Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista Pan-Amazônica de Saúde
versão impressa ISSN 2176-6215versão On-line ISSN 2176-6223
Rev Pan-Amaz Saude v.1 n.1 Ananindeua mar. 2010
http://dx.doi.org/10.5123/S2176-62232010000100013
Ocurrence of Human Bocavirus associated with acute respiratory infections in children up to 2 years old in the City of Belém, Pará State, Brazil
Allan Kaio SilvaI; Mirleide Cordeiro dos SantosII; Wyller Alencar de MelloII; Rita Catarina Medeiros de SousaI
INúcleo de Medicina Tropical,
Universidade Federal do Pará, Belém, Pará, Brasil
IIInstituto Evandro Chagas/SVS/MS, Ananindeua, Pará,
Brasil
Endereço para
correspondência
Correspondence
Dirección para correspondencia
Original Title: Ocorrência de Bocavírus Humano associado às infecções respiratórias agudas em crianças de 0 a 2 anos de idade na Cidade de Belém, Pará, Brasil. Translated by: American Journal Experts
ABSTRACT
INTRODUCTION: Acute Respiratory Infections
(ARI) are one of the main public health problems in the world. Most of these
infections are associated with several pathogens, and viruses are the most
prevalent agents. Recently, a new parvovirus named Human Bocavirus (HBoV)
has been described. Investigations on the relationship between this new agent
and cases of ARI in individuals are still scarce. Herein, we review a study
of HBoV in a population segment in the Amazon.
MATERIALS AND METHODS: In this study, samples of nasopharyngeal aspirates
from patients with ARI treated in Health Care Units in Belém, Brazil,
were analyzed. Identification of the virus was carried out by polymerase
chain reaction using pairs of specific oligonucleotides, followed by phylogenetic
analysis of the nucleotide sequences obtained.
RESULTS: Of the 397 samples studied, three specimens were HBoV-positive,
and one presented as a co-infection with the respiratory syncytial virus.
DISCUSSION: The positivity rate obtained in this investigation was
lower than that described in other studies; however, previous studies involved
hospitalized patients, which constitute a different population group. The
phylogenetic analyses revealed a significant similarity between the virus
strains found and those previously described.
CONCLUSION: This is the first report associating HBoV with ARI in
the Amazon.
Keywords: Respiratory Tract Infections; Bocavirus; Parvoviridae Infections.
INTRODUCTION
Due to their high mortality, Acute Respiratory Infections (ARI) are still one of the main public health problems worldwide, especially in developing countries7,28. These infections are generally associated with various pathogens with viruses being the most prevalent6,13. Among these viruses are Influenza A and B (Flu A and Flu B), Parainfluenza 1, 2 and 3 (HPIV), Adenovirus (AdV), Respiratory Syncytial Virus (RSV), Human Rhinovirus (HRV), Coronavirus (HCoV) and Human Metapneumovirus (hMPV)3,18.
Several studies are currently being conducted in order to determine the etiology of respiratory tract infections. In 12% to 39% of cases, no known agent is found. Under this premise, Allander et al2 have described the development of a novel methodology based on molecular biology techniques in order to investigate new agents possibly related to respiratory tract infections. Using this methodology, these authors detected a parvovirus, previously called Human Bocavirus (HBoV), which is taxonomically classified within the genus Bocavirus, subfamily Parvovirinae, family Parvoviridae2,14.
HBoV is a non-enveloped virus with icosahedral symmetry, a diameter of 18 to 26 nanometers and a capsid formed by approximately 60 capsomeres. The viral genome is non-segmented, composed of a single-stranded deoxyribonucleic acid (DNA) of both positive and negative sense and is approximately 5.3 Kb. The virus has three open reading frames (ORFs) that encodes four proteins: VP1 and VP2, virion structural proteins; NS1, non-structural protein; and the nucleoprotein NP1, which has an unknown function.
Studies have shown that HBoV apparently has a worldwide distribution5,9,11,16,23,25,27, with an incidence ranging from 1,5%5 to 19%1. Few studies have been conducted in Latin America to determine the association of HBoV with other respiratory infections. The high percentage of cases in which the virus is found in the presence of another infection (5 - 85%), either with another virus or other pathogens12,25,27, is noteworthy.
