Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista Pan-Amazônica de Saúde
versión impresa ISSN 2176-6215versión On-line ISSN 2176-6223
Rev Pan-Amaz Saude v.2 n.3 Ananindeua sep. 2011
http://dx.doi.org/10.5123/S2176-62232011000300008
COMMUNICATION | COMUNICAÇÃO | COMUNICACIÓN
Detection of corona-like virus in a case of flaccid paraparesis in Belém, Pará State, Brazil
Detecção de virus corona-like em um caso de paraparesia flácida em Belém, Estado do Pará, Brasil
Detección de virus corona-like en un caso de paraparesia flácida en Belém, Estado de Pará, Brasil
Delana Andreza Melo BezerraI; José Antonio Picanço Diniz JúniorII; Nelson Antonio Bailão RibeiroIII; Maria de Lourdes Contente GomesIV; Alexandre da Costa LinharesIV; Ana Lucia Monteiro WanzellerIV
IMestranda do Curso de Farmácia, Universidade Federal do Pará, Belém, Brasil
IIUnidade de Microscopia Eletrônica, Instituto Evandro Chagas/SVS/MS, Belém, Pará, Brasil
IIISeção de Arbovirologia e Febres Hemorrágicas, Instituto Evandro Chagas/SVS/MS, Belém, Pará, Brasil
IVSeção de Virologia, Instituto Evandro Chagas/SVS/MS, Ananindeua, Pará, Brasil
Endereço para correspondência
Correspondence
Dirección para correspondencia
ABSTRACT
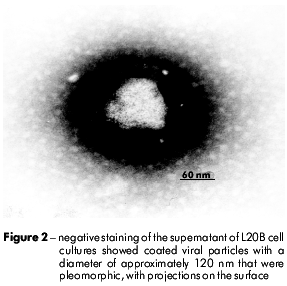
In this study, we analyzed a fecal sample of a female infant with paralysis and other clinical symptoms that resembled poliomyelitis. Negative staining electron microscopy showed viral particles with a diameter of approximately 120 nm and displaying a crown-like appearance with surface projections. Ultrathin sections showed particles budding from the membranes of the Golgi apparatus. Based on these results, we propose the association of this virus with the neurological disorder and tentatively assign it to the Coronaviridae family. Further studies are required on this proposed relationship.
Keywords: Coronaviridae; Paraparesis; Transmission Electron Microscopy; Motor Disorders.
RESUMO
Neste estudo, analisamos uma amostra fecal de criança do sexo feminino com paralisia e outros sintomas clínicos que se assemelharam à poliomielite. A microscopia eletrônica (contrastação negativa) mostrou partículas com 120 nm de diâmetro, exibindo projeções na superfície semelhantes a uma coroa. Cortes ultrafinos mostraram partículas brotando do complexo de Golgi. Com base nesses resultados, propomos a associação deste vírus com o distúrbio neurológico e o associamos provisoriamente à família Coronaviridae. Estudos adicionais são necessários para esclarecer a relação proposta.
Palavras chaves: Coronaviridae; Paraparesia; Microscopia Eletrônica de Transmissão; trastornos motores.
RESUMEN
En este estudio, analizamos una muestra fecal de niña con parálisis y otros síntomas clínicos semejantes a la poliomielitis. La microscopia electrónica (por contraste negativo) mostró partículas con 120 nm de diámetro, exhibiendo proyecciones en la superficie semejantes a una corona. Cortes ultrafinos mostraron partículas brotando del complejo de Golgi. Con base en esos resultados, proponemos la asociación de este virus al disturbio neurológico y lo asociamos provisoriamente a la familia Coronaviridae. Se necesitan estudios adicionales para aclarar la relación propuesta.
Palabras claves: Coronaviridae; Paraparesia; Microscopía Electrónica de Transmisión; trastornos motores.
INTRODUCTION
The acute flaccid paraparesis (AFP) may be associated with various etiologies, including viral infections. Coronavirus, an agent of AFP, belongs to the order Nidovirales, the family Coronaviridae and the sub-family Coronavirinae that comprises three genera: Alphacoronavirus, Betacoronavirus and Gamacoronavirus1. The origin of the term "corona" is from Latin and means "crown" because of the appearance of surface projections. The Coronaviridae family genome is non-segmented and is composed of positive-sense single-stranded RNA. The virions are spherical or pleomorphic, exhibit helical symmetry and are approximately 150 nm in diameter2.
