
ECLAMC is an international network, and is also part of ICBDSR and ReLAMC.
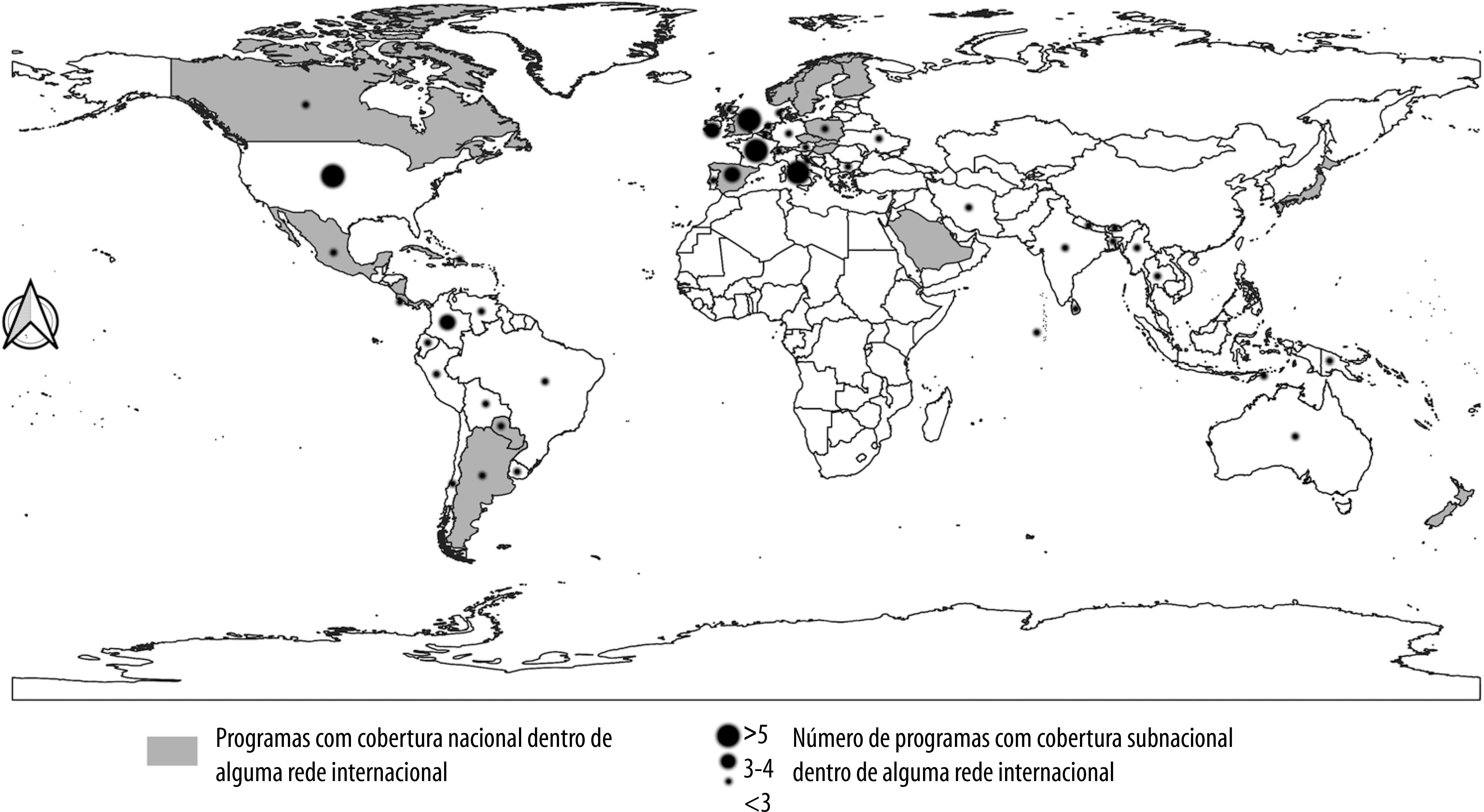
Figure 2 – Countries that are part of international networks through programs with national, subnational or both coverage
Figure 3 shows an interaction diagram between the programs that form part of the international CAs surveillance collaboration networks. All these networks, except for the collaboration network between the South-East Asian countries (SEAR-NBBD), had programs that were also part of other networks. One of them, ECLAMC, despite being an international collaboration network itself, collaborates with two other international surveillance systems, namely ReLAMC and ICBDSR. Detailed information on each network is presented below.
