Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista Pan-Amazônica de Saúde
versão impressa ISSN 2176-6215versão On-line ISSN 2176-6223
Rev Pan-Amaz Saude v.5 n.2 Ananindeua jun. 2014
http://dx.doi.org/10.5123/S2176-62232014000200004
ORIGINAL ARTICLE | ARTIGO ORIGINAL | ARTÍCULO ORIGINAL
Phlebotomine sand flies (Diptera: Psychodidae) in forest fragments of Belém metropolitan area, Pará State, Brazil, with considerations on vectors of American cutaneous leishmaniasis agents
Flebotomíneos (Diptera: Psychodidae) em fragmentos florestais da região metropolitana de Belém, Estado do Pará, Brasil, com considerações sobre vetores da leishmaniose tegumentar americana
Flebótomos (Diptera: Psychodidae) en fragmentos forestales de la región metropolitana de Belém, Estado de Pará, Brasil, con consideraciones sobre vectores de la leishmaniasis tegumentaria americana
João Victor Silva FerreiraI; Thiago Vasconcelos dos SantosII; Emerson Monteiro dos SantosIII; Inocêncio de Sousa GorayebI
ILaboratório de Entomologia, Coordenação de Zoologia, Museu Paraense Emílio Goeldi, Belém, Pará, Brasil
IILaboratório de Leishmanioses "Prof. Dr. Ralph Lainson", Instituto Evandro Chagas/SVS/MS, Ananindeua, Pará, Brasil
IIICampus Binacional, Universidade Federal do Amapá, Oiapoque, Amapá, Brasil
Correspondence
Endereço para correspondência
Dirección para correspondencia
ABSTRACT
In order to identify vectors of American cutaneous leishmaniasis (ACL) agents in urban forest fragments of Belém metropolitan area, Pará State, Brazil, an entomological study was conducted between 2009 and 2011 in seven ecologically isolated areas using CDC light traps. A total of 1,119 phlebotomine sand flies were collected and out of 22 species identified, six presented epidemiological importance due to their proven and/or putative implications in the transmission of ACL agents in the Brazilian Amazon. These results reveal the need of entomological surveillance in such areas.
Keywords: Psychodidae; Insect Vectors; Leishmaniasis, Cutaneous.
RESUMO
Com o objetivo de identificar agentes transmissores da leishmaniose tegumentar americana (LTA) em fragmentos florestais na área urbana da região metropolitana de Belém, Estado do Pará, Brasil, foi conduzido um estudo entre 2009 e 2011 em sete áreas ecologicamente isoladas usando armadilhas luminosas CDC. Um total de 1.119 flebotomíneos foi coletado e das 22 espécies identificadas, seis apresentaram importância epidemiológica devido as suas implicações comprovadas e/ou suspeitas na transmissão dos agentes de LTA na Amazônia brasileira. Tais resultados revelam a necessidade de vigilância entomológica nessas áreas.
Palavras-chave: Psychodidae; Insetos Vetores; Leishmaniose Cutânea.
RESUMEN
Con el objetivo de identificar agentes transmisores de la leishmaniasis tegumentaria americana (LTA) en fragmentos forestales en el área urbana de la región metropolitana de Belém, Estado de Pará, Brasil, se llevó a cabo un estudio entre 2009 y 2011 en siete áreas ecológicamente aisladas usando trampas luminosas CDC. Se colectó un total de 1.119 flebótomos e de las 22 especies identificadas, seis presentaron importancia epidemiológica debido a sus implicaciones comprobadas y/o putativas en la transmisión de los agentes de LTA en la Amazonia brasileña. Tales resultados revelan la necesidad de vigilancia entomológica en esas áreas.
Palabras clave: Psychodidae; Insectos Vectores; Leishmaniasis Cutánea.