The incubation period can vary in most cases between 1-2 weeks. Transmission occurs through aerosols or fecal-oral infection. The human symptoms can be enteric (diarrhea)2 or associated with the respiratory tract (RT). The replication of this virus may also occur in the nervous system. The laboratory diagnosis of enteroviruses can be performed with the isolation and identification of the viruses in cell cultures, serological tests or polymerase chain reaction reverse transcription (RT-PCR)3,4.
In this paper, we performed an ultrastructural identification of 89351 viral sample isolated from the fecal sample of a patient with AFP between 1998 and 2004 within the national surveillance network for acute motor deficiency (AMD).
MATERIALS AND METHODS
VIRUS ISOLATION
The sample studied was obtained through the national surveillance network for AMD, with the primary purpose of searching for the possible circulation of wild poliovirus in Brazil. A corona-like virus was analyzed from a fecal sample from a female infant with paralysis and other clinical symptoms that resembled poliomyelitis.
VIRUS INFECTION
To infect cell cultures with the corona-like virus, 1 mL of a 1:20 fecal suspension was inoculated into the center of a confluent (70% to 80%) monolayer of L-20B cells (a transgenic mouse cell line expressing the human poliovirus surface receptor). The viral particles were adsorbed to the cells for 1 h. Cell culture maintenance was performed according to Wanzeller et al5. When the cells showed a cytopathic effect (CPE), the supernatant was aliquoted and stored at -70o C.
TRANSMISSION ELECTRON MICROSCOPY
Negative staining
The procedure was previously described in Wanzeller et al5, and the sample was examined with a transmission electron microscope (TEM) (Zeiss EM 900).
Ultrathin sections
The L20B cell line was used for conventional electron microscopy. The method used was similar to that described in Wanzeller et al5. The sections were stained and observed with TEM (Zeiss EM 900).
RESULTS
CULTURE
The corona-like virus caused CPE in the L20B cell line 24 h p.i., and the cells were completely destroyed 48 h p.i., (Figure 1). The sample was negative when tested for poliovirus.

TRANSMISSION ELECTRON MICROSCOPY
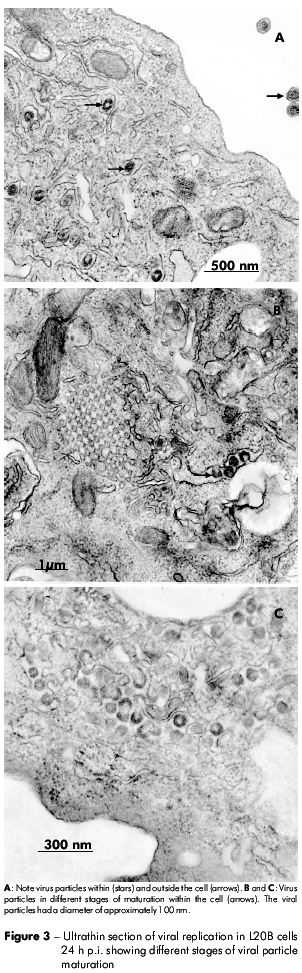
The negatively stained viral particles were approximately 120 nm in diameter. These particles had an envelope that consisted of a bilaminar membrane covered with projecting structures on the surface (Figure 2). In figure 3, different stages of viral replication can be observed.
The sample studied was tested with primers to Coronaviridae family, demonstrating a positive result by RT-PCR technique (data not shown).
DISCUSSION
A number of morphological features of this virus in L20B cells were similar to those previously described for other members of the Coronaviridae family2,6.
The budding of the gastroenteric coronavirus and severe acute respiratory syndrome coronavirus (SARS-CoV)7 in all stages of maturation has been primarily observed to be inside membranes of the Golgi structure or endoplasmic reticulum and has been considered a hallmark of members of the Coronaviridae family7,2,8,9.
The stages of viral replication observed in this study are in complete agreement with those of others coronavirus10. Lai et al2 described changes that occur during the replication process: I) the viral proteins appear as an electron-dense layer in the cytoplasm; II) viral envelope proteins adhere to vesicular membranes within the cytoplasm; and III) an electron-dense area, possibly corresponding to viral spikes, can be observed in the lumen of vesicles.
In this study, we also found these characteristics7,8, reinforcing the association of this virus with the neurological disorder presented by this patient. We have tentatively assigned this virus to the Coronaviridae family.