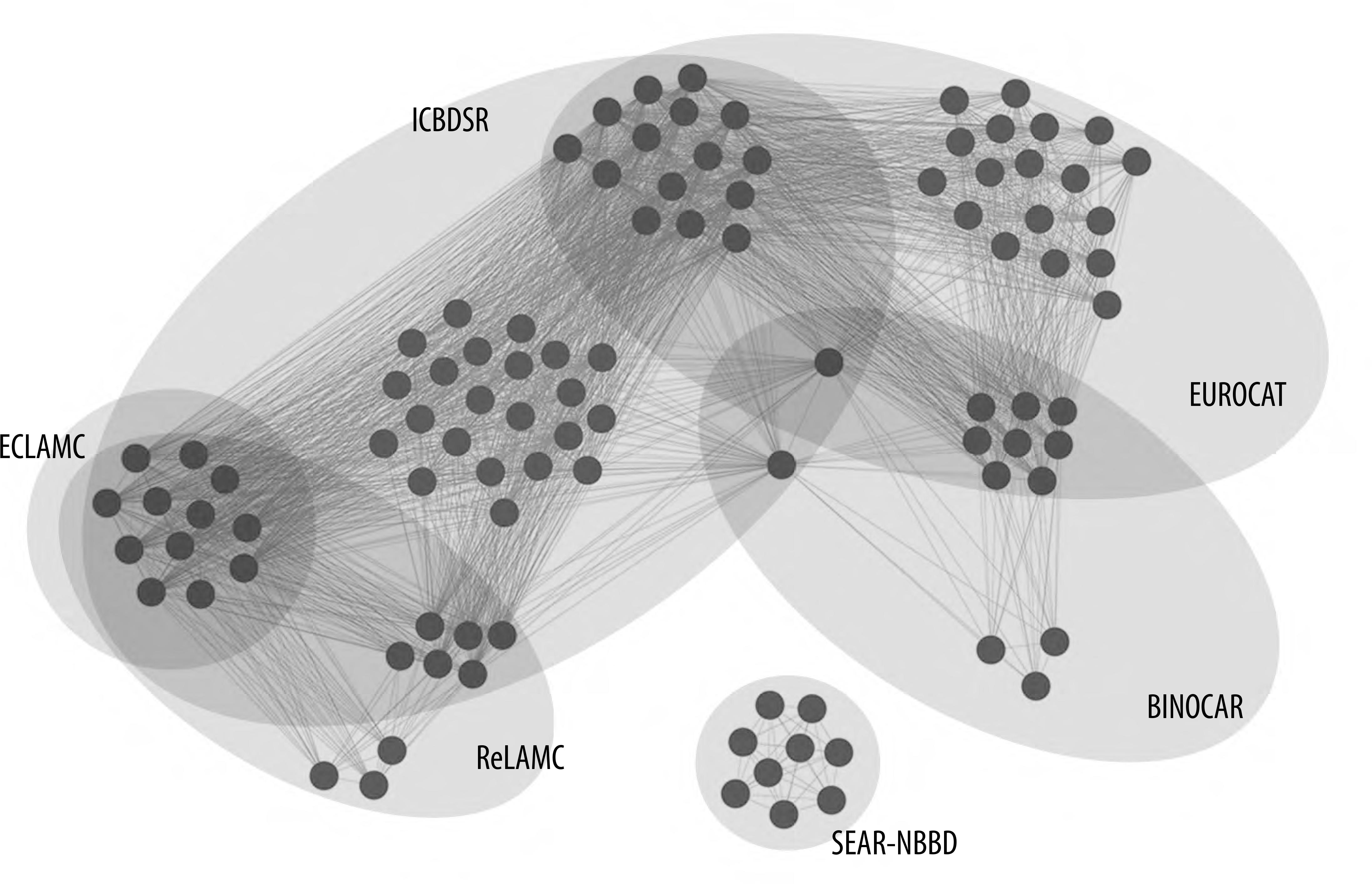
Figure 3 – Network of networks: number and distribution of programs that are part of international networks of congenital anomaly índicesNotes:Each sphere represents a monitoring program for congenital malformations contained in some network international, represented by shaded ellipses.For ECLAMC and SEAR-NBBD, each sphere represents a country participating in the hospital network.