INTRODUCTION
Phlebotomine sand flies (Diptera: Psychodidae: Phlebotominae) are group of insects of medical importance due to the fact that they are considered natural vectors of Leishmania Ross, 1903 species (Euglenozoa: Trypanosomatidae), etiological agents of leishmaniasis1,2. Of the total of 900 sand fly species described, approximately 70 have been implicated in the epidemiology of this disease3,4.
In Brazil, American visceral leishmaniasis (AVL), whose transmission only takes place by a proven phlebotomine sand fly species5,6, differs from American cutaneous leishmaniasis (ACL) that is caused by a variety of dermotropic agents and involves a high diversity of phlebotomine sand fly species with zoophilic behavior in the environment, which bite mammalian hosts. Consequently the disease presents several different enzootic transmission cycles7.
In the last ten years, an increase of ACL cases has been reported in several states of the Amazon Region8. In Pará State, the disease has been spreading in several mesoregions, including the Northeast region of Pará, where the capital Belém is located. The urbanization of ACL is unusual because of the sylvatic habits of the vectors involved1. However, the increasing adaptation of some phlebotomine species to artificial ecotopes and peridomiciliar conditions, as occurs in southeastern Brazil9, suggests that forest fragments with eco-epidemiological conditions in the center of big cities could be potential sources of human infection. In Belém, this fact was highlighted when an outbreak of ACL caused by, until that moment, an unknown parasite described as Leishmania (Viannia) lindenbergi (Silveira et al., 2002) occurred in a small forest fragment surrounded by an urban area10.
Belém metropolitan area is composed by the Municipalities of Ananindeua, Belém, Marituba, Benevides, Santa Isabel do Pará and Santa Bárbara do Pará, has a total of 154 forest fragments and four urban parks with areas varying from 1 to 1,200 hectares11. Moreover, previous and recent data have been demonstrated that some of these fragments can support enzootic transmission cycles evolving at least four Leishmania species as follows: Leishmania (Leishmania) amazonensis Lainson & Shaw, 1972, L. (V.) lainsoni (Silveira et al., 1987), L. (V.) naiffi (Lainson & Shaw, 1989) and L. (V.) lindenbergi12,13,14,15.
Regarding that fact, from 1940's to 1980's several entomological studies were carried out in forest areas of Belém and surrounding areas, such as Utinga (Belém), Aurá (Ananindeua) and Pirelli (Marituba) providing an extensive knowledge on the distribution of 62 Phlebotominae species, emphasizing the records of incriminated vectors of ACL, Lutzomyia (Nyssomyia) flaviscutellata (Mangabeira, 1942), Lu. (Trichophoromyia) ubiquitalis (Mangabeira, 1942) and the suspected vectors Lu. (N.) antunesi (Coutinho, 1939) and Lu. (Psychodopygus) davisi (Root, 1934). However, owing to the progressive urbanization and environmental changes over the years, these data seem to be outdated16,17.
Due to these previous described facts, this study aimed to investigate the Phlebotominae in seven forest fragments in Belém metropolitan area in order to update their fauna and identify potential vectors of cutaneous leishmaniasis.
