Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista Pan-Amazônica de Saúde
versão impressa ISSN 2176-6215versão On-line ISSN 2176-6223
Rev Pan-Amaz Saude v.1 n.3 Ananindeua set. 2010
http://dx.doi.org/10.5123/S2176-62232010000300015
ARTIGO DE REVISÃO | REVIEW ARTICLE | ARTÍCULO DE REVISIÓN
Methods for assessment of antimalarial activity in the different phases of the Plasmodium life cycle
Métodos para avaliação da atividade antimalárica nas diferentes fases do ciclo de vida do Plasmodium
Métodos para evaluación de la actividad antimalárica en las diferentes fases del ciclo de vida del Plasmodium
Fátima Nogueira; Virgílio Estólio do Rosário
Unidade de Ensino e Investigação da Malária, Centro de Malária e Outras Doenças Tropicais, Laboratório Associado, Instituto de Higiene e Medicina Tropical, Universidade Nova de Lisboa, Lisboa, Portugal
Endereço para correspondência
Correspondence
Dirección para correspondencia
ABSTRACT
Malaria is a mosquito-borne disease caused by parasites of the genus Plasmodium. In humans, parasites multiply in the liver and then infect red blood cells. The Plasmodium life cycle consists of a sexual phase in the mosquito vector (sporogony) and an asexual phase in the vertebrate host (schizogony); both life cycle phases can be detected in assays. In general, in vivo and in vitro are the two basic approaches routinely used to evaluate the antimalarial activity of compounds. The antimalarial activity measured in an in vivo test results from a variety of factors associated with both the parasite and the host. Conversely, in vitro tests reflect more accurately the "isolated" effects of the compounds on parasite metabolism. In vivo assessment of antiplasmodial activity can be achieved using rodent models and by assessing transmission-blocking activity using mosquitoes. There are several in vitro tests for the assessment of antimalarial activity based upon observation of parasite development in blood cells through thick films, isotopic assays, quantification of parasite proteins and DNA dye intercalation assays. Besides antimalarial activity, a promising antimalarial compound should also lack toxicity to host cells; the degree of selectivity of a compound towards the malaria parasite includes such assessment. In this manuscript, we intend to summarize the most frequently used methods for assessing the antimalarial activity of compounds in the different stages of the Plasmodium life cycle.
Keywords: Malaria; Drug Resistance; Antimalarials; Cytotoxicity Tests, Immunologic; Plasmodium.
RESUMO
A malária é uma doença transmitida por mosquitos e causada por parasitas do gênero Plasmodium. Em seres humanos, os parasitas multiplicam-se no fígado e, em seguida, infectam os eritrocitos. O ciclo de vida do Plasmodium consiste em uma fase sexuada no mosquito vetor (esporogonia) e uma fase assexuada no hospedeiro vertebrado (esquizogonia); ambas as fases podem ser detectadas por meio de testes. Em geral, a avaliação da atividade antimalárica de compostos é feita por testes in vivo e in vitro. A atividade antimalárica determinada por testes in vivo decorre de uma variedade de fatores associados tanto ao parasita quanto ao hospedeiro. Por outro lado, os testes in vitro mostram com mais precisão os efeitos "isolados" dos compostos sobre o metabolismo do parasita. A análise in vivo da atividade antiplasmódica pode ser realizada com o uso de modelos de roedores e pela avaliação da atividade bloqueadora do potencial vetor utilizando mosquitos. Há diversos testes in vitro para a avaliação da atividade antimalárica com base na observação do desenvolvimento do parasita em células sanguíneas por meio de gota espessa, ensaios isotópicos, quantificação de proteínas do parasita e testes de intercalação no DNA com o uso de corantes. Além da atividade antimalárica, um composto antimalárico promissor não deve apresentar toxicidade em relação às células hospedeiras; o grau de seletividade de um composto em relação ao parasita da malária envolve esta análise. Neste artigo, pretende-se resumir os métodos mais comumente utilizados para avaliar a atividade antimalárica de compostos durante os diferentes estágios do ciclo de vida do Plasmodium.
Palavras-chave: Malária; Resistência a Medicamentos; Antimaláricos; Testes Imunológicos de Citotoxicidade; Plasmodium.
RESUMEN
La malaria es una enfermedad transmitida por mosquitos y causada por parásitos del género Plasmodium. En seres humanos, los parásitos se multiplican en el hígado y en seguida, infectan los eritrocitos. El ciclo de vida del Plasmodium consiste en una fase sexuada en el mosquito vector (esporogonia) y una fase asexuada en el huésped vertebrado (esquizogonia); ambas fases pueden detectarse por intermedio de pruebas. En general, la evaluación de la actividad antimalárica de compuestos se hace por ensayos in vivo e in vitro. La actividad antimalárica determinada por ensayo in vivo resulta de una variedad de factores asociados tanto al parásito como al huésped. Por otro lado, las pruebas in vitro muestran con más precisión los efectos "aislados" de los compuestos sobre el metabolismo del parásito. El análisis in vivo de la actividad antiplasmódica pode realizarse con el uso de modelos de roedores y por la evaluación de la actividad bloqueadora del potencial vector utilizando mosquitos. Existen diversos testes in vitro para la evaluación de la actividad antimalárica basados en la observación del desarrollo del parásito en células sanguíneas a través de gota espesa, ensayos isotópicos, cuantificación de proteínas del parásito y pruebas de intercalación en el ADN con el uso de colorantes. Además de la actividad antimalárica, un compuesto antimalárico prometedor no debe presentar toxicidad en relación a las células huéspedes; el grado de selectividad de un compuesto en relación al parásito de la malaria está involucrado en este análisis. En este artículo, se pretende resumir los métodos más comúnmente utilizados para evaluar la actividad antimalárica de compuestos durante las diferentes etapas del ciclo de vida del Plasmodium.
Palabras clave: Malaria; Resistencia a Medicamentos; Antimaláricos; Pruebas Inmunológicas de Citotoxicidad; Plasmodium.
INTRODUCTION
Approximately 300 million people worldwide are affected by malaria and between 1 and 1.5 million people die from it every year. Previously extremely widespread, malaria is now mainly confined to Africa, Asia and Latin America. Malaria species predominate with different prevalence depending on geographic region; for example Plasmodium vivax is more prevalent than P. falciparum in Latin America. In general, the absence of a vaccine and the problem of drug resistance has hindered many malaria control programs.
PLASMODIUM LIFE CYCLE
Malaria infects both a vertebrate and an invertebrate host. Mosquitoes ingest Plasmodium gametocytes in a blood meal from an infected vertebrate host, and the gametocytes then develop into infectious form (sporozoites) within the salivary glands of the mosquito. Sporogony in the mosquito takes approximately 10 - 20 days, and thereafter the mosquito remains infective for 1 - 2 months. The schizogonic phase starts with the inoculation of the parasite into the blood of the vertebrate host by the bite of a female Anopheles mosquito. In a short period of time, the sporozoites invade liver cells through a complex cycle1 and initiate schizogony, which has two stages, the hepatic stage (pre-erythrocytic schizogony) and the erythrocytic stage (erythrocytic schizogony). Within hepatocytes, the trophozoites start their intracellular asexual division. At the completion of this stage, thousands of exo-erythrocytic merozoites are released from each infected hepatocyte. The time taken for the completion of the tissue phase is variable and depends on the infecting species: 8 - 25 days for P. falciparum, 8 - 27 days for P. vivax, 9 - 17 days for P. ovale, 15 - 30 days for P. malariae and 9 - 12 days for P. knowlesi2. This interval is called the pre-patent period. In the case of P. vivax and P. ovale, some sporozoites may go into latency, also known as the cryptobiotic phase, where they are called hypnozoites. They can lie dormant for months or years, and upon reactivation they cause clinical relapse. The merozoites released from the hepatocytes enter red blood cells and start asexual division, developing through the stages of rings, trophozoites, early schizonts and mature schizonts. Each mature schizont consists of thousands of erythrocytic merozoites. These merozoites are then released by the lysis of the erythrocyte, and they immediately invade uninfected erythrocytes. This repetitive cycle of invasion - multiplication - release - invasion continues. It synchronously occurs as the merozoites are released at approximately the same time, resulting in the characteristic clinical manifestations of the disease. A small proportion of the merozoites in the erythrocyte undergo transformation into gametocytes, male and female, and the full cycle will be reinitiated if another female mosquito feeds on the infected vertebrate host.
ASSESSMENT OF ANTIMALARIAL ACTIVITY
In general, two basic approaches are routinely used to study or measure antimalarial activity of compounds: a) in vivo and b) in vitro. These can be complementary, as data obtained from in vitro assays require confirmation from the use of live models.
A - An in vivo test is associated with animal models and their compulsory ethical issues. In these assays, one measures mainly a) the clearance of parasites as detected by optical microscopy or other more sensitive methods (polymerase chain reaction - PCR), b) the time that elapses between last drug dose and clearance of parasitemia, and c) the drug dosage that clears parasites in a dose response manner.
When adjusted to humans, in vivo studies usually represent the following of a selected group of symptomatic and parasitemic individuals that underwent carefully controlled treatment with subsequent monitoring of the parasitological and/or clinical responses over time. Diminished therapeutic efficacy of a drug/compound can be masked by immune-mediated clearance of parasites among hosts with acquired immunity. When performed with human subjects, in vivo tests most closely reflect actual clinical or epidemiological situations; that is, the therapeutic response of currently circulating parasites infecting the actual population in which the drug will be used.
The screening of antimalarial activity of new compounds follows standard procedures*. Non-human primates have provided experimental models for P. falciparum malaria3. Nevertheless, the use of this animal model has been difficult because of the economic and ethical considerations, a limited supply of monkeys, the narrow range of parasite lines that are adapted to primate infection and the different pathology that is seen in these models4,5,6. Consequently, most in vivo experimental studies on malaria have relied on different combinations of various murine strains and Plasmodium spp. of rodents7,8; however, the biological differences between parasite species must be taken into account9.