Estudio Colaborativo Latino Americano de Malformaciones Congénitas (ECLAMC)
Created in 1967 as a CAs investigation program restricted to the Argentinean capital Buenos Aires, ECLAMC expanded its activities to other South American countries in 1969. It is a program that undertakes clinical and epidemiological investigation of CAs risk factors in Latin American hospitals, using a case-control methods, i.e.: each newborn baby with CAs (case) is matched with the next newborn baby with no malformations, of the same sex and in the same hospital (control). The main objective of this program is to prevent CAs through research.4 ECLAMC is a voluntary and collaborative network formed of Latin American hospital personnel who wish to take part in the study and, after more than fifty years of existence, it has hundreds of participating hospitals in 12 Latin American countries.8 In addition to dozens of academic papers published in a wide variety of areas related to defects, the Latin American strategy provides primary and tertiary prevention manuals. For health professionals who work with CAs description, coding and/or analysis, the program has also created a Congenital Malformations Browser, an online tool that links the name of the CAs or the code contained in the International Statistical Classification of Diseases and Related Health Problems – 10th Revision (ICD-10) with images of the most common malformations.9
International Clearinghouse for Birth Defects Surveillance and Research (ICBDSR)
ICBDSR (or just Clearinghouse) is a voluntary non-profit international network linked to WHO. Created in 1974, the Clearinghouse currently comprises 45 congenital anomaly surveillance programs with different characteristics that are present on almost all continents (except Africa). These comprise 18 programs with national coverage, 26 programs with subnational coverage and 1 with multinational coverage, i.e. ECLAMC. Jointly, ICBDSR monitors approximately 4 million births a year. The main mission of this organization is to promote and conduct CAs surveillance around the world, as well as scientific research on causes and determinants, with the objective of preventing congenital defects.3 The network also provides educational resources on CAs prevention, surveillance and research.2
European Surveillance of Congenital Anomalies(EUROCAT)
EUROCAT is a network of population-based data registries, created in 1979 with the main objectives of collecting population data and carrying out epidemiological surveillance of CAs on the European continent. The acronym EUROCAT derives from its original name: European Concerted Action on Congenital Anomalies and Twins. In January 2020, this network comprised 42 registries, between full and associated members, covering over 1.7 million births in 23 different countries (for the purposes of this study, the nations comprising the United Kingdom were counted as separate countries), which accounts for approximately 29% of the annual population of births in Europe.10 Besides providing epidemiological information about CAs, EUROCAT aims to work as an information and research center for the population, health workers and managers working with the subject of congenital malformations, teratogenic agents and other risk factors. Another objective of the network is to assess the effectiveness of primary prevention measures and the impact of prenatal screening programs among the European population.10
British and Irish Network of Congenital Anomaly Researchers (BINOCAR)
BINOCAR comprises a network of CAs registries spread over England, Scotland, Wales and Ireland that began its activities in 1985. Despite having undergone a series of changes over its history, the network has maintained its principal objective: to provide resources and robust information about the epidemiology, causes and outcomes of congenital defects among the populations covered.12 BINOCAR comprises three subnational programs for congenital anomaly surveillance in Ireland, another subnational program in Scotland, a national registry in Wales and a further seven subnational registries in England – which since 2015 have been incorporated into the new National Congenital Anomaly and Rare Disease Registration Service (NCARDRS).13 Apart from the registries mentioned above, the network has two registries focused on specific outcomes: the National Down Syndrome Cytogenetic Register, in England and Wales; and the Cleft Registry and Audit Network, in England, Wales and Northern Ireland.
South-East Asia Region’s Newborn and Birth Defects Database (SEAR-NBBD)
This network was created in 2014, fruit of joint efforts between the World Health Organization South-East Asia Region Office (WHO SEARO) and the United States Centers for Disease Control and Prevention (US CDC). The purpose of this surveillance system is to collect perinatal surveillance data in order to establish robust assessment of neonatal health and monitor the occurrence of CAs in South-East Asia. The main objective of the network is to promote primary prevention and improve the health of children with CAs.14 currently, SEAR-NBBD promotes hospital-based surveillance in nine Asian countries which provide data based on a variable number of participating hospitals.16
Red Latinoamericana de Malformaciones Congénitas (ReLAMC)
Created in 2016, in a post-epidemic context of microcephaly linked to congenital Zika virus infection in Northeast Brazil,17 the objective of ReLAMC is to provide up to date epidemiological information on congenital anomalies based on the hospital and population records of the participating Latin American countries. The data generated by this network can support research into CAs causes and prevention, as well as assisting with Public Health decision making.18 The network currently has ten record sources, five of which have national coverage, four have subnational coverage and one has multinational coverage, i.e. ECLAMC. It should be noted that ReLAMC goes beyond the objectives of ECLAMC, as it includes population-based data records.