MATERIALS AND METHODS
STUDY AREA
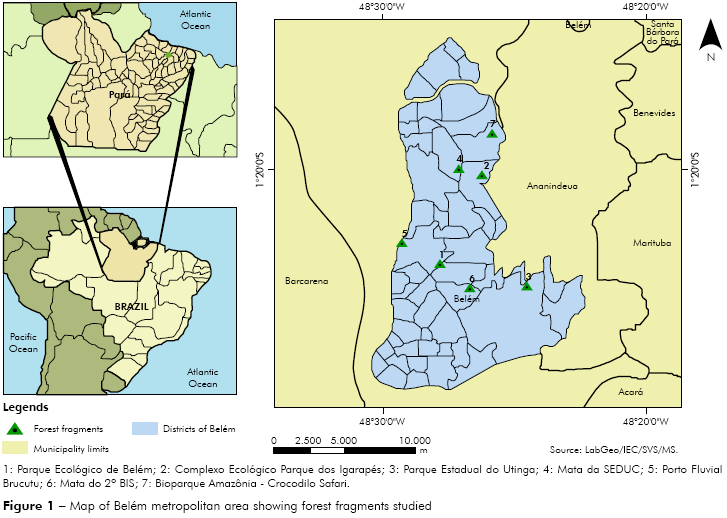
Size, geometry and the distance between vegetation fragments can influence many ecological processes, such as death, migration and colonizations of plants and animals18, so it was selected seven different and ecologically isolated areas (Figure 1): 1- Parque Ecológico do Município de Belém (1o23'47.11"S; 48o27'56.62"W), with 212 hectares, is a unit of environmental conservation under the responsibility of Secretaria Municipal de Meio Ambiente (SEMMA). It is destinated to fauna and flora preservation, and scientific studies and researches, as well as promoting education for integration between man and nature; 2- Complexo Ecológico Parque dos Igarapés (1o20'15.36"S; 48o26'19.14"W) is a private area to provide recreation and ecological tourism in a place of environmental sustainability and preservation of the remaining forest in the urban area; 3- Parque Estadual do Utinga (1o24'39.55"S; 48o24'37.27"W), with 1,200 hectares, is considered the biggest urban park of Belém and with the best ecological conditions due to their size, form and the degree of isolation of forest fragments; 4- Mata da SEDUC (1o19'56.87"S; 48o27'13.38"W), a forest fragment that belongs to Secretaria de Estado de Educação, presenting some depredated areas of rainforest; 5- Porto Fluvial Brucutu (1o22'58.47"S; 48o29'22.16"W), a small forest fragment that serves as air force operational area at Val-de-cães air base; 6- Mata do 2o BIS (1o24'45.15"S; 48o26'47.82"W), a forest operational area of Army's Infantry; 7- Bioparque Amazônia - Crocodilo Safari (1o18'33.58"S; 48o25'52.80"W), a private ecological park with approximately 80 hectares providing contact with four different ecosystems by tracking and ecotourism far away 15 km from the town.
CAPTURING
During the rainy seasons from 2009 to 2011, 11 capture campaigns (three days each) were performed in each area described previously: 2009 (March, April, May and June), 2010 (January, February, March, April and May) and 2011 (January and February). Phlebotomine sand flies captures were undertaken using six battery-operated CDC miniature light traps installed at 1.5 m from ground and 10 m distant from each other in a horizontal transect started on the edge of vegetation, from 6:00 pm to 6:00 am providing a total of 12 h capture per trap-night. The sampling effort was calculated, in hours per trap, multiplying the trap exposure time (12 h) with the total days of campaign (33). The phlebotomine sand flies were triaged, killed in ethyl acetate, washed in a detergent solution (neutral soap 5%), saline solution (NaCl 0.9%) and then stored in alcohol 70% until mounting.
MOUNTING AND IDENTIFICATION
The specimens were mounted in microscope slides individually using berlese's fluid (GBI labs, USA). Damaged specimens could not be identified and therefore they were excluded from the analysis. The identification and nomenclature were carried out in accordance with Young and Duncan4 taxonomic criteria, assisted with a regional pictorial key17. Abbreviations of sand fly names were written with two letters for genus and three for subgenera to avoid confusion to Leishmania abbreviation terms (just one letter for both genus and subgenera). A part of sampled material was deposited in the Phlebotomine Sand fly Collection of the Instituto Evandro Chagas (Coleção de Flebotomíneos do Instituto Evandro Chagas - ColFleb IEC), Ananindeua, Pará, Brazil.