The transmission of HBoV is undefined because it has been found several times in the respiratory tract5,9,23,27, serum15, blood1,17 and urine24, as well as in cases of gastroenteritis4,24. Nevertheless, studies have shown that in patients younger than 2 years of age1,24 hospitalized for a respiratory tract infection, HBoV is the fourth most frequently detected agent, suggesting that the respiratory tract is the main route of transmission.
The symptoms observed in cases of HBoV infection include those of a common cold with fever and rhinorrhea as well as wheezing and dyspnea. In the event of complications, the occurrence of bronchitis, bronchiolitis and pneumonia have been noted11,20,23. Studies also suggest that HBoV can establish latent or persistent infections in mucosal lymphocytes or contribute to tonsillar hyperplasia22.
The seasonal profile of HBoV infection cases has yet to be well established. However, in countries with a temperate climate, the occurrence of this virus is more pronounced in the winter and early spring11,27.
To date, studies have demonstrated the existence of three types of HBoV (HBoV1, HBoV2 and HBoV3). Type 3 has only been found in feces4, and type 2 has been found in both blood and feces4,17. HBoV1 is the only type associated with ARI.
As for the genetic variability, Allander et al2 described two very conserved lineages (ST1 and ST2). This seems to be a very stable virus, and few mutations are found in the VP1 and VP2 genes sequences. Divergences in nucleotide sequences are caused by point mutations, which result in a few changes in the amino-acid chain16,25.
In countries with a tropical climate, such as Brazil, little research has been conducted on the occurrence and genetic variability of HBoV. Thus, there is a clear need to conduct studies that will generate epidemiological data on this virus in order to better define the role of HBoV in cases of ARI in Amazônia.
MATERIALS AND METHODS
STUDY POPULATION
Between 2004 and 2007, combined swab samples (nasal/throat) and nasopharyngeal aspirates were collected in an outpatient setting by the Sistema de Vigilância Virológica da Rede Influenza (Virological Surveillance System of the Influenza Network), the Laboratório de Vírus Respiratórios (LVR - Respiratory Virus Laboratory) of the Instituto Evandro Chagas (IEC). A screening procedure allowed selection of children younger than 2 years of age who presented with signs and symptoms of ARI lasting no longer than five days.
SAMPLE PROCESSING
The collected samples were centrifuged at 1000 rpm for 10 min. The pellets from the centrifuged samples were used to prepare slides according to the protocol in the commercial kit Light Diagnostics™ Respiratory Panel I Viral Screening and Identification IFA for Indirect Immunofluorescence (IDIF) that was used to identify the viruses Influenza A and B, Parainfluenza 1-3, Adenovirus and RSV. The supernatant was used in an attempt to isolate HBoV using molecular biology techniques.
IDENTIFICATION USING MOLECULAR BIOLOGY
For the extraction, we standardized a protocol in which we initially added 150 µL of TNE (Tris-Na-EDTA) buffer, 20 µL of 10% sodium dodecyl sulfate (SDS) and 10 µL of proteinase K (10 mg/mL) to 120 µL of the sample in a microtube. This mixture was incubated at 56° C for 30 min in a thermal block (Eppendorf). We then added 200 µL of saturated phenol, vortexed the sample for 1 min and centrifuged it at 20° C for 3 min at 10,000 rpm. We transferred 150 µL of the supernatant to a microtube containing 150 µL of phenol, chloroform and isoamyl alcohol. This mixture was then vortexed for 1 min and centrifuged at 20° C for 3 min at 10,000 rpm. A total of 100 µL of the resulting supernatant was transferred to a microtube containing 100 µL of chloroform, vortexed for 1 min and centrifuged at 20° C for 3 min at 10,000 rpm. Finally, we carefully removed 40 µL of the supernatant and stored it at -20° C.