External factors, such as host immunity, variations of drug absorption and metabolism, are reduced by the use of naive animals from well-known lines in the experiments. Also, potential misclassification of re-infection is not an issue, as assays use direct inoculation of blood stage parasites in well-protected animal house surroundings. However, antimalarial activity measured in an in vivo test does not always consider relevant side factors, such as metabolism and host genetics, which can affect the outcome of the experiment.
B - In vitro tests avoid some of the confounding factors listed above by removing parasites from the host and placing them into a controlled experimental environment. In the most frequently used procedure, P. falciparum parasites, obtained from culture or a human patient, are exposed to precisely known quantities of a drug or compound. Then the inhibition of parasite maturation is monitored to examine a dose dependent effect of the drug in preventing parasite development from ring into schizont. For assays related to sporozoite infectivity, P. berghei sporozoites in a HepG2 liver cell culture are usually used to study the drug dose effect on schizont maturation. The correlation of in vitro response data with clinical response in patients is not consistent, as clinical cure is affected by the patient's immune system. Another example of the limitation of in vitro assays is the use of prodrugs, such as proguanil, which requires host conversion into active metabolites. Although adaptation of erythrocytic forms of P. vivax to continuous culture has been achieved, their use in assays is complex and infeasible. In general, these tests are demanding and relatively expensive, but innovations are being implemented which may make them easier to adapt to routine field-work and make them more efficient as high-throughput tools for drug activity screens.
ASSESSMENT OF ANTIMALARIAL ACTIVITY OF COMPOUNDS DURING ERYTHROCYTIC STAGE
Antimalarial drug screening and validation is relatively time consuming and complicated. The first phase of drug screening is usually comprised of two steps. The first step involves the use of whole cell assays for the determination of the effect of the compound on the growth of the human parasite P. falciparum in vitro. The second step involves the testing of the in vivo efficacy of selected drugs in small animal models of malaria, mainly using the rodent parasites P. berghei, P. yoelii and P. chabaudi in laboratory mice.
PARASITE CULTURE
Most approaches to in vitro drug-sensitivity assays are based on the culturing of malaria parasites in erythrocytes, which was developed in the 1970s10,11. This development allowed for the removal of the parasite from the patient's bloodstream and its transfer into a highly controlled laboratory environment. It also allows research in non-endemic regions where there is no supply of fresh parasite samples. Short-term culture of P. falciparum parasites is fairly simple and requires little laboratory equipment, and it can be easily performed under field conditions. Although continuous culture poses considerably more technical and logistic challenges, it has nevertheless become an essential tool in laboratories working on malaria drug development and basic research. Together, with the capability to cryopreserve live parasites12, culturing provides the basis for studies that require high parasite densities (proteomics) and for cloning. Although P. falciparum is asynchronous during in vitro culture, artificial synchronization (Figure 1) of erythrocytic stages of the parasite in culture is useful for studies searching for erythrocytic stage malaria vaccine candidates.

IN VITRO TESTS
The main objective in drug-sensitivity testing is to evaluate or measure the parasite (P. falciparum) sensitivity to increasing drug dosages in vitro; data are presented as percentages of parasite growth compared to untreated cultures. This approach allows for an almost complete exclusion of interfering host-related factors, such as host immunity and host metabolism of the compound, thus offering a direct evaluation of drug impact. In vitro tests are basically without risk to the patient, are non-invasive (apart from the collection of a blood sample), and are crucial for drug development. In vitro tests for screening of antimalarial activity generally utilize the method of continuous culture developed by Trager and Jensen11 and the microculture technique developed by Rieckmann et al13. The short-term in vitro culture of freshly collected field isolates of P. vivax and P. ovale has also been established14,15, also allowing for the assessment of antimalarial activity of different drugs on these plasmodium species.
In terms of drug screening, several in vitro methods are commonly used for the assessment of P. falciparum growth inhibition: a) schizont counting on thick films, known as the World Health Organization (WHO) Schizont Maturation assay16; b) incorporation of a radiolabeled nucleic acid precursor (e.g., radiolabeled hypoxanthine) known as the isotopic assay17,18; c) the quantification of parasite proteins histidine-rich protein 2 (HRP2) and lactate dehydrogenase (LDH) by enzyme-linked immunosorbent assay (ELISA)19,20; and d) DNA dye intercalation assays21,22,23,24.
Schizont counting on thick films
This drug sensitivity test is based on parasite counts following the evaluation of the morphology of parasite growth (rings into schizonts) by light microscopy. The WHO Mark III microtest assay (Table 1) is a low-cost alternative standardized by the WHO*. Experienced microscopists should carry out this test, as it is labor-intensive and prone to individual variability. It is based on the maturation of P. falciparum in a 24 to 36 h microculture, by microscopically counting the number of parasites that develop into schizonts (i.e. parasites with three or more chromatins) in Giemsa-stained thick films. Despite these drawbacks, the WHO assay is economical and simple to perform in the field. A number of published reports from Brazil have utilized this technique25,26,27,28.
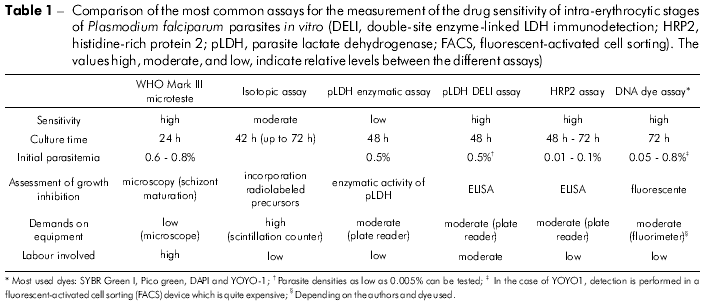
Isotopic assays
The radiolabeled hypoxanthine incorporation assay (Table 1) has been used as the gold standard in P. falciparum drug susceptibility testing18. As an alternative to using radiolabeled hypoxanthine, Elabbadi et al29 proposed ethanolamine, which has the major advantage that the culture medium can be supplemented with hypoxanthine to improve parasite growth. Several other precursors (e.g., palmitate, serine, choline, inositol and isoleucine) have also been suggested for use in isotopic assays29. However, regulations regarding the handling of radioactive materials have become considerably more restrictive, aiming to limit the application to domains where there is no alternative methodology available. Another limitation is the high purchase cost of the necessary equipment, such as liquid scintillation counters and harvesting machines. In addition, the relatively high parasite densities required for this test (approximately 0.5%) limits its application to the use of culture-adapted parasite strains or field samples with adequately high parasitemia. These limitations make it costly and difficult to routinely use in research and clinical settings, particularly in resource-limited environments.
Quantification of parasite proteins
A method that is simple to establish, highly reproducible, requires little technical equipment and could be applicable to a field laboratory is the ELISA. A few commercialized ELISA tests are already available, targeting P. falciparum lactate dehydrogenase (pLDH), pLDH activity or HRP230,19,31,32,33. The principle behind the use of pLDH production as a measure of parasite growth comes from the different enzyme structure of pLDH compared to human LDHs34 and the rapid decline of pLDH levels when the parasites die35.
The double-site enzyme-linked pLDH immunodetection (DELI) assay (Table 1) has been used to assess P. falciparum antimalarial drug susceptibility36,30,19. It is highly sensitive, more sensitive than the detection of P. falciparum in blood smears and equally as sensitive as PCR19. This technique can be used to measure in vitro drug sensitivity of P. falciparum with greater ease, greater speed, and simpler equipment than that required for isotopic assays. In contrast with the latter, lower parasite densities can be tested in the DELI assay (as low as 0.005%), thereby extending the number of isolates that can be investigated.
The pLDH enzyme activity assay is based on the observation that the LDH enzyme of P. falciparum has the ability to rapidly use 3-acetyl pyridine NAD (APAD) as a coenzyme in the reaction, leading to the formation of pyruvate from lactate. In contrast, human red blood cell LDH carries out this reaction at a slow rate in the presence of APAD. The development of APADH is measured, and there is a correlation between levels of parasitemia and the activity of parasite LDH37,38.
The HRP2 assay (Table 1) is based on the measurement of the increase in HRP2 concentrations in P. falciparum culture samples. There is a commercial kit available (Malaria Ag CELISA, Cellabs Pty. Ltd., Brookvale, NSW, Australia; http://www.cellabs.com.au), which takes only about 2.5 h to perform. If parasite growth is inhibited by antimalarial drugs, the inhibition is reflected in the increment of HRP2 levels and can therefore easily be quantified by antibody-mediated detection39. HRP2 has been reported to show extensive protein sequence diversity (mainly insertions) in P. falciparum isolates from geographically different areas40, and some isolates from the Amazon region of Peru even the lack of pfhrp2 gene41. Importantly, it has been demonstrated that the HRP2 protein diversity has an effect on the sensitivities of the HRP2 detection antibodies. For these reasons, there are growing concerns about its application for field studies. However, this method is widely used by the Amazon Network for the Surveillance of Antimalarial Drug Resistance (RAVREDA)* for drug susceptibility testing.
DNA dye intercalation assays
Several screening methods have been published that offer the potential for high-throughput screening of drugs against P. falciparum, and most are based on automated quantification of parasite growth after staining parasites with fluorescent DNA binding dyes42,21,43,44,23,24. The principle behind these assays is the contrast between host erythrocytes, which lack DNA and RNA, and the malaria parasites, which have both DNA and RNA; thus, parasites are readily stained with dyes that show enhanced fluorescence in the presence of nucleic acids.
DNA dye-based technologies (Table 1), such as those using DAPI (4, 6-diamidino-2-phenylindole), Pico green, YOYO-1, and SYBR Green I, have been shown to have comparable results to isotopic assays. In these assays, the fluorescence of the dyes is measured using either fluorescence microplate readers or fluorescence activated cell sorters (FACS). To date, only two of these dyes have been successfully used for high-throughput screening of existing libraries of compounds: YOYO-1 and DAPI42,24. The use of these assays has increased because they are relatively simple and inexpensive to run compared to their radioactive and ELISA counterparts.