RESULTS
With 360 h/trap of sampling, a total of 1,119 phlebotomine sand fly specimens were collected in the seven studied areas that belong to 22 species/subspecies. All them belonging to the genus Lutzomyia (Lu.) França, 1924 and were distributed in nine subgenera: Psychodopygus (Psy.) Mangabeira, 1941 - four species; Trichophoromyia (Tho.) Barretto, 1962 -three species; Nyssomyia (Nys.) Barretto, 1962 - three species; Evandromyia (Eva.) Mangabeira, 1941 - three species; Viannamyia (Via.) Mangabeira, 1941 - two species; Sciopemyia (Sci.) Barretto, 1962 - two species; Lutzomyia (Lut.) - one species; Trichopygomyia (Thy.) Barretto, 1962 - one species; and Pressatia (Pre.) Mangabeira, 1942 - one species; and group Oswaldoi Theodor, 1965 - two species (Table 1).

The most frequent species were Lu. (Tho.) brachipyga (Mangabeira, 1942) (32.88%), Lu. (Psy.) davisi (Root, 1934) (23.27%), Lu. (Tho.) ubiquitalis (23.05%) and Lu. (Nys.) flaviscutellata (8.14%). A more diverse and abundant fauna was found at Parque Estadual do Utinga (790 specimens, 22 species), followed by Mata da Seduc (250 specimens, five species) and Complexo Ecológico Parque dos Igarapés (37 specimens, five species). Lu. (Nys.) flaviscutellata was the most widely distributed species in the areas, being captured in all the seven capture points sampled and Lu. (Psy.) davisi and Lu. rorotaensis were found in five of them.
DISCUSSION
The Phlebotominae fauna of Belém metropolitan area has been studied since the 1940's16, but few recent data regards the distribution on forest fragments remaining to the progressive vegetal suppression have been reported. As like other recent studies of the sand flies of the Amazon biome, known as having the most diverse fauna19, the present report shows a high diversity of species (22 species) distributed in ten supraspecific taxa (nine subgenera and one species group). However, despite this high diversity, is noteworthy its reduction, since in accordance with Ryan17 the sand fly fauna of Belém consisted of 62 species. On the other side these present results are in accordance with a recent survey carried out in Ananindeua municipality, where a diversity of 23 species in a forest fragment surrounded by urban area was found15.
In fact the present study does not seem to have an extensive sampling effort when compared with the previous ones16,17. Anyway, it is the first time that ecologically isolated areas are analyzed and give us relevant eco-epidemiological information.
The sand flies captured in this study provided an updated picture of the phlebotomine fauna in forested areas of the Belém metropolitan region and also indicates, with previous data, that some species could be potentially involved in the transmission of ACL. Besides 22 species, six of them presented epidemiological importance due to their proven and/or putative implications in the transmission of ACL in Brazilian Amazon as follows: Lu. (Tho.) ubiquitalis, Lu. (Psy.) davisi, Lu. (Nys.) flaviscutellata, Lu. (Nys.) antunesi, Lu. (Psy.) paraensis (Costa Lima, 1941) and Lu. (Psy.) ayrozai (Barretto & Coutinho, 1940). Entomoepidemiological aspects of the most frequent potential vectors discussed below.
Lu. (Psy.) davisi, the second most numerous species (26.27%) and the first one in epidemiological importance is considered as other species of Psychodopygus with a strong anthropophilic tendency20. Moreover, several previous studies about frequency and infection rates have shown that this species should be a potential vector of zoonotic cutaneous leishmaniasis since some specimens have been recorded naturally infected with L. (V.) braziliensis and L. (V.) naiffi in Brazilian Amazon19,21,22,23.
Lu. (Tho.) ubiquitalis was the third numerous species found in this study (23.05%) and the second most epidemiologically important. In previous studies at Belém metropolitan area, in the Municipality of Benevides, this phlebotomine was found to be infected with L. (V.) lainsoni14 and latter incriminated as the main vector in the transmission of this parasite on the basis of its antropophilic behavior under laboratory conditions24.