A polymerase chain reaction (PCR) was conducted using the following pairs of specific oligonucleotides: BoV 118F and BoV 542R (NP1), VP/+/1 and VP/-/726 (VP1) and VP/+/1005 and VP/-/2072 (VP2). The primers and the amplicon sizes can be found in table 1.

For all the genes, we used a reaction with a final volume of 50 (µL containing: 5 of extracted DNA, 0.5 µL of each primer (50 pmol/µL), 5 µL of reaction buffer (10x), 2 µL of dNTPs (5 mM), 5 µL of MgCl2 (25 mM), 1.25 U of Taq DNA polymerase and 31.5 µL of DNAse/RNAse-free water. The adopted program was specified by Allander et al2.
PCR samples positive for HBoV were tested for the VP1/VP2 gene, prepared for partial sequencing with the Kit Big Dye® terminator Cycle Sequencing (Applied Biosystem) according to the manufacturer's instructions and sequenced with the ABIPrism 3130xl automated sequencer (Applied Biosystem).
All the reactions were performed in a Mastercycler ep Gradient S (Eppendorf) automated thermocycler. Negative and positive controls were always performed to avoid contamination by exogenous DNA.
SEQUENCE ANALYSIS AND EDITING AND PHYLOGENETIC TREE CONSTRUCTION
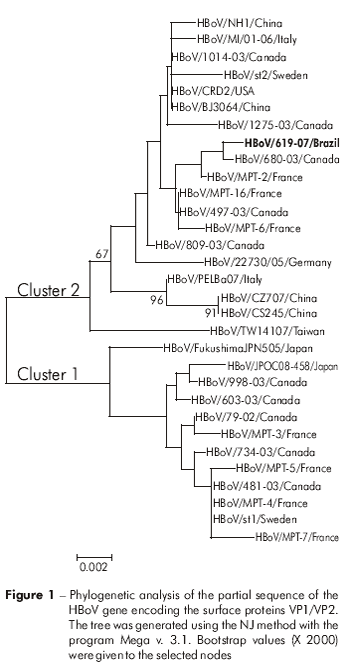
The nucleotide sequences obtained from HBoV were analyzed and edited using the program BioEdit v. 7.0 and compared with sequences of other viral isolates available in the GenBank database (http://www.ncbi.nlm.nih.gov), using the programs Clustal W v. 1.726 and Mega v. 3.121. The neighbor joining (NJ) method was used to construct phylogenetic trees on the program Mega v 3.1. The distance matrix in the NJ method was calculated based on the aligned sequences by using Kimura's19 two-parameter formula. We used a Bootstrap analysis with two thousand replicates to enhance reliability of cluster analysis10.
RESULTS
We analyzed 397 samples, 3 (0.76%) of which were positive for HBoV infection, as confirmed by sequencing.
For all the positive cases, the infection occurred during the dry season and affected male children (on average, 29 weeks old) who presented rhinitis and cough. In one case, a HBoV/RSV coinfection was found. This child also presented fever and nasal congestion (Table 2).

For the phylogenetic analysis, the NP1 segment was 100% similar to the strains described by Allander et al2. For the sequence analysis of the VP1/VP2 gene, one sample was sequenced and was 98.3% to 99.7% similar to strains available in GenBank (Figure 1).
DISCUSSION
In the City of Belém, Pará, from January 2004 to December 2007, three (0.76%) cases of Human Bocavirus-associated acute respiratory infection were diagnosed from a total of 397 samples collected locally. Two positive samples were detected in 2004 and another in 2007.
These data reveal a low percentage of HBoV-positive cases when compared to the data available in the literature, which report detection in up to 19% of cases1. However, we should emphasize that the samples used in this study originated from patients in an outpatient setting, whereas the studies with higher rates of association included samples from hospitalized patients9,27.
The low positivity rates did not lead to conclusions relative to the epidemiological patterns of prevalence and seasonality caused by the virus in the study population. However, we emphasize the occurrence of the virus in the period of lowest seasonal rainfall.
One of the samples presented coinfection with RSV In fact, a high incidence of association between HBoV and other viruses, frequently in association with RSV, is often described in the literature2,8.