IN VIVO ASSESSMENT OF ANTIPLASMODIAL ACTIVITY
Drug resistance in malaria has been defined as the "ability of a parasite strain to survive and/or multiply despite the administration and absorption of a drug given in doses equal to or higher than those usually recommended, but within the limits of tolerance of the subject". Another reason for failing to clear the parasite from the patient's body may be that not enough of the drug, or its active metabolite, reaches the parasite, an event that is termed therapeutic failure. Unlike drug resistance, therapeutic failure is a process directly related to the parasite and highly dependent on both the host and the properties of the drug.
Obviously, antimalarial drug discovery technologies are targeted to eliminate human disease. There is large heterogeneity in the behavior of patients and between different populations owing to gene polymorphisms, redundant targets, differently regulated pathways and environment challenges.
Follow-up of patients
In humans, the most traditional approach to determine P. falciparum susceptibility to antimalarial drugs is the assessment of the therapeutic (in vivo) response, which was originally defined by WHO in terms of parasite clearance [sensitive (S) and three increasing degrees of resistance (RI, RII, RIII)]. Because re-infection is difficult to exclude in areas with intense malaria transmission, WHO introduced a modified protocol based on clinical outcome (adequate clinical response, early treatment failure and late treatment failure) for areas with intense transmission, where parasitemia in the absence of clinical signs or symptoms is common*. Currently, the classification system extends the follow-up period to 28 days and includes different criteria for the definition of Early Treatment Failure, Late Clinical Failure and Late Parasitologic Failure, for areas with intense and low to moderate transmission†. The development of molecular tools has made it possible to distinguish incidents of recrudescence from re-infections45. All in vivo tests should be carried out with standard therapeutic doses and performed according to WHO guidelines‡. For P. vivax, criteria are different from those for P. falciparum†. The proliferation of antimalarial drug trials in the last ten years provides the opportunity to launch a concerted global surveillance effort to monitor antimalarial drug efficacy. The proposed World Antimalarial Resistance Network (WARN)§ aims to establish a comprehensive clinical database from which standardized estimates of antimalarial efficacy can be derived and monitored over time for diverse geographical and endemic regions46. The widespread adoption of such an approach will permit accurate and timely recognition of trends in drug efficacy, guiding the appropriate interventions to deal with established multidrug-resistant parasite strains and accelerate action when new strains of drug-resistant plasmodia first emerge.
Rodent models for in vivo screening of antimalarial activity
Four species of African rodent malaria parasites have been adapted for growth in laboratory mice: P. berghei, 194847; P. chabaudi, 196548; P. vinckei, 195249; and P. yoelii, 196550 (Table 2). These model parasites have become widely used primarily due to the ease of handling and maintaining rats and mice in the laboratory; additionally, their life cycle can be maintained in the laboratory through cyclical transmission, using Anopheles stephensi and A. gambiae, which are also easily maintained in the laboratory.
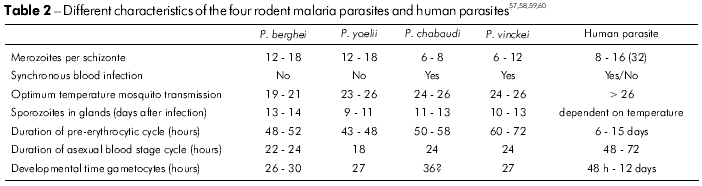
Although direct extrapolation from rodent model biology to P. falciparum biology might not be applicable in all situations, each of the four rodent malaria species has similar characteristics to the four human malaria species, making them suitable for parallel study. For example, antigenic variation in P. chabaudf51 in vivo drug testing with P. berghei52, or P. chabaudi53, cerebral malaria with P. bergei ANKA54, pre-erythrocytic stage vaccines with P. yoelii55 and chronobiology of P. vinckei56.
One of the most used in vivo dose-response tests for antimalarial activity is the four-day suppression test, originally designed by Peters61. Briefly, female mice are infected with rodent malaria parasites and the different treatment doses are given after infection on day 0 and repeated once daily for three days. On day 4 and day 7 post-infection, parasitemia is assessed. After day 7, no further manipulations are performed on the mice and they are monitored for survival.
Non-human primate, rodent or avian parasites have been used as surrogates of P. falciparum62. However, in spite of their value, there are significant biological differences between these species and the human parasites63,64. Recently, a reliable murine model of P. falciparum malaria has been established and is a valuable research tool, particularly in the field of drug discovery. The development of P. falciparum-infected humanized mice (HM) by successfully infecting nude or scid mice with P. falciparum parasites65,66,67,68,69 has facilitated the harvesting of exo-erythrocytic, intra-erythrocytic, and cytoadherent intraerythrocytic stages of the parasite. It is possible to devise new falciparum murine models using HM immunodeficient mice expressing human adhesion molecules in the endothelium that also have specific defects in phagocyte activity/activation. These tools will hopefully open up additional ways to study the biology of the erythrocytic stages of P. falciparum in vivo; further, its molecular responses to selective pressures imposed by antimalarials or the human immune system reconstituted in mice could also be examined70,71,65,72. In fact, Angulo-Barturen and collaborators73 succeeded in obtaining a dose-response relationship for antimalarials using a P. falciparum HM model.
SAFETY (CYTOTOXICITY) ASSAYS
A cytotoxicity assay is a rapid and cost-effective tool to help choose optimal candidates and to sort out likely failures before a compound is entered into the costly development process. Cytotoxicity primarily is considered as the potential of a compound to induce cell death. A reasonably good correlation has been found between basal cytotoxicity and acute toxicity in animals and humans74. Besides antimalarial activity, a promising antimalarial compound must also lack toxicity to host cells. The degree of selectivity a compound shows for the malaria parasite can be expressed by its selectivity index [SI = IC50cells/IC50 parasite (IC50, concentration that inhibits 50% of parasites)]. The higher the SI, the more promise a compound holds, due to its selectivity for the malaria parasite75. However, an SI below 2.0 indicates that a compound may possess strong antimalarial activity but may be a general toxin and should not be considered as a candidate. In practice, a drug is potentially interesting as an antimalarial if its IC50 is below 10 µM76.
Though a number of organisms can be used, in vitro cell culture methods are generally accepted as an effective method for cytotoxicity screening of new compounds. The advantages of these systems over classical methods, such as long-term studies on experimental animals like rats, mice or even the less frequent brine shrimp lethality assay77, include relatively well-controlled variables, decreased costs, a reduced time to completion, and reduced numbers of animals necessary to complete the study. There are six common targets of xenobiotic toxicity: liver, blood, kidney, nerve, lung and skin. There are many cell types available for each target: liver, HepG278 and BEL-7402; blood, HL60 and K160; kidney, Vero and HEK293; nerve, XF498 and NG108-15; lung, WI-38 and A549; skin, SK-MEL-2 and Lox. HepG2 is one of the most frequently used cell lines by pharmaceutical companies (GlaxoSmithKline79) to screen for cytotoxicity in libraries of compounds.
In works involving active anti-plasmodium compounds, the most frequently used cytotoxicity endpoints tested are based on a) the breakdown of the cellular permeability barrier (LDH leakage assay)80, b) protein content81, c) neutral red accumulation82, and d) reduced mitochondrial function [MTT assay (3-(4,5-dimethylthiazol-2-yl)-2,5-dimethyltetrazolium bromide]83. Each of these methods can be performed in microcultures, using multiple sample concentrations on 96-well or 384-well plates, thus allowing high throughput testing of large libraries of compounds.
a) The LDH leakage assay is based on the measurement of LDH activity in the extracellular medium. LDH activity is measured based on the diaphorase-mediated conversion of resazurin into the fluorescent resorufin product after LDH-catalyzed enzymatic conversion of lactate to pyruvate. The loss of intracellular LDH and its release into the culture medium is an indicator of irreversible cell death due to cell membrane damage. This assay is characterized by its reliability, speed and simple evaluation80.
b) The protein content assay is an indirect measurement of the cellular protein content of adherent and suspension cultures. It works by coloration with sul-forhodamine B (SRB) and subsequent determination of optical density (490 nm) in a microtiter plate reader. The assay provides a colorimetric endpoint test, but SRB also fluoresces strongly with laser excitation at 488 nm and can be measured quantitatively at the single-cell level by static fluorescence cytometry81.
c) The neutral red assay is based on the ability of viable cells to incorporate and bind the supravital dye neutral red in the lysosomes82. The dye is then extracted and quantified by absorbance reading using a spectrophotometer.
d) Finally, the MTT assay has been used in different cell lines to assess cell viability. The assay involves conversion of the tetrazolium salt MTT, by mitochondrial dehydrogenases in viable proliferating cells, to an insoluble purple product formazan. Amounts of formazan are then rapidly quantitated using an automated spectrophotometric microplate reader83.
ASSESSMENT OF TRANSMISSION-BLOCKING ACTIVITY OF CANDIDATE DRUGS
The rapid emergence of resistance to antimalarial drugs from diverse chemical classes* is a major factor affecting the treatment and control of malaria84. The use of compounds capable of interrupting the transmission of malaria has been advocated as a way of preventing the development of drug resistance and limiting the spread of resistant parasites85,86,87,88. Gametocytocidal antimalarials, and the artemisinines in particular, might accelerate the process of malaria control as they reduce post-treatment transmission89,90,91. However, artemisinin derivatives do not completely prevent post-treatment transmission92, and in view of emerging artemisinin combined therapy (ACT) resistance in South-East Asia93,94,95, alternative drugs effective against gametocytes are urgently needed.
Transmission-blocking antimalarial drugs can affect the sexual stages of the parasite in the human host (gametocytocidal activity) or the development of the parasite in the mosquito (sporontocidal activity).