Despite it was not used a more specific trap (Disney) for catching Lu. (Nys.) flaviscutellata, this species was found in all seven areas studied, with frequencies varying from 2.7% to 62.5%, indicating a irregular distribution in the metropolitan area of Belém. Moreover, it's known that this species is also distributed in other areas of this region: contiguous areas to the Parque Estadual do Utinga, as Mocambo, Utinga, Catu and Água Preta forests25, and others far away as Enviromental Protected Area of Pirelli and Bosque Rodrigues Alves (unpublished data). These data have considered the possibility of Lu. (Nys.) flaviscutellata is being adapted to different environments along over Belém reinforcing its epidemiological importance since this species is so far recognized as the main vector of L. (V.) amazonensis7,12,26.
Lu. (Nys.) antunesi was rarely found (0.71%) and in only two areas sampled (Parque Estadual do Utinga and Complexo Ecológico Parque dos Igarapés). This fact was unexpected, mainly for the absence of specimens in the samples of the Mata do 2o BIS. A entomological survey was done to describe an outbreak of L. (V.) lindenbergi during 1996 in this same area, although no female of Lu. (Nys.) antunesi was found infected for this parasite, it remains in the list of possible vectors, at that moment, the most frequent (50.7%) among the species captured and predominant (83.7%) within the antropophilic ones captured10.
To explain that fact, it would be possible that, once the sampling of this area was undertaken only at rainy season, probably carried out in the beginning of the emergence of adult population. Moreover, recent studies on the seasonal variation of Lu. (Nys.) antunesi in Colombia27 and in Ananindeua Municipality15, near from the present study area, showed that this species had been increased in abundance inversely correlating with the precipitation. Other probable reason is that Lu. antunesi is known to be attracted by Shannon trap10, because of technical conditions it was not possible to be used.
It's necessary to give attention for the most frequent species found, Lu. (Tho.) brachipyga (32.88%). Although most of the species from Trichophoromyia subgenus does not seem to be very anthropophilic and Lu. (Tho.) ubiquitalis is, up to now, the only one incriminated in the transmission of ACL14,24. In Bolivia, Lu. (Tho.) vellascoi Le Pont & Desjeux, 1992 was found infected by an unidentified Leishmanian parasite in a ACL endemic area where a first human case attributed to L. (V.) lainsoni was identified28 and recently in Peru, Lu. (Tho.) auraensis (Mangabeira, 1942) was found infected with L. (V.) lainsoni and L. (V.) braziliensis29. Moreover, two dissected females of Lu. (Tho.) sp. [ambiguous identification of Lu. (Tho.) brachipyga or Lu. (Tho.) adelsonsouzai (Santos, Silva Barata Andrade & Galati, 2014)] were found housing flagellates potentially compatible to peripylarian Leishmania parasites in an area under occurrence of ACL at Brazilian Amazon (TV Santos, unpublished data). All these findings indicate that some Trichophoromyia species could play an important role in the transmission of leishmaniasis agents and their high frequency in entomological surveys, such as Lu. (Tho.) brachipyga in this study may present epidemiological relevancy.
The Parque Estadual do Utinga presented the most richness species (22 species out of 22 total) and abundance of specimens (70.7%) than the other areas analyzed. These data are in accordance with previous studies about this complex of contiguous areas that were indicated as the best ecological conditions to environmental preservation due their size and degree of isolation11, such characteristics also favor to maintain Leishmania life cycles7,8.
CONCLUSION
With these present data, it can be concluded that the phlebotomine sand fly fauna of Belém metropolitan area presents 22 species currently, with at least six beings of epidemiological interest in the potential transmission of five leishmaniasis agents, indicating that continuous investigations on the sampled fragments are need for the entomological surveillance.