Discussion
The six international collaboration networks covered by this narrative literature review have different characteristics as they seek to achieve different purposes, although all of them have the common objective of promoting surveillance of congenital defects. ECLAMC was the first network to be created in this sense, originating in 1967. Its case determination method (case-control design), as well as its longevity, enable ECLAMC to formulate time trends for CAs frequency. This contributes, progressively, to the enrichment of the specialized literature. Another important characteristic of the Latin American hospital-based registry is the egalitarian and voluntary way it is organized: the hospitals are not obliged to report their data periodically, and the health professionals involved are considered to be investigators and co-directors of the program, with equal access to the data collected.4
Besides ECLAMC, another network focused on hospital-based surveillance, SEAR-NBBD, was created in 2014 and its coverage area comprises some regions of South-East Asia. The region is especially relevant for the subject, as it has the world’s second highest congenital defect prevalence rate among children under five years old.20 Hospital-based CAs surveillance can be more feasible in relation to the reality of low and middle-income countries that wish to begin a monitoring program, given that population-based surveillance can involve high financial costs.2 Indeed, the two international hospital-based networks identified in this review are located in Latin America and South-East Asia, which concentrate countries with significant political, social and financial challenges to deal with.22
It should be noted that data obtained from hospital-based surveillance must be interpreted with caution, especially with regard to extrapolating results to entire populations, given that is coverage is limited to participating clinics or hospitals located within a restricted area and does not extend to total births to mothers living in a given geographic area. In addition, it is possible that there is preferential transferral of suspected cases of congenital anomalies to specialized hospitals that are more likely to be taking part in collaboration networks.2 Together, these factors may lead to incongruities in the statistics obtained and may even result in false alarms.4
On the other hand, BINOCAR and EUROCAT bring together European population-based registries (the only exception in the case of EUROCAT, is the hospital-based Spanish Collaborative Study of Congenital Malformations - ECEMC).11 In turn, ICBDSR and ReLAMC are of a more hybrid nature, having both population-based and hospital-based registries. Notwithstanding, it is important to highlight that the majority of the programs covered by the networks provided subnational coverage, which may also lead to biased interpretations if the results of the registries that monitor CAs in a delimited geographical area are extrapolated, in isolation, to larger regions.
The congenital anomaly surveillance networks truly comprise a source of reference for research groups and prevention and surveillance programs worldwide. Their data are used to report the frequency of rare outcomes, which would be hard to achieve using only individual registries. The aggregated experience of different programs has also served as support for countries (especially low and middle-income countries) that are planning to develop their own surveillance systems.3 EUROCAT, for example, provides standardized definitions, diagnosis methods and terminology, making it possible to compare data from heterogeneous programs.24 ICBDSR, jointly with WHO and US CDC, offers training courses and informative material for professionals involved in congenital defect surveillance.2 ECLAMC, in turn, contributes by developing a photographic atlas that assists with identifying more common congenital malformations.3
Some programs take part in more than one surveillance network, such as the Congenital Anomaly Registry and Information Service (CARIS), which is part of ICBDSR, EUROCAT and BINOCAR. It is a population-based registry created in 1998 and which currently covers all of Wales (approximately 35,000 births a year). The Clearinghouse and EUROCAT networks have the largest number of programs, and also share the largest number of individual registries. However, the recently created SEAR-NBBD network is isolated, in comparison to the other international networks. ECLAMC, apart from being an international in itself, shares the information it obtains with other networks, such as ICBDSR and ReLAMC. Brazil, through some associated hospitals, is an historic member of ECLAMC. Brazil participates in ReLAMC as a ‘guest’ in which its contribution is its Live Birth Information System (SINASC).