EFFECT ON MICROGAMETOGENESIS (EXFLAGELLATION ASSAY)
Exflagellation is the process in which an activated microgametocyte produces flagellar microgametes. The microgametes avidly adhere to neighboring erythrocytes to form multicellular exflagellation centers, which can be observed microscopically96. Gametocyte activation is triggered in vitro by simultaneous exposure to two stimuli: a drop in temperature of more than 5o C97 and a pH shift from 7.5 to 8.098,99,100. The standard method to monitor successful gametogenesis is by counting exflagellation centers under the microscope. However, as this method is labor-intensive, somewhat subjective, and cannot be automated, it is not suited for medium-throughput applications. DNA replication during microgametogenesis has been previously investigated, using DNA staining with various fluorescent dyes and subsequent analysis of individual cells by fluorescence microscopy or of cell populations by flow cytometry101,102. However, these methods are not easily adapted to higher throughput assays. Recently, an assay adapted to the 96-well format to monitor activation of gametocytes based on the incorporation of radioactive hypoxanthine into the newly synthesized DNA of microgametes has been developed103.
EVALUATION OF THE IMPACT OF ANTIMALARIALS ON SPOROGONIC DEVELOPMENT
Post-treatment gametocyte carriage in humans can be examined104, despite a number of ethical considerations. Alternatively, a rodent model where the animals are infected and then treated can be used. Experimental mice are infected by intraperitoneal inoculation of infected red blood cells, and five days after infection, the presence of mature gametocytes is verified by the presence of exflagellation observed microscopically in blood drops. Gametocytemic mice are then treated with the compound to be tested and placed over cages containing mosquitoes. Ten days after the blood meal, mosquitoes are dissected and their midguts are examined under the light microscope (400x) to assess for the presence or absence of oocysts105,106. Traditionally, oocysts were visualized by staining with 0.5% mercurochrome. The development of fluorescent protein (GFP) transformed rodent parasites has made the detection of oocysts in the midgut easier because GFP-generated fluorescence can be easily and rapidly detected in a fluorescent microscope (Figure 2).
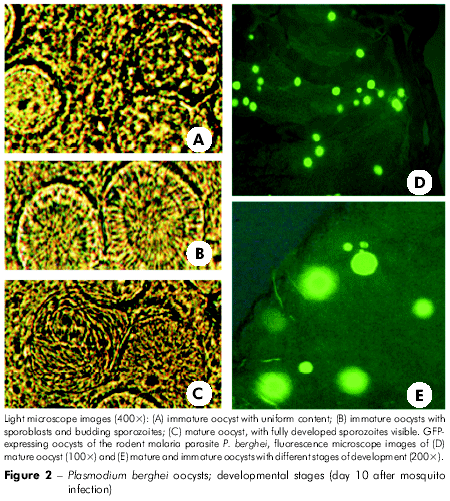
Evaluation of the impact on oocyst maturation can be established by observing the oocyst developmental status. Example categories of development include the following: a) immature oocyst, before the formation of sporoblasts; b) immature oocyst, with visible sporoblasts and budding sporozoites; and c) mature oocyst, containing fully developed sporozoites. Oocyst development can also be quantified by measuring oocyst diameter using an ocular micrometer.
Transmission-blocking activity is ultimately evaluated by removing mosquito salivary glands on day 21 postinfection and examining them for the presence or absence of sporozoites by phase-contrast microscopy86,87,88, or by allowing these mosquitoes to feed on uninfected mice and then checking for parasitemia in the mice on subsequent days.
ASSESSMENT OF ANTIMALARIAL ACTIVITY OF COMPOUNDS DURING THE HEPATIC STAGE
This stage is a link between the extracellular sporozoite, inoculated by the mosquito vector, and the blood stage, which is responsible for producing disease.
ANTIMALARIAL EXO- ERYTHROCYTIC SCHIZONTOCIDAL ASSAY
The development of pharmaceutical products inhibiting the growth of hepatic forms of Plasmodium is relevant for two main reasons. First, such compounds could be used as prophylactic agents by people exposed for a limited duration in an area where malaria is endemic (e.g., refugees and travelers) and, second, the emergence of drug-resistant strains is theoretically limited during the liver phase because of the lower parasitic load compared to the blood phase. Although hepatic stages provide attractive targets for antimalarial chemotherapy, the list of effective and widely available drugs is still limited. The only currently available prophylactic drugs are atovaquone and the two related drugs, primaquine and tafenoquine. Atovaquone has been demonstrated to be efficient in the inhibition of parasite development107, however, its use is limited because of its high cost. Hematological toxicity restricts the use of primaquine, particularly in Africa because of the elevated frequency of Glucose-6-phosphate dehydrogenase (G6PD) deficiency in the human population108. The identification of new drugs is slowed down by the lack of a reliable and sensitive method that can be utilized in high-throughput screening.
Despite the significant advances, the exo-erythrocytic (EE) stages of mammalian malarial parasites remain the east understood of all stages of the parasite's life cycle. The earliest and most prolific cultures of EE stages were achieved using the avian parasites P. fallax, P. gallinaceum and P. cathemerium, cultured in chicken embryos or in vitro109,110,111,112. However, the biological disparity between the EE cycle in avian and mammalian malarial parasites, the differences in host immune systems, and the role of the hepatocyte greatly limit the use of the culture system as a model for human malaria. Some laboratories have pursued the use of primary hepatocytes from susceptible natural and laboratory hosts113. Primary hepatocytes have the advantage that they can be obtained from animals that are histocompatible to experimentally immunized hosts. In addition, they may allow for the testing of drugs requiring metabolic activation, although qualitative and quantitative inter-species differences commonly exist in the pathways of hepatic drug metabolism. However, primary hepatocytes also suffer from a number of major disadvantages: (1) the metabolic profile of the hepatocyte will vary during the period of culture; (2) the density of parasites obtained using these cells is relatively low; (3) for technical reasons, preparations will vary from experiment to experiment and between laboratories; and (4) the preparations will vary depending upon the nutritional status and other characteristics of the host.
Advances were made by the introduction of the hepatoma cell lines HepG2-A16 and Huh7114. However, in drug-sensitivity studies hepatoma cell lines may not faithfully reproduce drug metabolism by primary hepatocytes. For example, although HepG2 cells can perform cytochrome P-450 dependent mixed function oxidase and conjugation reactions, differences have been observed in the activities of some of the drug metabolizing enzymes as compared with freshly isolated primary human hepatocytes115,116. Primaquine, which is believed to require activation by the host cell, has a low activity against P. berghei EE forms cultured in HepG2 cells117 compared to P. yoelii grown in primary Thamnomys hepatocytes118. This may reflect the inability of HepG2 to transform primaquine into its active state. To obtain a more complete picture of the activity of individual drugs, several different culture systems should ideally be used.
The in vitro cultivation of the EE forms of P. berghei (ANKA strain) or P. yoelii (265BY strain), by infection of the HepG2 cell line with sporozoites produced in Anopheles stephensi mosquitoes119,78, constitutes one of the most used assays to assess the antimalarial activity of compounds during the hepatic stage.
In vitro drug sensitivity assays are largely based on counting the number of hepatic cells containing schizonts in sporozoite-infected cultures120,113,114. Although alternative methods have been proposed to detect exo-erythrocytic schizonts including the use of quantitative RT-PCR (qRT-PCR) methods121,122,123, the use of ribosomal RNA probes124, the use of infrared imaging system combined with a colony counter125, and detection in culture and in live mice using real-time luminescence imaging126, the number of infected cells is still largely measured by fluorescence microscopy analysis. These methods are not only prone to large variations between observers but are also time consuming given the low infection rates (generally less than 2%) observed in cultured hepatocytes125. Transgenic rodent malaria parasites (GFP-expressing parasites) expressing the fluorescent reporter protein luciferase have been developed127,128. The luminescence-based analysis of the effects of various drugs on in vitro hepatocyte infection using these parasites shows that these models can effectively be used for in vitro and in vivo screening of compounds targeting Plasmodium liver stages129. This strategy is a reproducible system in which a cloned parasite grows to maturity in a defined host cell, at the same rate and with the same morphology as that observed in a convenient laboratory host. Further, it represents a routine and reproducible assay for the growth of EE stages, which is both objective and statistically robust.
THE ORDEAL OF CHOICE
The abundance of available drug-sensitivity assays obviously leaves the researcher with the difficulty of choice. Both the WHO microtest and the isotopic assays have proven their reliability in their respective disciplines after more than two decades of use; the WHO assay in the field, the isotopic assay in well-equipped research laboratories. However, both assays are associated with serious drawbacks. The WHO assay is both subjective and labor-intensive, whereas the isotopic assay lacks sensitivity and involves the handling of radioactive material. The new colorimetric assays, the ELISA-based DELI and HRP2 assays and the DNA dye intercalation assays overcome many of these disadvantages and might currently be the methods of choice. They are sensitive, relatively simple and rapid, do not require highly specialized equipment, and have the potential to replace traditional assays. However, they are still a long way from the 'drug resistance dipstick'. As more information on the genetic background of resistance becomes available, and as new and improved tools (such as real-time PCR) are developed, molecular techniques might become an extremely valuable tool for the surveillance and early detection of antimalarial drug resistance in the future.
Drug assays are fundamental to malaria research and should always be complemented with clinical studies, host genetic studies related to drug metabolism, molecular markers for the quicker identification of presence of resistance in the field, and new diagnostic methods mainly applied to drug susceptibility characterization.
ACKNOWLEDGMENTS
The authors would like to acknowledge the many valuable suggestions made by Dr. Marcelo Silva (PhD), researcher at Centro de Malária e Outras Doenças Tropicais LA, Universidade Nova de Lisboa, Lisbon, Portugal.