ACKNOWLEDGMENTS
To dr. Adelson Alcimar Almeida de Souza (in memoriam) for providing facilities (technical and logistic) for the laboratory work; to the technicians of Instituto Evandro Chagas: Edna de Freitas Leão, Fábio Márcio Medeiros da Silva, Iorlando da Rocha Barata, José Aprígio Nunes Lima, Luciene Aranha da Silva Santos, Maria Sueli Barros Pinheiro and Roberto Carlos Feitosa Brandão for their technical assistance in the laboratory work; to the biologist Paoola Cristina Vieira Amorim and to the technicians of the Laboratório Central da Secretaria do Estado de Saúde Pública (LACEN/SESPA): Dídimo Roberto Pimentel, Antonio Edivaldo Fernandes de Aguiar, Gilberto Gomes Barbosa, João Guilherme Duarte Pereira, Luis Carlos Feitosa Brandão, Marcos César da Silva Faria and Raimundo Brandão Baia, for their assistance in the laboratory and field work; to the technicians of the Museu Paraense Emílio Goeldi: Domingo Dalcides dos Reis Guimarães and Luis Augusto Quaresma, for their general technical assistance; to Clistenes Pamplona Catete (LabGeo/IEC) for his support in the mapping and to the LACEN/SESPA for logistic support that was given to the fieldwork.
FINANCIAL SUPPORT
The authors are also gratiful to the National Council for Scientific and Technological Development (CNPq) for their financial support given to João Victor Silva Ferreira (official announcements PIBIC/MPEG/MCTI n. 001/2009 and n. 001/2010).
REFERENCES
1 Killick-Kendrick R. Phlebotomine vectors of the leishmaniases: a review. Med Vet Entomol. 1990 Jan;4(1):1-24. Doi: 10.1111/j.1365-2915.1990.tb00255.x [Link]
2 Ready PD. Biology of phlebotomine sand flies as vectors of disease agents. Annu Rev Entomol. 2013 Jan;58:227-50. Doi: 10.1146/annurev-ento-120811-153557 [Link]
3 Seccombe AK, Ready PD, Huddleston LM. A catalogue of Old World phlebotomine sandflies (Diptera: Psychodidae, Phlebotominae). Occ Pap Syst Entomol. 1993;8:1-57. [Link]
4 Young DG, Duncan MA. Guide to the identification and geographic distribution of Lutzomyia sand flies in Mexico, the West Indies, Central and South America (Diptera: Psychodidae). Gainesville: Associated Publishers American Entomological Institute; 1994. 881 p. [Link]
5 Lainson R, Rangel EF. Lutzomyia longipalpis and the eco-epidemiology of American visceral leishmaniasis, with particular reference to Brazil: a review. Mem Inst Oswaldo Cruz. 2005 Dec;100(8):811-27. Doi:10.1590/S0074-02762005000800001 [Link]
6 Missawa NA, Veloso MAE, Maciel GBML, Michalsky EM, Dias ES. Evidence of transmission of visceral leishmaniasis by Lutzomyia cruzi in the municipality of Jaciara, state of Mato Grosso, Brazil. Rev Soc Bras Med Trop. 2011 Jan-Feb;44(1):76-8. Doi: 10.1590/S0037-86822011000100017 [Link]
7 Rangel EF, Lainson R. Proven and putative vectors of American cutaneous leishmaniasis in Brazil: aspects of their biology and vectorial competence. Mem Inst Oswaldo Cruz. 2009 Nov;104(7):937-54. Doi: 10.1590/S0074-02762009000700001 [Link]
8 Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Manual de vigilância da leishmaniose tegumentar americana. 2. ed. atual. Brasília: Ministério da Saúde; 2007. 180 p. (Série A. Normas e manuais técnicos).[Link]
9 Silva LMR, Cunha PR. A urbanização da leishmaniose tegumentar americana no município de Campinas - São Paulo (SP) e região: magnitude do problema e desafios. An Bras Dermatol. 2007 nov-dez;82(6):515-9. Doi: 10.1590/S0365-05962007000600003 [Link]
10 Silveira FT, Ishikawa EA, Souza AAA, Lainson R. An outbreak of cutaneous leishmaniasis among soldiers in Belém, Pará state, Brazil, caused by Leishmania (Viannia) lindenbergi n.sp. A new leishmanial parasite of man in the Amazon Region. Parasite. 2002 Mar;9(1):43-50.