Among the networks identified, no surveillance program was found for any territory on the African continent. This situation is especially sensitive because it is in Africa that the world’s highest rates of mortality due to congenital defects are recorded: on the African continent, prevalence can vary between 5.2 and 75.4 cases per 10,000 live births.20 Given the importance of the subject and the need to understand the situation in order to be able to propose feasible interventions, some CAs surveillance initiatives area being established in African countries, such as a hospital-based surveillance program in Kampala, Uganda, as well as educational and instructional actions on the theme.6 In the future, the development of a continental network to integrate existing programs, as well as to strengthen new surveillance strategies, will be able to drive forward understanding and action with regard to congenital defects in Africa.
For the purposes of this study, international surveillance networks focusing on a specific outcome were not taken into consideration, such as the WHO initiative for surveillance of rubella and congenital rubella syndrome,27 nor did it take into consideration networks that did not extend beyond the boundaries of a country, such as the National Birth Defects Prevention Network (NBDPN) in the United States.28 Another initiative that was not used in our analyses, but which is worthy of mention is the International Union for Circumpolar Health Birth Defect Working Group (IUCH-BDWG), which has catalogued surveillance systems in existence and which are being developed in the more northerly regions of the world.30 However, to the best of our knowledge, so far that initiative has not provided systematic information about the programs that effectively form part of it, nor related statistics and information.
The results discussed here need to be interpreted in the light of some limitations. It is possible that international congenital defect surveillance networks exist that have not produced records capable of being captured by the methodological approach we used. Despite some networks not making available all necessary information on their websites or in scientific articles, the search we performed in the literature and in specialized documents helped to fill some gaps in variables of interest in this review. The landscape of the networks is not static: EUROCAT and ReLAMC, for instance, have some ‘candidate’ and ‘guest’ registries, respectively, that were not taken into consideration in this study, but which may become network members in the near future.
This review listed the world’s main international congenital defect surveillance collaboration networks. In the case of the six networks identified, we reviewed historical aspects and individual characteristics, as well as the main similarities and differences between them. Important aspects of congenital defect surveillance were also discussed. It was possible to perceive that, for congenital defect investigation and prevention (the frequency of which, individually, can be very rare), collaboration is a key word, both for strengthening existing programs and also for creating surveillance initiatives in regions where so far they are lacking. Moreover, the networks revealed themselves to be important sources of production and dissemination of knowledge among professionals involved in congenital anomaly prevention and surveillance, as well as the public in general.
Acknowledgements
We extend our thanks to all the professionals who collaborate with the registering of congenital anomalies worldwide.
REFERENCES
1. World Health Organization - WHO. Congenital anomalies [Internet]. Geneva: World Health Organization; 2016 [cited 2020 Jan 28]. Available from: https://www.who.int/news-room/fact-sheets/detail/congenital-anomalies [ Links ]
2. World Health Organization – WHO. Center for Disease Control and Prevention –CDC. International Clearinghouse for Birth Defects Surveillance and Research - ICBDSR. Birth defects surveillance: a manual for programme managers [Internet]. Geneva: World Health Organization; 2014 [cited 2020 May 26]. 116 p. Available from: https://www.who.int/publications-detail/9789241548724 [ Links ]