REFERENCES
1 Mota MM, Rodriguez A. Invasion of mammalian host cells by Plasmodium sporozoites. Bioessays. 2002 Feb;24(2):149-56. [ Links ]
2 Ng OT, Ooi EE, Lee CC, Lee PJ, Ng LC, Pei SW, et al. Naturally acquired human Plasmodium knowlesi infection, Singapore. Emerg Infect Dis. 2008 May;14(5):814-6. [ Links ]
3 Carlton JM, Muller R, Yowell CA, Fluegge MR, Sturrock KA, Pritt JR, et al. Plasmodium falciparum: studies on mature exoerythrocytic forms in the liver of the chimpanzee, Pan troglodytes. Exp Parasitol. 1990 Jan;70(1):1-11. DOI:10.1016/0014-4894(90)90080-V [ Links ]
4 Evans KJ, Hansen DS, van Rooijen N, Buckingham LA, Schofield L. Severe malarial anemia of low parasite burden in rodent models results from accelerated clearance of uninfected erythrocytes. Blood. 2006 Feb;107(3):1192-9. DOI:10.1182/blood-2005-08-3460 [ Links ]
5 Sanni LA, Fonseca LF, Langhorne J. Mouse models for erythrocytic-stage malaria. Methods Mol Med. 2002;72:57-76. DOI:10.1385/1-59259-271-6:57 [ Links ]
6 Stevenson MM, Gros P, Olivier M, Fortin A, Serghides L. Cerebral malaria: human versus mouse studies. Trends Parasitol. 2010 Jun;26(6):274-5. DOI:10.1016/J.PT.2010.03.008 [ Links ]
7 Carlton JM, Muller R, Yowell CA, Fluegge MR, Sturrock KA, Pritt JR, et al. Profiling the malaria genome: a gene survey of three species of malaria parasite with comparison to other apicomplexan species. Mol Biochem Parasitol. 2001 Dec;118(2):201-10. DOI:10.1016/S0166-6851(01)00371-1 [ Links ]
8 Hernandez-Valladares M, Rihet P, ole-MoiYoi OK, Iraqi FA. Mapping of a new quantitative trait locus for resistance to malaria in mice by a comparative mapping approach with human Chromosome 5q31-q33. Immunogenetics. 2004 May;56(2):115-7. DOI 10.1007/s00251-004-0667-0 [ Links ]
9 Walliker D. Ronald Ross: malariologist and polymath. A biography. Parasitol Today. 1998 May;14(5):210. DOI:10.1016/S0169-4758(98)01230-7
10 Haynes JD, Diggs CL, Hines FA, Desjardins RE. Culture of human malaria parasites Plasmodium falciparum. Nature. 1976 Oct;263(5580):767-9. DOI:10.1038/263767a0 [ Links ]
11 Trager W, Jensen JB. Human malaria parasites in continuous culture. Science. 1976 Aug;193(4254):673-5. DOI:10.1126/science.781840 [ Links ]
12 Diggs C, Joseph K, Flemmings B, Snodgrass R, Hines F. Protein synthesis in vitro by cryopreserved Plasmodium falciparum. Am J Trop Med Hyg. 1975 Sep;24(5):760-3. [ Links ]
13 Rieckmann KH, Campbell GH, Sax LJ, Mrema JE. Drug sensitivity of Plasmodium falciparum: an in-vitro microtechnique. Lancet. 1978 Jun;1(8054):22-3.
14 Basco LK, Le Bras J. In vitro susceptibility of Cambodian isolates of Plasmodium falciparum to halofantrine, pyrona rid in e an d a rtemi sin in derivatives. Ann Trop Med Parasitol. 1994 Apr;88(2):137-44. [ Links ]
15 Russell BM, Udomsangpetch R, Rieckmann KH, Kotecka BM, Coleman RE, Sattabongkot J. Simple in vitro assay for determining the sensitivity of Plasmodium vivax isolates from fresh human blood to antimalarials in areas where P. vivax is endemic. Antimicrob Agents Chemother. 2003 Jan;47(1):170-3. DOI:10.1128/AAC.47.1.170-173.2003 [ Links ]
16 Wernsdorfer WH, Kouznetsov RL. Drug-resistant malaria: occurrence, control, and surveillance. Bull World Health Organ. 1980;58(3):341-52. [ Links ]
17 Chulay JD, Haynes JD, Diggs CL. Plasmodium falciparum: assessment of in vitro growth by [3H] hypoxanthine incorporation. Exp Parasitol. 1983 Feb;55(1):138-46. DOI:10.1016/0014-4894(83)90007-3 [ Links ]
18 Desjardins RE, Canfield CJ, Haynes JD, Chulay JD. Quantitative assessment of antimalarial activity in vitro by a semiautomated microdilution technique. Antimicrob Agents Chemother. 1979 Dec;16(6):710-8. [ Links ]
19 Druilhe P, Moreno A, Blanc C, Brasseur PH, Jacquier P. A colorimetric in vitro drug sensitivity assay for Plasmodium falciparum based on a highly sensitive double-site lactate dehydrogenase antigen-capture enzyme-linked immunosorbent assay. Am J Trop Med Hyg. 2001 May-Jun;64(5-6):233-41. [ Links ]
20 Noedl H, Wernsdorfer WH, Miller RS, Wongsrichanalai C. Histidine-rich protein II: a novel approach to malaria drug sensitivity testing. Antimicrob Agents Chemother. 2002 Jun;46(6):1658-64. [ Links ]
21 Corbett Y, Herrera L, Gonzalez J, Cubilla L, Capson TL, Coley PD, et al. A novel DNA-based microfluorimetric method to evaluate antimalarial drug activity. Am J Trop Med Hyg. 2004 Feb;70(2):119-24. [ Links ]
22 Johnson JD, Dennull RA, Gerena L, Lopez-Sanchez M, Roncal NE, Waters NC. Assessment and continued validation of the MSF assay for use in malaria drug screening. Antimicrob Agents Chemother. 2007 Jun;51(6):1926-33. DOI:10.1128/AAC.01607-06 [ Links ]
23 Smilkstein M, Sriwilaijaroen N, Kelly JX, Wilairat P, Riscoe M. Simple and inexpensive fluorescence-based technique for high-throughput antimalarial drug screening. Antimicrob Agents Chemother. 2004 May;48(5):1803-6. DOI:10.1128/AAC.48.5.1803-1806.2004 [ Links ]
24 Weisman JL, Liou AP, Shelat AA, Cohen FE, Guy RK, DeRisi JL. Searching for new antimalarial therapeutics amongst known drugs. Chem Biol Drug Des. 2006 Jun;67(6):409-16. DOI:10.1111/j.1747-0285.2006.00391.x [ Links ]
25 Best Plummer W, Pinto Pereira L. Diminished Plasmodium falciparum sensitivity to quinine exposure in vitro and in a sequential multi-drug regimen: a preliminary investigation in Guyana, South America. Int J Infect Dis. 2008 Nov;12(6):e27-31. DOI:10.1016/j.ijid.2008.03.024 [ Links ]
26 Calvosa VS, Adagu IS, Povoa MM. Plasmodium falciparum: emerging mefloquine resistance in vitro in Para State, north Brazil. Trans R Soc Trop Med Hyg. 2001 May-Jun;95(3):330-1.