11 Ferreira LV, Muñoz SH, Parolin P, Chaves PP. O efeito da fragmentação e isolamento florestal das áreas verdes da região metropolitana de Belém. Pesqui Bot. 2012;63:357-67. [Link]
12 Lainson R, Shaw JJ. Leishmaniasis in Brazil: I. observations on enzootic rodent leishmaniasis-incrimination of Lutzomyia flaviscutellata (mangabeira) as the vector in the lower amazonian basin. Trans R Soc Trop Med Hyg. 1968;62(3):385-95. Doi: 10.1016/0035-9203(68)90090-4 [Link]
13 Silveira FT, Shaw JJ, Braga RR, Ishikawa E. Dermal leishmaniasis in the Amazon region of Brazil: Leishmania (Viannia) lainsoni sp.n., a new parasite from the state of Pará. Mem Inst Oswaldo Cruz. 1987 Apr-Jun;82(2):289-91. Doi: 10.1590/S0074-02761987000200018 [Link]
14 Silveira FT, Souza AAA, Lainson R, Shaw JJ, Braga RR, Ishikawa EA. Cutaneous leishmaniasis in the Amazon region: natural infection of the sandfly Lutzomyia ubiquitalis (Psychodidae: Phlebotominae) by Leishmania (Viannia) lainsoni in Pará state, Brazil. Mem Inst Oswaldo Cruz. 1991 Apr-Jun;86(1):127-30. Doi: 10.1590/S0074-02761987000200018 [Link]
15 Lima JAN, Oliveira DMS, Brandao RCF, Silva FMM, Santos LAS, Pinheiro MSB, et al. Epidemiological evaluation of phlebotomine (Diptera: Psychodidae) fauna in area of coverage in the field of Instituto Evandro Chagas, municipality of Ananindeua, Brazil. In: 28o Annual Meeting of the Brazilian Society of Protozoology, 39o Annual Meeting on Basic Research in Chagas Diesase; 2012 out 1-3; Caxambu: Sociedade Brasileira de Protozoologia; 2012.
16 Damasceno RG, Arouck R, Causey OR. Estudos sobre Phebotomus no Vale Amazônico, parte VI. Contribuição ao conhecimento da distribuição geográfica e da incidência por tipo de captura de 64 espécies identificadas. Rev Serv Espec Saude Publica. 1949 jul;2(3):817-42. [Link]
17 Ryan L. Flebótomos do estado do Pará, Brasil (Diptera: Psychodidae). Belém: Instituto Evandro Chagas; 1986. 154 p. (Documento técnico; No.1). [Link]
18 Laurance WF, Lovejoy TE, Vasconcelos HL, Bruna EM, Didham RK, Stouffer FC, et al. Ecosystem decay of Amazonian Forest fragments: a 22 year investigation. Conserv Biol. 2002 Jun;16(3):605-18. Doi: 10.1046/j.1523-1739.2002.01025.x [Link]
19 Souza AAA, Silveira FT, Lainson R, Barata IR, Silva MGS, Lima JAN, et al. The Phlebotominae fauna of Serra dos Carajás, Pará, Brazil, and its possible implication in the transmission of American tegumentary leishmaniasis. Rev Pan-Amaz Saude. 2010 Mar;1(1):45-51. Doi: 10.5123/S2176-62232010000100007 [Link]
20 Castellón EG, Arias JR, Freitas RA, Naiff RD. Os flebotomíneos da região Amazônica, estrada Manaus Humaitá, estado do Amazonas, Brasil (Diptera: Psychodidae; Phlebotominae). Acta Amaz. 1994 jan-jun;24(1/2):91-102. [Link]
21 Grimaldi Jr G, Momen H, Naiff RD, McMahon-Pratt D, Barrett TV. Characterization and classification of leishmanial parasites from humans, wild mammals, and sand flies in the Amazon region of Brazil. Am J Trop Med Hyg. 1991 Jun;44(6):645-61. [Link]
22 Gil LHS, Basano SA, Souza AAA, Silva MGS, Barata IR, Ishikawa EAY, et al. Recent observations on the sand fly (Diptera: Psychodidae) fauna of the state of Rondônia, Western Amazônia, Brazil: the importance of Psychodopygus davisi as a vector of zoonotic cutaneous leishmaniasis. Mem Inst Oswaldo Cruz. 2003 Sep;98(6):751-5. Doi: 10.1590/S0074-02762003000600007 [Link]
23 Silva FMM, Santos TV, Santos LAS, Leão EF, Brandão RCFS, Silveira FT. Phlebotomine sand fly fauna (Diptera: Psychodidae) and natural flagellate infections in American cutaneous leishmaniasis endemic area under the influence of Belo Monte hydroelectric plant at state of Pará, Amazonian Brazil. In: 5th World Congress on Leishmaniasis - WorldLeish 5. Abstracts; 2013 May 13-17; Porto de Galinhas; 2013. p. 928.