When comparing our sequences with those from GenBank, we observed 100% similarity for NP1 and 98.6% to 99.3% similarity for the partial sequences of VP1/VP2. These data demonstrate the high level of conservation for NP1 and reinforces the low genomic mutability of VP1/VP216,25.
CONCLUSION
This study shows a low index (0.76%) of HBoV in 0 to 2 years of age children with a coinfection with other respiratory viruses receiving ambulatory care in the capital of Pará.
The strains circulating in the region of the study had few mutations. When analyzed using their non-structural protein, an overall similarity was found relative to the strain lineage from a study conducted in Sweden, designated ST1. In this study, we emphasize that this is the first incidence of HBoV-associated respiratory infections detected in the Legal Amazon.
REFERENCES
1 Allander T, Jartti T, Gupta S, Niesters HGM, Lehtinen P, Osterback R, et al. Human bocavirus and acute wheezing in children. Clin Infect Dis. 2007 Apr;44(7):904-10. DOI:10.1086/512196 [ Links ]
2 Allander T, Tammi MT, Eriksson M, Bjerkner A, Tiveljung-Lindell A, Andersson B. Cloning of a human parvovirus by molecular screening of respiratory tract samples. Proc Natl Acad Sci U S A. 2005 Sep;102(36):12891-6. Erratum in: Proc Natl Acad Sci U S A. 2005;102(43):15712. DOI:10.1073/pnas.0504666102 [ Links ]
3 Arruda E, Cintra AOL, Hayden FG. Respiratory tract viral infections. In: Guerrant RL, Walker DH, Weller PF, editors. Tropical infectious disease: principles, pathogens, and practice. Philadelphia: Churchill Livingstone Elsevier; 2006. p. 637-59.
4 Arthur JL, Higgins GD, Davidson GP, Givney RC, Ratcliff RM. A novel bocavirus associated with acute gastroenteritis in Australian children. PLoS Pathog. [Internet]. 2009 Apr [citado 2009 jun 6];5(4):1-11. Disponível em: http://www.plospathogens.org/article/info:doi/10.1371/journal.ppat.1000391. DOI:10.1371/journal.ppat.1000391. [ Links ]
5 Bastien N, Brandt K, Dust K, Ward D, Li Y. Human bocavirus infection, Canada. Emerg Infect Dis. 2006 May;12(5):848-50. [ Links ]
6 Beckett CG, Kosasih H, Ma'roef C, Listiyaningsih E, Elyazar IR, Wuryadi S, et al. Influenza surveillance in Indonesia: 1999-2003. Clin Infect Dis. 2004 Aug;39(4):443-9. DOI:10.1086/422314 [ Links ]
7 Chiesa AM, Westphal MF, Kashiwagi NM. Geoprocessing and health promotion: social and environmental inequalities, Brazil. Rev Saude Publica. 2002 Oct;36(5):559-67. DOI:10.1590/S0034-89102002000600004. [ Links ]
8 Christensen A, Nordbø SA, Krokstad S, Rognlien AG, Døllner H. Human bocavirus commonly involved in multiple viral airway infections. J Clin Virol. 2008 Jan;41(1):34-7. DOI:10.1016/j.jcv.2007.10.025 [ Links ]
9 Chung JY, Han TH, Kim CK, Kim SW. Bocavirus infection in hospitalized children, South Korea. Emerg Infect Dis. 2006 Aug;12(8):1254-6. [ Links ]
10 Felsenstein J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution. 1985 Jul;39(4):783-91. [ Links ]
11 Foulongne V, Olejnik Y, Perez V, Elaerts S, Rodière M, Segondy M. Human bocavirus in French children. Emerg Infect Dis. 2006 Aug;12(8):1251-3. [ Links ]
12 Gagliardi TB, Iwamoto MA, Paula FE, Proença-Modena JL, Saranzo AM, Criado MF, et al. Human bocavirus respiratory infections in children. Epidemiol Infect. 2009 Jul;137(7):1032-6. DOI:10.1017/S0950268808001842 [ Links ]