27 Ferreira ID, Martinelli A, Rodrigues LA, Carmo EL, Rosário VE, Póvoa MM, et al. Plasmodium falciparum from Para State (Brazil) shows satisfactory in vitro response to artemisinin derivatives and absence of the S769N mutation in the SERCA-type PfATPase6. Trop Med Int Health. 2008 Feb;13(2):199-207. [ Links ]
28 Le Bras J, Deloron P, Hartmann JF, Coulanges P, Dourado HV, Larouze B. Application of an in vitro semi-microtest to the study of drug sensitivity of 66 Plasmodium falciparum isolates from 15 countries. Trans R Soc Trop Med Hyg. 1984;78(4):485-8. [ Links ]
29 Elabbadi N, Ancelin ML, Vial HJ. Use of radioactive ethanolamine incorporation into phospholipids to assess in vitro antimalarial activity by the semiautomated microdilution technique. Antimicrob Agents Chemother. 1992 Jan;36(1):50-5. [ Links ]
30 Brockman A, Singlam S, Phiaphun L, Looareesuwan S, White NJ, Nosten F. Field evaluation of a novel colorimetric method--double-site enzyme-linked lactate dehydrogenase immunodetection assay--to determine drug susceptibilities of Plasmodium falciparum clinical isolates from northwestern Thailand. Antimicrob Agents Chemother. 2004 Apr;48(4):1426-9. DOI:10.1128/AAC.48.4.1426-1429.2004 [ Links ]
31 Kaddouri H, Nakache S, Houze S, Mentre F, Le Bras J. Assessment of the drug susceptibility of Plasmodium falciparum clinical isolates from Africa by using a Plasmodium lactate dehydrogenase immunodetection assay and an inhibitory maximum effect model for precise measurement of the 50-percent inhibitory concentration. Antimicrob Agents Chemother. 2006 Oct;50(10):3343-9. DOI:10.1128/AAC.00367-06 [ Links ]
32 Noedl H, Attlmayr B, Wernsdorfer WH, Kollaritsch H, Miller RS. A histidine-rich protein 2-based malaria drug sensitivity assay for field use. Am J Trop Med Hyg. 2004 Dec;71(6):711-4. [ Links ]
33 Noedl H, Wernsdorfer WH, Kollaritsch H, Looareesuwan S, Miller RS, Wongsrichanalai C. Malaria drug-susceptibility testing. HRP2-based assays: current data, future perspectives. Wien Klin Wochenschr. 2003;115 Suppl 3:23-7. [ Links ]
34 Brown WM, Yowell CA, Hoard A, Vander Jagt TA, Hunsaker LA, Deck LM, et al. Comparative structural a nalysis and kin eti c properties of l actate dehydrogenases from the four species of human malarial parasites. Biochemistry. 2004 May;43(20):6219-29. DOI: 10.1021/bi049892w [ Links ]
35 Piper KP, Roberts DJ, Day KP. Plasmodium falciparum: analysis of the antibody specificity to the surface of the trophozoite-infected erythrocyte. Exp Parasitol. 1999 Feb;91(2):161-9. DOI:10.1006/EXPR.1998.4368 [ Links ]
36 Barends M, Jaidee A, Khaohirun N, Singhasivanon P, Nosten F. In vitro activity of ferroquine (SSR 97193) against Plasmodium falciparum isolates from the Thai-Burmese border. Malar J. 2007 Jun;6:81. DOI:10.1186/1475-2875-6-81 [ Links ]
37 Basco LK, Marquet F, Makler MM, Le Bras J. Plasmodium falciparum and Plasmodium vivax: lactate dehydrogenase activity and its application for in vitro drug susceptibility assay. Exp Parasitol. 1995 Mar;80(2):260-71.![]() DOI:10.1006/Expr.1995.1032 [ Links ]
DOI:10.1006/Expr.1995.1032 [ Links ]
38 Makler MT, Ries JM, Williams JA, Bancroft JE, Piper RC, Gibbins BL, et al. Parasite lactate dehydrogenase as an assay for Plasmodium falciparum drug sensitivity. Am J Trop Med Hyg. 1993 Jun;48(6):739-41. [ Links ]
39 Noedl H, Bronnert J, Yingyuen K, Attlmayr B, Kollaritsch H, Fukuda M. Simple histidine-rich protein 2 double-site sandwich enzyme-linked immunosorbent assay for use in malaria drug sensitivity testing. Antimicrob Agents Chemother. 2005 Aug;49(8):3575-7. DOI:10.1128/AAC.49.8.3575-3577.2005 [ Links ]
40 Baker J, McCarthy J, Gatton M, Kyle DE, Belizario V, Luchavez J, et al. Genetic diversity of Plasmodium falciparum histidine-rich protein 2 (PfHRP2) and its effect on the performance of PfHRP2-based rapid diagnostic tests. J Infect Dis. 2005 Sep;192(5):870-7. DOI:10.1086/432010 [ Links ]
41 Gamboa D, Ho MF, Bendezu J, Torres K, Chiodini PL, Barnwell JW, et al. A large proportion of P. falciparum isolates in the Amazon region of Peru lack pfhrp2 and pfhrp3: implications for malaria rapid diagnostic tests. PLoS One. 2010 Jan;5(1):e8091. DOI:10.1371/journal.pone.0008091 [ Links ]
42 Baniecki ML, Wirth DF, Clardy J. High-throughput Plasmodium falciparum growth assay for malaria drug discovery. Antimicrob Agents Chemother. 2007 Feb;51(2):716-23. DOI:10.1128/AAC.01144-06 [ Links ]
43 Johnson JD, Dennull RA, Gerena L, Lopez-Sanchez M, Roncal NE, Waters NC. Assessment and continued validation of the malaria SYBR green I-based fluorescence assay for use in malaria drug screening. Antimicrob Agents Chemother. 2007 Jun;51(6):1926-33. Doi:10.1128/AAC.01607-06 [ Links ]
44 Madrid PB, Wilson NT, DeRisi JL, Guy RK. Parallel synthesis and antimalarial screening of a 4-aminoquinoline library. J Comb Chem. 2004 May-Jun;6(3):437-42. DOI: 10.1021/cc0340473 [ Links ]
45 Snounou G, Beck HP. The use of PCR genotyping in the assessment of recrudescence or reinfection after antimalarial drug treatment. Parasitol Today. 1998 Nov;14(11):462-7. DOI:10.1016/S0169-4758(98)01340-4 [ Links ]
46 Price RN, Dorsey G, Ashley EA, Barnes KI, Baird JK, Guerin PJ, et al. World Antimalarial Resistance Network I: clinical efficacy of antimalarial drugs. Malar J. 2007 Sep;6:119. DOI:10.1186/1475-2875-6-119 [ Links ]
47 van den Berghe L, Vincke I, Chardome M. Tissular phase of Plasmodium berghei. Ann Soc Belg Med Trop (1920). 1950 Mar;30(1):79-82.
48 Landau I, Chabaud AG. Natural infection by 2 plasmodia of the rodent Thamnomys rutilans in the Central African Republic. C R Acad Sci Hebd Seances Acad Sci D. 1965 Jun;261(1):230-2. [ Links ]
49 Bafort JM. The biology of rodent malaria with particular reference to Plasmodium vinckei vinckei Rodhain 1952. Ann Soc Belges Med Trop Parasitol Mycol. 1971 May;51(1):5-203.
50 Landau I, Chabaud AG. Secondary hepatic schizonts in spontaneous malaria in rodents. C R Acad Sci Hebd Seances Acad Sci D. 1968 Apr;266(17):1730-3. [ Links ]
51 Phillips RS, Brannan LR, Balmer P, Neuville P. Antigenic variation during malaria infection: the contribution from the murine parasite Plasmodium chabaudi. Parasite Immunol. 1997 Sep;19(9):427-34. [ Links ]
52 van Vianen PH, van Engen A, Thaithong S, van der Keur M, Tanke HJ, van der Kaay HJ, et al. Flow cytometric screening of blood samples for malaria parasites. Cytometry. 1993;14(3):276-80. [ Links ]
53 Ancelin ML, Calas M, Bonhoure A, Herbute S, Vial HJ. In vivo antimalarial activities of mono- and bis quaternary ammonium salts interfering with Plasmodium phospholipid metabolism. Antimicrob Agents Chemother. 2003 Aug;47(8):2598-605. DOI: 10.1128/AAC.47.8.2598-2605.2003 [ Links ]
54 Bagot S, Nogueira F, Collette A, Rosario V, Lemonier F, Cazenave PA, et al. Comparative study of brain CD8+ T cells induced by sporozoites and those induced by blood-stage Plasmodium berghei ANKA involved in the development of cerebral malaria. Infect Immun. 2004 May;72(5):2817-26. DOI:10.1128/IAI.72.5.2817-2826.2004 [ Links ]
55 Becker SI, Wang R, Hedstrom RC, Aguiar JC, Jones TR, Hoffman SL, et al. Protection of mice against Plasmodium yoelii sporozoite challenge with P. yoelii merozoite surface protein 1 DNA vaccines. Infect Immun. 1998 Jul;66(7):3457-61. [ Links ]
56 Gautret P, Chabaud AG, Landau I. Plasmodium vinckei vinckei and P. yoelii nigeriensis: pattern of gametocyte production and development. Parassitologia. 1995 Apr;37(1):17-24. [ Links ]
57 Carter R, Beach RF. Gametogenesis in culture by gametocytes of Plasmodium falciparum. Nature. 1977 Nov;270(5634):240-1.
58 Landau I, Chabaud AG. Latency of Plasmodium merozoites and drug-resistance: a review. Parasite. 1994 Jun;1(2):105-14. [ Links ]
59 Killick-Kendrick R, Rajapaksa N, Peters W, Garnham PC, Cheong WH, Cadigan FC. Malaria parasites of the orang-utan. Trans R Soc Trop Med Hyg. 1973;67(1):1-2.
60 Landau I, Boulard Y, Seureau C, Peters W. Retarded schizogony of Plasmodium yoelii yoelii in rodents treated with ethionine or subjected to a methionine deficiency: histological and ultrastructural studies. Ann Parasitol Hum Comp. 1982;57(1):1-20. [ Links ]
61 Peters W. The chemotherapy of rodent malaria: XXII. The value of drug-resistant strains of P. berghei in screening for blood schizontocidal activity. Ann Trop Med Parasitol. 1975 Jun;69(2):155-71. [ Links ]
62 Fidock DA, Rosenthal PJ, Croft SL, Brun R, Nwaka S. Antimalarial drug discovery: efficacy models for compound screening. Nat Rev Drug Discov. 2004 Jun;3(6):509-20.DOI:10.1038/nrd1416 [ Links ]
63 Carlton JM, Angiuoli SV, Suh BB, Kooij TW, Pertea M, Silva JC, et al. Genome sequence and comparative analysis of the model rodent malaria parasite Plasmodium yoelii yoelii. Nature. 2002 Oct;419(6906):512-9. DOI:10.1038/nature01099 [ Links ]
64 Gautret P, Coquelin F, Mora-Silvera E, Chabaud AG, Landau I. Chronosexuality of Plasmodium species of Central African Muridae. Parassitologia. 1998 Sep;40(3):255-8. [ Links ]
65 Moore JM, Kumar N, Shultz LD, Rajan TV. Maintenance of the human malarial parasite, Plasmodium falciparum, in scid mice and transmission of gametocytes to mosquitoes. J Exp Med. 1995 Jun;181(6):2265-70.
66 Moreno A, Badell E, Van Rooijen N, Druilhe P. Human malaria in immunocompromised mice: new in vivo model for chemotherapy studies. Antimicrob Agents Chemother. 2001 Jun;45(6):1847-53. DOI:10.1128/AAC.45.6.1847-1853.2001 [ Links ]
67 Moreno A, Ferrer E, Arahuetes S, Eguiluz C, Van Rooijen N, Benito A. The course of infections and pathology in immunomodulated NOD/LtSz-SCID mice inoculated with Plasmodium falciparum laboratory lines and clinical isolates. Int J Parasitol. 2006 Mar;36(3):361-9. DOI:10.1016/J.IJPARA.2005.10.012 [ Links ]
68 Moreno Sabater A, Moreno M, Moreno FJ, Eguiluz C, van Rooijen N, Benito A. Experimental infection of immunomodulated NOD/LtSz-SCID mice as a new model for Plasmodium falciparum erythrocytic stages. Parasitol Res. 2005 Jan;95(2):97-105. DOI:10.1007/s00436-004-1249-7 [ Links ]
69 Tsuji M, Ishihara C, Arai S, Hiratai R, Azuma I. Establishment of a SCID mouse model having circulating human red blood cells and a possible growth of Plasmodium falciparum in the mouse. Vaccine. 1995;13(15):1389-92. DOI:10.1016/0264-410X(95)00081-B [ Links ]
70 Jimenez-Diaz MB, Mulet T, Viera S, Gomez V, Garuti H, Ibanez J, et al. Improved murine model of malaria using Plasmodium falciparum competent strains and non-myelodepleted NOD-scid IL2Rgammanull mice engrafted with human erythrocytes. Antimicrob Agents Chemother. 2009 Oct;53(10):4533-6. DOI:10.1128/AAC.00519-09 [ Links ]
71 Legrand N, Ploss A, Balling R, Becker PD, Borsotti C, Brezillon N, et al. Humanized mice for modeling human infectious disease: challenges, progress, and outlook. Cell Host Microbe. 2009 Jul;6(1):5-9. DOI:10.1016/J.CHOM.2009.06.006 [ Links ]
72 Moreno JE, Rubio-Palis Y, Paez E, Perez E, Sanchez V. Abundance, biting behaviour and parous rate of anopheline mosquito species in relation to malaria incidence in gold-mining areas of southern Venezuela. Med Vet Entomol. 2007 Dec;21(4):339-49.