The association between coronaviruses and neuropathology in animals has been shown by Pasick et al11, who reported a variety of neurotropic strains that cause diseases, such as encephalitis, acute poliomyelitis and chronic inflammatory demyelination caused by mouse hepatitis virus (MHV4).
There are few reports in humans concerning the potential association of coronaviruses with neurological diseases. Among these studies, Yeh et al12 detected human coronavirus in the cerebrospinal fluid of a child who was diagnosed with disseminated acute encephalomyelitis similar to that presented in diseases such as multiple sclerosis.
CONCLUSION
Based on the viral morphology presented in this study, we provisionally proposed the inclusion of this virus in the Coronaviridae family and attributed an association of this virus with neurological diseases, such as AMD.
This is the first detection of the association of a viral particle similar to coronavirus with patients with AFP in Brazil; however, it is still not possible to confirm this association, and future studies are necessary to determine this correlation.
ACKNOWLEDGEMENTS
To Walber Victor de Moraes Pinto for help in the optical illustrations. Edna Silveira, Antonia dos Santos Alves, Euda Galiza Primo Souza, Jainara Alves and Nazaré Valadares for contribution on the laboratory techniques.
REFERENCES
1 International Committee on Taxonomy of Viruses. Virus Taxonomy. [Internet]. 2011. (citado 2012 maio 18). Disponível em: http://www.ictvonline.org/virusTaxonomy.asp?version=2011.
2 Lai MMC, Holmes KV. Coronaviridae. In: Knipe DM, Howley PM, editors. Fields virology. 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2001. p.1163-85.
3 Romero JR. Reverse-transcription polymerase chain reaction detection of the enteroviroses. Arch Pathol Lab Med. 1999 Dec;123(12):1161-9. [Link]
4 Sawyer MH. Enterovirus infections: diagnosis and treatment. Pediatr Infect Dis J. 1999 Dec;18(12):1033-9.
5 Wanzeller ALM, Diniz JAP, Gomes MLC, Cruz ACR, Soares MCP, Souza W, et al. Ultrastructural, antigenic and physicochemical characterization of the Mojuí dos Campos (Bunyavirus) isolated from bat in the Brazilian Amazon Region. Mem Inst Oswaldo Cruz. 2002 Apr;97(3):307-11. [Link]
6 Ng ML, Tan SH, See EE, Ooi EE, Ling AE. Proliferative growth of SARS coronavirus in Vero E6 cells. J Gen Virol. 2003 Dec;84(Pt 12):3291-303. [Link]
7 Goldsmith CS, Tatti KM, Ksiazek TG, Rollin PE, Comer JA, Lee WW, et al. Ultrastructural characterization of SARS coronavirus. Emerg Infect Dis. 2004 Feb;10(2):320-6.
8 Novoa RR, Calderita G, Arranz R, Fontana J, Granzow H, Risco C. Virus factories: associations of cell organelles for viral replication and morphogenesis. Biol Cell. 2005;97:147-72. DOI:10.1042/BC20040058 [Link]
9 Salanueva IJ, Carrascosa JL, Risco C. Structural maturation of the transmissible gastroenteritis coronavirus. J Virol. 1999 Oct;73(10):7952-64.
10 Snijder EJ, van der Meer Y, Zevenhoven-Dobbe J, Onderwater JJ, van der Meulen J, Koerten HK, et al. Ultrastructure and origin of membrane vesicles associated with the severe acute respiratory syndrome coronavirus replication complex. J Virol. 2006 Jun;80(12):5927-40.
11 Pasick JMM, Kalicharran K, Dales S. Distribution and tafficking of JHM coronavirus structural proteins and virions in primary neurons and the OBL-21 neuronal cell line. J Virol. 1994 May;68(5):2915-28. [Link]
12 Yeh EA, Collins A, Cohenc ME, Duffner PK, Faden H. Disseminated encephalomyelitis detection of coronavirus in the central nervous system of a child with acute. Pediatrics. 2004 Apr;113(1):73-6.
 Correspondência / Correspondence / Correspondencia:
Correspondência / Correspondence / Correspondencia:
Ana Lucia Monteiro Wanzeller
Instituto Evandro Chagas,
Laboratório de Cultivo Celular da Seção de Virologia
Rodovia BR316, km 7, s/no, Levilândia
CEP: 67030-000
Ananindeua-Pará-Brasil
E-mail: anawanzeller@iec.pa.gov.br
Received / Recebido em / Recibido en: 10/5/2011
Accepted / Aceito em / Aceito en:14/8/2011