13 Galiano M, Videla C, Puch SS, Martínez A, Echavarría M, Carballal G. Evidence of human metapneumovirus in children in Argentina. J Med Virol. 2004 Feb;72(2):299-303. DOI:10.1002/jmv.10536 [ Links ]
14 International Committee on Taxonomy of Viruses. Virus index database [Internet]. 2009. [citado 2009 jul 5]. Disponível em: http://www.danforthcenter.org/iltab/ictvnet/asp/iVirusIndex.asp. [ Links ]
15 Jones MS, Kapoor A, Lukashov VV, Simmonds P, Hecht F, Delwart E. New DNA viruses identified in patients with acute viral infection syndrome. J Virol. 2005 Jul;79(13):8230-6. DOI:10.1128/JVI.79.13.8230-8236.2005 [ Links ]
16 Kaplan NM, Dove W, Abu-Zeid AF, Shamoon HE, Abd-Eldayem SA, Hart CA. Human bocavirus infection among children, Jordan. Emerg Infect Dis. 2006 Sep;12(9):1418-20. [ Links ]
17 Kapoor A, Slikas E, Simmonds P, Chieochansin T, Naeem A, Shaukat S, et al. A newly identified bocavirus species in human stool. J Infect Dis. 2009 Jan;199(2):196-200. DOI:10.1086/595831 [ Links ]
18 Kesson AM. Respiratory virus infections. Paediatr Respir Rev. 2007 Sep;8(3):240-8. DOI:10.1016/j.prrv.2007.07.003 [ Links ]
19 Kimura M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol. 1980 Dec;16(2):111-20. [ Links ]
20 Kleines M, Scheithauer S, Rackowitz A, Ritter K, Häusler M. High prevalence of human bocavirus detected in young children with severe acute lower respiratory tract disease by use of a standard PCR protocol and a novel real-time PCR protocol. J Clin Microbiol. 2007 Mar;45(3):1032-4. DOI:10.1128/JCM.01884-06 [ Links ]
21 Kumar S, Tamura K, Nei M. MEGA3: Integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform. 2004 Jun;5(2):150-63. [ Links ]
22 Lu X, Gooding LR, Erdman DD. Human bocavirus in tonsillar lymphocytes. Emerg Infect Dis. 2008 Aug;14(8):1332-4. [ Links ]
23 Ma X, Endo R, Ishiguro N, Ebihara T, Ishiko H, Ariga T, et al. Detection of human bocavirus in Japanese children with lower respiratory tract infections. J Clin Microbiol. 2006 Mar;44(3):1132-4. DOI:10.1128/JCM.44.3.1132-1134.2006 [ Links ]
24 Pozo F, García-García ML, Calvo C, Cuesta I, Pérez-Breña P, Casas I. High incidence of human bocavirus infection in children in Spain. J Clin Virol. 2007 Nov;40(3):224-8. DOI:10.1016/j.jcv.2007.08.010 [ Links ]
25 Smuts H, Hardie D. Human bocavirus in hospitalized children, South Africa. Emerg Infect Dis. 2006 Sep;12(9):1457-8. [ Links ]
26 Thompson JD, Higgins DG, Gibson TJ. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994 Nov;22(22):4673-80. [ Links ]
27 Weissbrich B, Neske F, Schubert J, Tollmann F, Blath K, Blessing K, et al. Frequent detection of bocavirus DNA in German children with respiratory tract infections. BMC Infect Dis. 2006 Jul;6:109. DOI:10.1186/1471-2334-6-109 [ Links ]
28 World Health Organization. The world health report 2005: make every mother and child count [Internet]. Geneva; 2005. [citado 2006 mai 15]. Disponível em: http://www.who.int/whr/2005/annex/annexes3-4_en.pdf. [ Links ]
 Correspondência/Correspondence/Correspondencia:
Correspondência/Correspondence/Correspondencia:
Allan Kaio Silva
Universidade Federal do Pará
Núcleo de Medicina Tropical
E-mail:alansilva@iec.pa.gov.br
Recebido em/Received/Recibido en: 30/07/2009
Aceito em/Accepted/Aceito en: 01/10/2009











 texto em
texto em 