73 Angulo-Barturen I, Jimenez-Diaz MB, Mulet T, Rullas J, Herreros E, Ferrer S, et al. A murine model of falciparum-malaria by in vivo selection of competent strains in non-myelodepleted mice engrafted with human erythrocytes. PLoS One. 2008 May;3(5):e2252. DOI:10.1371/journal.pone.0002252 [ Links ]
74 Clemedson C, Kolman A, Forsby A. The integrated acute systemic toxicity project (ACuteTox) for the optimisation and validation of alternative in vitro tests. Altern Lab Anim. 2007 Mar;35(1):33-8. [ Links ]
75 Jonville MC, Kodja H, Humeau L, Fournel J, De Mol P, Cao M, et al. Screening of medicinal plants from Reunion Island for antimalarial and cytotoxic activity. J Ethnopharmacol. 2008 Dec;120(3):382-6. [ Links ]
76 Mahmoudi N, Julian-Ortiz JV, Ciceron L, Galvez J, Mazier D, Danis M, et al. Identification of new antimalarial drugs by linear discriminant analysis and topological virtual screening. J Antimicrob Chemother. 2006 Mar;57(3):489-97. DOI:10.1093/JAC/DKI470 [ Links ]
77 Solis PN, Wright CW, Anderson MM, Gupta MP, Phillipson JD. A microwell cytotoxicity assay using Artemia salina (brine shrimp). Planta Med. 1993 Jun;59(3):250-2. DOI:10.1055/s-2006-959661 [ Links ]
78 Hollingdale MR, Leland P, Schwartz AL. In vitro cultivation of the exoerythrocytic stage of Plasmodium berghei in a hepatoma cell line. Am J Trop Med Hyg. 1983 Jul;32(4):682-4. [ Links ]
79 Gamo FJ, Sanz LM, Vidal J, Cozar C, Alvarez E, Lavandera JL, et al. Thousands of chemical starting points for antimalarial lead identification. Nature. 2010 May;465(7296):305-10. DOI:10.1038/nature09107 [ Links ]
80 Decker T, Lohmann-Matthes ML. A quick and simple method for the quantitation of lactate dehydrogenase release in measurements of cellular cytotoxicity and tumor necrosis factor (TNF) activity. J Immunol Methods. 1988 Nov;115(1):61-9. [ Links ]
81 Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Warren JT, et al. New colorimetric cytotoxicity assay for anticancer-drug screening. J Nat Cancer Inst. 1990 Jul;82(13):1107-12. [ Links ]
82 Zhang SZ, Lipsky MM, Trump BF, Hsu IC. Neutral red (NR) assay for cell viability and xenobiotic-induced cytotoxicity in primary cultures of human and rat hepatocytes. Cell Biol Toxicol. 1990 Apr;6(2):219-34. [ Links ]
83 Pieters R, Huismans DR, Leyva A, Veerman AJ. Adaptation of the rapid automated tetrazolium dye based (MTT) assay for chemosensitivity testing in childhood leukemia. Cancer Lett. 1988 Aug;41(3):323-32. [ Links ]
84 Wongsrichanalai C, Varma JK, Juliano JJ, Kimerling ME, MacArthur JR. Extensive drug resistance in malaria and tuberculosis. Emerg Infect Dis. 2010 Jul;16(7):1063-7. [ Links ]
85 Butcher GA. Antimalarial drugs and the mosquito transmission of Plasmodium. Int J Parasitol. 1997 Sep;27(9):975-87. DOI:10.1016/S0020-7519(97)00079-9 [ Links ]
86 Coleman RE . Sporontocidal activity of the antimalarial WR-238605 against Plasmodium berghei ANKA in Anopheles stephensi. Am J Trop Med Hyg. 1990 Mar;42(3):196-205. [ Links ]
87 Coleman RE, Nath AK, Schneider I, Song GH, Klein TA, Milhous WK. Prevention of sporogony of Plasmodium falciparum and P. berghei in Anopheles stephensi mosquitoes by transmission-blocking antimalarials. Am J Trop Med Hyg. 1994 May;50(5):646-53. [ Links ]
88 Coleman RE, Polsa N, Eikarat N, Kollars Jr TM, Sattabongkot J. Prevention of sporogony of Plasmodium vivax in Anopheles dirus mosquitoes by transmission-blocking antimalarials. Am J Trop Med Hyg. 2001 Sep;65(3):214-8. [ Links ]
89 Garner P, Graves PM. The benefits of artemisinin combination therapy for malaria extend beyond the individual patient. PLoS Med. 2005 Apr;2(4):e105. DOI:10.1371/journal.pmed.0020105 [ Links ]
90 Price RN, Nosten F, Luxemburger C, Paiphun L, Chongsuphajaisiddhi T, White NJ, et al. Effects of artemisinin derivatives on malaria transmissibility. Lancet. 1996 Jun;347(9016):1654-8. [ Links ]
91 Sutherland CJ, Ord R, Dunyo S, Jawara M, Drakeley CJ, Pinder M, et al. Reduction of malaria transmission to Anopheles mosquitoes with a six-dose regimen of co-artemether. PLoS Med. 2005 Apr;2(4):e92. DOI:10.1371/JOURNAL.PMED.0020092 [ Links ]
92 Targett G, Drakeley C, Jawara M, von Seidlein L, Coleman R, Deen J, et al. Artesunate reduces but does not prevent posttreatment transmission of Plasmodium falciparum to Anopheles gambiae. J Infect Dis. 2001;183(8):1254-9. DOI:10.1086/319689 [ Links ]
93 Duffy PE, Sibley CH. Are we losing artemisinin combination therapy already? Lancet. 2005 Dec;366(9501):1908-9. DOI:10.1016/S0140-6736(05)67768-9 [ Links ]
94 Jambou R, Legrand E, Niang M, Khim N, Lim P, Volney B, et al. Resistance of Plasmodium falciparum field isolates to in-vitro artemether and point mutations of the SERCA-type PfATPase6. Lancet. 2005 Dec;366(9501):1960-3. DOI:10.1016/S0140-6736(05)67787-2 [ Links ]
95 Noedl H, Krudsood S, Leowattana W, Tangpukdee N, Thanachartwet W, Looareesuwan S, et al. In vitro antimalarial activity of azithromycin, artesunate, and quinine in combination and correlation with clinical outcome. Antimicrob Agents Chemother. 2007 Feb;51(2):651-6. DOI:10.1128/AAC.01023-06 [ Links ]
96 Pradel G. Proteins of the malaria parasite sexual stages: expression, function and potential for transmission blocking strategies. Parasitology. 2007 Dec;134(Pt 14):1911-29. DOI:10.1017/S0031182007003381 [ Links ]
97 Sinden RE. The cell biology of sexual development in Plasmodium. Parasitology. 1983 Apr;86(Pt 4):7-28. [ Links ]
98 Billker O, Lindo V, Panico M, Etienne AE, Paxton T, Dell A, et al. Identification of xanthurenic acid as the putative inducer of malaria development in the mosquito. Nature. 1998 Mar;392(6673):289-92. DOI:10.1038/32667 [ Links ]
99 Garcia GE, Wirtz RA, Barr JR, Woolfitt A, Rosenberg R. Xanthurenic acid induces gametogenesis in Plasmodium, the malaria parasite. J Biol Chem. 1998 May;273(20):12003-5. [ Links ]
100 Nijhout MM, Carter R. Gamete development in malaria parasites: bicarbonate-dependent stimulation by pH in vitro. Parasitology. 1978 Feb;76(1):39-53.