3. Bermejo-Sánchez E, Botto LD, Feldkamp ML, Groisman B, Mastroiacovo P. Value of sharing and networking among birth defects surveillance programs: an ICBDSR perspective. J Community Genet [Internet]. 2018 Oct [cited 2020 May 26];9(4):411-5. Available from: https://doi.org/10.1007/s12687-018-0387-z [ Links ]
4. Castilla EE, Orioli IM. ECLAMC: the Latin-American collaborative study of congenital malformations. Community Genet [Internet]. 2003 [cited 2020 May 26];7(2-3):76-94. Available from: https://doi.org/10.1159/000080776 [ Links ]
5. Luquetti DV, Koifman RJ. Surveillance of birth defects: Brazil and the US. Ciênc Saúde Coletiva [Internet]. 2011 [cited 2020 May 26];16(supl. 1):777-85. Available from: https://doi.org/10.1590/S1413-81232011000700008 [ Links ]
6. Kouame BD, N’Guetta-Brou IA, Kouame GS, Sounkere M, Koffi M, Yaokreh JB, et al. Epidemiology of congenital abnormalities in West Africa: results of a descriptive study in teaching hospitals in Abidjan: Cote d’Ivoire. Afr J Paediatr Surg [Internet]. 2015 Jan-Mar [cited 2020 May 26];12(1):51-5. Available from: https://doi.org/10.4103/0189-6725.150983 [ Links ]
7. Poletta FA, López Camelo JS, Gili JA, Leoncini E, Castilla EE, Mastroiacovo P. Methodological approaches to evaluate teratogenic risk using birth defect registries: advantages and disadvantages. PLoS One [Internet]. 2012 Oct [cited 2020 Jan 21];7(10):e46626. Available from: https://dx.plos.org/10.1371/journal.pone.0046626 [ Links ]
8. Poletta FA, Gili JA, Castilla EE. Latin American collaborative study of congenital malformations (ECLAMC): a model for health collaborative studies. Public Health Genomics [Internet]. 2014 [cited 2020 Jan 21];17(2):61-7. Available from: https://doi.org/10.1159/000356568 [ Links ]
9. Castilha EE, Zarante I, Guatibonza P, Contreras G, Mosquera D, et al. Congenital malformations browser [Internet]. [S.l.]; 2013 [cited 2020 Jan 21]. Available from: http://en.atlaseclamc.org/ [ Links ]
10. Kinsner-Ovaskainen A, Lanzoni M, Garne E, Loane M, Morris J, Neville A, et al. A sustainable solution for the activities of the European network for surveillance of congenital anomalies: EUROCAT as part of the EU Platform on Rare Diseases Registration. Eur J Med Genet [Internet]. 2018 Sep [cited 2020 May 26];61(9):513-7. Available from: https://doi.org/10.1016/j.ejmg.2018.03.008 [ Links ]
11. European Comission. EUROCAT network | EU RD Platform [Internet]. [S.l.]: European Comission; 2019 [cited 2020 Jan 20]. Available from: https://eu-rd-platform.jrc.ec.europa.eu/eurocat/eurocat-network/eurocat-network-overview [ Links ]
12. British and Irish Network of Congenital Anomaly Researchers - BINOCAR [Internet]. Londres: BINOCAR; 2019 [cited 2020 Jan 21]. Available from: http://www.binocar.org/ [ Links ]
13. Stevens S, Miller N, Rashbass J. Development and progress of the National Congenital Anomaly and Rare Disease Registration Service. Arch Dis Child [Internet]. 2018 Mar [cited 2020 May 26];103(3):215–8. Available from: http://dx.doi.org/10.1136/archdischild-2017-312833 [ Links ]
14. World Health Organization - WHO. Improving newborn and child health: a strategic framework (2018–2022) [Internet]. Geneva: World Health Organization: 2018 [cited 2020 May 26]. 116 p. Available from: https://apps.who.int/iris/handle/10665/274311 [ Links ]
15. World Health Organization - WHO. Birth defects in SEARO [Internet]. Geneva: World Health Organization; 2020 [cited 2020 Jan 21]. Available from: https://www.who.int/southeastasia/health-topics/birth-defects [ Links ]
16. World Health Organization - WHO. Perinatal surveillance and prevention of birth defects: progress review [Internet]. Bali: World Health Organization; 2017 [cited 2020 May 26]. 32 p. Available from: https://www.who.int/docs/default-source/searo/cah/bds-bali-mtg-17.pdf?sfvrsn=49a46cd8_2 [ Links ]
17. Schuler-Faccini L, Ribeiro EM, Feitosa IML, Horovitz DDG, Cavalcanti DP, Pessoa A, et al. Possible association between zika virus infection and microcephaly — Brazil, 2015. MMWR Morb Mortal Wkly Rep [Internet]. 2016 Jan [cited 2020 May 26];65(3):59-62. Available from: https://doi.org/10.15585/mmwr.mm6503e2 [ Links ]