24 Lainson R, Shaw JJ, Souza AAA, Silveira FT, Falqueto A. Further observations on Lutzomyia ubiquitalis (Psychodidae: Phlebotominae), the sandfly vector of Leishmania (Viannia) lainsoni. Mem Inst Oswaldo Cruz. 1992 Jul-Sep;87(3):437-9. Doi: 10.1590/S0074-02761992000300016 [Link]
25 Shaw JJ, Lainson R, Ward RW. Leishmaniasis in Brazil: VII. Further observations on the feeding habitats of Lutzomyia flaviscutellata (Mangabeira) with particular reference to its biting habitats at different heights. Trans R Soc Trop Med Hyg. 1972;66(5):718-23. Doi: 10.1016/0035-9203(72)90085-5 [Link]
26 Ward R, Lainson R, Shaw JJ. Further evidence of the role of Lutzomyia flaviscutellata (Mangabeira) as the vector of Leishmania mexicana amazonensis in Brazil. Trans R Soc Trop Med Hyg. 1973;67(4):608-9. Doi: 10.1016/0035-9203(73)90092-8 [Link]
27 Vásquez Trujillo A, González Reina AE, Góngora Orjuela A, Suárez EP, Palomares JE, Buitrago Alvarez LS. Seasonal variation and natural infection of Lutzomyia antunesi (Diptera: Psychodidae: Phlebotominae), an endemic species in the Orinoquia region of Colombia. Mem Inst Oswaldo Cruz. 2013 Jun;108(4):463-9. Doi:10.1590/S0074-0276108042013011 [Link]
28 Martinez E, Le Ponte F, Mollinedo S, Cupollilo EA. First case of cutaneous leishmaniasis due to Leishmania (Viannia) lainsoni in Bolivia. Trans R Soc Trop Med Hyg. 2001 Jul-Aug:95(4):375-7. Doi: 10.1016/S0035-9203(01)90185-3 [Link]
29 Valdivia HO, Santos MB, Fernandez R, Baldeviano GC, Zorrilla VO, Vera H, et al. Natural Leishmania infection of Lutzomyia (Trichophoromyia) auraensis in Madre de Dios, Peru, detected by a fluorescence resonance energy transfer-based real-time polymerase chain reaction. Am J Trop Med Hyg. 2012 Sep;87(3):511-7. Doi: 10.4269/ajtmh.2012.11-0708 [Link]
 Correspondence / Correspondência / Correspondencia:
Correspondence / Correspondência / Correspondencia:
Thiago Vasconcelos dos Santos
Instituto Evandro Chagas,
Seção de Parasitologia
Rodovia BR 316, km 7, s/no.
Bairro: Levilândia CEP: 67030-000
Ananindeua-Pará-Brasil
Phone #: +55 (91) 3214-2003
E-mail: thiagovasconcelos@iec.pa.gov.br
Received / Recebido em / Recibido en: 11/12/2013
Accepted / Aceito em / Aceito en: 2/5/2014