101 Billker O, Dechamps S, Tewari R, Wenig G, Franke-Fayard B, Brinkmann V. Calcium and a calcium-dependent protein kinase regulate gamete formation and mosquito transmission in a malaria parasite. Cell. 2004 May;117(4):503-14. DOI:10.1016/S0092-8674(04)00449-0 [ Links ]
102 Tewari R, Ogun SA, Gunaratne RS, Crisanti A, Holder AA. Disruption of Plasmodium berghei merozoite surface protein 7 gene modulates parasite growth in vivo. Blood. 2005 Jan;105(1):394-6. DOI 10.1182/blood-2004-06-2106 [ Links ]
103 Raabe AC, Billker O, Vial HJ, Wengelnik K. Quantitative assessment of DNA replication to monitor microgametogenesis in Plasmodium berghei. Mol Biochem Parasitol. 2009 Dec;168(2):172-6.![]() DOI:10.1016/J.MOLBIOPARA.2009.08.004 [ Links ]
DOI:10.1016/J.MOLBIOPARA.2009.08.004 [ Links ]
104 Zoungrana A, Coulibaly B, Sie A, Meissner P, Mockenhaupt FP, Kouyaté B, et al. Safety and efficacy of methylene blue combined with artesunate or amodiaquine for uncomplicated falciparum malaria: a randomized controlled trial from Burkina Faso. PLoS One. 2008 Feb;3(2):e1630. doi:10.1371/journal.pone.0001630 [ Links ]
105 Lucantoni L, Yerbanga RS, Lupidi G, Pasqualini L, Esposito F, Habluetzel A. Transmission blocking activity of a standardized neem (Azadirachta indica) seed extract on the rodent malaria parasite Plasmodium berghei in its vector Anopheles stephensi. Malar J. 2010 Mar;9:66. DOI:10.1186/1475-2875-9-66 [ Links ]
106 Vale N, Prudencio M, Marques CA, Collins MS, Gut J, Nogueira F, et al. Imidazoquines as antimalarial and antipneumocystis agents. J Med Chem. 2009 Dec;52(23):7800-7. DOI: 10.1021/jm900738c [ Links ]
107 Davies CS, Pudney M, Nicholas JC, Sinden RE. The novel hydroxynaphthoquinone 566C80 inhibits the development of liver stages of Plasmodium berghei cultured in vitro. Parasitology. 1993 Jan;106(Pt 1):1-6. [ Links ]
108 Shanks GD, Biomndo K, Guyatt HL, Snow RW. Travel as a risk factor for uncomplicated Plasmodium falciparum malaria in the highlands of western Kenya. Trans R Soc Trop Med Hyg. 2005 Jan;99(1):71-4. DOI:10.1016/J.TRSTMH.2004.04.001 [ Links ]
109 Beaudoin RL, Strome CP, Clutter WG. Cultivation of avian malaria parasites in mammalian liver cells. Exp Parasitol. 1974 Dec;36(3):355-9. Doi:10.1016/0014-4894(74)90075-7
110 Dubin IN, Laird RL, Drinnon VP. Further observations on the development of sporozoites of Plasmodium gallinaceum into cryptozoites in tissue culture. J Natl Malar Soc. 1950 Jun;9(2):119-27.
111 Hawking F. Growth of Protozoa in tissue culture 1. Plasmodium gallinaceum, exoerythrocytic forms. Trans R Soc Trop Med Hyg. 1945 Dec;39(3):245-63. DOI:10.1016/0035-9203(45)90026-5 [ Links ]
112 Laird RL, Dubin IN, Drinnon VP. The infection of chicks with pre-erythrocytic stages of Plasmodium gallinaceum grown in tissue culture. J Natl Malar Soc. 1950 Jun;9(2):128-31.
113 Millet P, Landau I, Baccam D, Miltgen F, Peters W. Cultivation of exo-erythrocytic schizonts of rodent Plasmodium in hepatocytes: a new experimental model for chemotherapy of malaria. C R Acad Sci III. 1985;301(8):403-6. [ Links ]
114 Sinden RE, Suhrbier A, Davies CS, Fleck SL, Hodivala K, Nicholas JC. The development and routine application of high-density exoerythrocytic-stage cultures of Plasmodium berghei. Bull World Health Organ. 1990;68 Suppl:115-25. [ Links ]
115 Dawson JR, Adams DJ, Wolf CR. Induction of drug metabolizing enzymes in human liver cell line Hep G2. FEBS Lett. 1985 Apr;183(2):219-22. [ Links ]
116 Doostdar H, Demoz A, Burke MD, Melvin WT, Grant MH. Variation in drug-metabolizing enzyme activities during the growth of human HepG2 hepatoma cells. Xenobiotica. 1990 Apr;20(4):435-41. [ Links ]
117 Millet P, Collins WE, Aikawa M, Cochrane AH, Nguyen-Dinh P. Use of non-human primate hepatocytes for in vitro study of the pre-erythrocytic stages of malaria parasites. Bull World Health Organ. 1990;68 Suppl:60-5. [ Links ]
118 Mazier D, Landau I, Miltgen F, Druilhe P, Lambiotte M, Baccam D, et al. In vitro infection of adult Thamnomys hepatocytes by sporozoites of Plasmodium yoelii: development of schizonts and release of infective merozoites. Ann Parasitol Hum Comp. 1982;57(1):99-100.
119 Gruner AC, Hez-Deroubaix S, Snounou G, Hall N, Bouchier C, Letourneur F, et al. Insights into the P. y. yoelii hepatic stage transcriptome reveal complex transcriptional patterns. Mol Biochem Parasitol. 2005 Aug;142(2):184-92. DOI:10.1016/j.molbiopara.2005.03.019 [ Links ]
120 Mahmoudi N, Ciceron L, Franetich JF, Farhati K, Silvie O, Eling W, et al. In vitro activities of 25 quinolones and fluoroquinolones against liver and blood stage Plasmodium spp. Antimicrob Agents Chemother. 2003 Aug;47(8):2636-9. DOI:10.1128/AAC.47.8.2636-2639.2003 [ Links ]
121 Bruña-Romero O, Hafalla JCR, González-Aseguinolaza G, Sano G, Tsuji M, Zavala F. Detection of malaria liver-stages in mice infected through the bite of a single Anopheles mosquito using a highly sensitive real-time PCR. Int J Parasitol. 2001 Nov;31(13):1499-502. DOI:10.1016/S0020-7519(01)00265-X [ Links ]
122 Siau A, Silvie O, Franetich JF, Yalaoui S, Marinach C, Hannoun L, et al. Temperature shift and host cell contact up-regulate sporozoite expression of Plasmodium falciparum genes involved in hepatocyte infection. PLoS Pathog. 2008 Aug;4(8):e1000121. DOI:10.1371/JOURNAL.PPAT.1000121 [ Links ]
123 Witney AA, Doolan DL, Anthony RM, Weiss WR, Hoffman SL, Carucci DJ. Determining liver stage parasite burden by real time quantitative PCR as a method for evaluating pre-erythrocytic malaria vaccine efficacy. Mol Biochem Parasitol. 2001 Dec;118(2):233-45. DOI:10.1016/S0166-6851(01)00372-3 [ Links ]
124 Li J, Wirtz RA, Schneider I, Muratova OV, McCutchan TF, Appiah A, et al. Plasmodium falciparum: stage-specific ribosomal RNA as a potential target for monitoring parasite development in Anopheles stephensi. Exp Parasitol. 1993 Feb;76(1):32-8. DOI:10.1006/EXPR.1993.1004 [ Links ]
125 Gego A, Silvie O, Franetich JF, Farhati K, Hannoun L, Luty AJ, et al. New approach for high-throughput screening of drug activity on Plasmodium liver stages. Antimicrob Agents Chemother. 2006 Apr;50(4):1586-9. DOI:10.1128/AAC.50.4.1586-1589.2006 [ Links ]
126 Tarun AS, Baer K, Dumpit RF, Gray S, Lejarcegui N, Frevert U, et al. Quantitative isolation and in vivo imaging of malaria parasite liver stages. Int J Parasitol. 2006 Oct;36(12):1283-93. DOI:10.1016/J.IJPARA.2006.06.009 [ Links ]
127 Ono K, Okajima T, Tani M, Kuroda S, Sun D, Davidson VL, et al. Involvement of a putative. [Fe-S]-cluster-binding protein in the biogenesis of quinohemoprotein amine dehydrogenase. J Biol Chem. 2006;281(19):13672-84. [ Links ]
128 Waters AP, Thomas AW, van Dijk MR, Janse CJ. Transfection of malaria parasites. Methods. 1997 Oct;13(2):134-47. DOI:10.1006/METH.1997.0506 [ Links ]
129 Ploemen IH, Prudencio M, Douradinha BG, Ramesar J, Fonager J, van Gemert GJ, et al. Visualisation and quantitative analysis of the rodent malaria liver stage by real time imaging. PLoS One. 2009 Nov;4(11):e7881. [ Links ]
 Correspondência / Correspondence / Correspondencia:
Correspondência / Correspondence / Correspondencia:
Fátima Nogueira
Instituto de Higiene e Medicina Tropical,
Universidade Nova de Lisboa
Rua da Junqueira, no 100
CEP: 1349-008 Lisboa-Portugal
Tel: 00351213652600
E-mail:fnogueira@ihmt.unl.pt
Recebido em / Received / Recibido en: 22/7/2010
Aceito em / Accepted / Aceito en: 28/9/2010
*World Health Organization. Assessment of therapeutic efficacy of antimalarial drugs for uncomplicated falciparum malaria in areas with intense transmission. Geneva: World Health Organization; 1996. Available at: http://www.who.int/drugresistance/malaria/en/Assessment_malaria_96.pdf.
*World Health Organization. Drug resistance: malaria. Available at: http://www.who.int/drugresistance/malaria/en/.
*Pan American Health Organization. RAVREDA-AMI: Amazon Network for the Surveillance of Antimalarial Drug Resistance (RAVREDA) / Amazon Malaria Initiative (AMI). Available at: http://www.paho.org/English/ad/dpc/cd/ravreda-ami.htm.
*World Health Organization. Assessment of therapeutic efficacy of antimalarial drugs for uncomplicated falciparum malaria in areas with intense transmission [Internet]. Geneva: World Health Organization; 1996. Available at: http://www.who.int/drugresistance/malaria/en/Assessment_malaria_96.pdf.
†World Health Organization. Monitoring Antimalarial Drug Resistance, Report of a WHO Consultation [Internet]. Geneva: World Health Organization; 2001. Available at: http://www.who.int/drugresistance/publications/WHO_CDS_CSR_EPH_2002_17/en/index.html.
‡World Health Organization. Drug resistance: malaria. Available at: http://www.who.int/drugresistance/malaria/en/.
§WorldWide Antimalarial Resistance Network [Internet]. 2011. Available at: http://www.wwarn.org/.
*World Health Organization. Emergence and Spread of Antimicrobial Resistance. Geneva: World Health Organization. Available at: http://www.who.int/drugresistance/AMR_Emergence_Spread/en/index.html.











 texto em
texto em 