18. Wilder-Smith A, Preet R, Brickley EB, Ximenes RAA, Miranda-Filho DB, Turchi Martelli CM, et al. ZikaPLAN: addressing the knowledge gaps and working towards a research preparedness network in the Americas. Glob Health Action [Internet]. 2019 Oct [cited 2020 May 26];12(1). Available from: https://doi.org/10.1080/16549716.2019.1666566 [ Links ]
19. Global Birth Defects. New surveillance network in Latin America—ReLAMC—building strength [Internet]. [S.l.]: Global Birth Defects; 2019 [cited 2020 Jan 21]. Available from: https://globalbirthdefects.tghn.org/news/new-surveillance-network-latin-americarelamcbuilding-strength/ [ Links ]
20. World Health Organization - WHO. Global health observatory data repository - number of deaths by region congenital anomalies [Internet]. Geneva: World Health Organization; 2018 [cited 2020 May 26]. Available from: https://apps.who.int/gho/data/view.main-eu.CM1002015REG6-CH15?lang=en [ Links ]
21. Mumpe-Mwanja D, Barlow-Mosha L, Williamson D, Valencia D, Serunjogi R, Kakande A, et al. A hospital-based birth defects surveillance system in Kampala, Uganda. BMC Pregnancy Childbirth [Internet]. 2019 Oct [cited 2020 Jan 27];19(372). Available from: https://doi.org/10.1186/s12884-019-2542-x [ Links ]
22. Kohli A. Coping with globalization: Asian versus Latin American strategies of development, 1980-2010. Rev Econ Polit [Internet]. 2012 Oct-Dec [cited 2020 May 26];32(4):531-56. Available from: https://doi.org/10.1590/S0101-31572012000400001 [ Links ]
23. Castilla EE, Orioli IM, Lopez-Camelo JS, Dutra MG, Nazer-Herrera J. Preliminary data on changes in neural tube defect prevalence rates after folic acid fortification in South America. Am J Med Genet [Internet]. 2003 Dec [cited 2020 May 26];123A(2):123-8. Available from: https://doi.org/10.1002/ajmg.a.20230 [ Links ]
24. Tucker FD, Morris JK, Neville A, Garne E, Kinsner-Ovaskainen A, Lanzoni M, et al. EUROCAT: an update on its functions and activities. J Community Genet [Internet]. 2018 [cited 2020 May 26];9(4):407-10. Available from: http://dx.doi.org/10.1007/s12687-018-0367-3 [ Links ]
25. Flores A, Valencia D, Sekkarie A, Hillard CL, Williams J, Groisman B, et al. Building capacity for birth defects surveillance in Africa: Implementation of an intermediate birth defects surveillance workshop. J Glob Heal Perspect [Internet]. 2015 Jan [cited 2020 Jan 27];1. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26753106 [ Links ]
26. Ajao AE, Adeoye IA. Prevalence, risk factors and outcome of congenital anomalies among neonatal admissions in OGBOMOSO, Nigeria. BMC Pediatr [Internet]. 2019 Apr [cited 2020 Jan 27];19(1):88. Available from: https://doi.org/10.1186/s12887-019-1471-1 [ Links ]
27. Grant GB, Desai S, Dumolard L, Kretsinger K, Reef SE. Progress toward rubella and congenital rubella syndrome control and elimination — Worldwide, 2000 –2018. MMWR Morb Mortal Wkly Rep [Internet]. 2019 Oct [cited 2020 Jan 28];68(39):855-9. Available from: http://dx.doi.org/10.15585/mmwr.mm6839a5 [ Links ]
28. Kirby RS. The prevalence of selected major birth defects in the United States. Semin Perinatol [Internet]. 2017 Oct [cited 2020 May 26];41(6):338-44. Available from: https://doi.org/10.1053/j.semperi.2017.07.004 [ Links ]
29. Matas JL, Agana DFG, Germanos GJ, Hansen MA, Modak S, Tanner JP, et al. Exploring classification of birth defects severity in national hospital discharge databases compared to an active surveillance program. Birth Defects Res [Internet]. 2019 Nov [cited 2020 Jan 30];111(18):1343-55. Available from: https://doi.org/10.1002/bdr2.1539 [ Links ]
30. Arbour L, Melnikov V, McIntosh S, Olsen B, Osborne G, Vaktskjold A. The current state of birth outcome and birth defect surveillance in northern regions of the world. Int J Circumpolar Health [Internet]. 2009 Dec [cited 2020 May 26];68(5):4430-58. Available from: https://doi.org/10.3402/ijch.v68i5.17376 [ Links ]
31. International Union for Circumpolar Health - IUCH. Congenital anomalies and birth outcome working group [Internet]. [S.l.]: International Union for Circumpolar Health; 2013 [cited 2020 Jan 30]. Available from: https://iuch.net/working-groups/#tab-id-5 [ Links ]
Received: April 09, 2020; Accepted: May 12, 2020










 text in
text in