
14 relacionados ao sorotipo G2, a eficácia de 75% contra os episódios mais graves, são discutidos à luz do contexto atual dos conhecimentos sobre o tema.]]>
14 related to serotype G2, a 75% efficacy rate against severe episodes, and the non-occurrence of intussusception, are discussed in the context of current knowledge on this issue.]]>
Reanálise da vacina tetravalente (RRV-TV) no contexto da prevenção das gastrenterites por rotavírus
Review of a tetravalent vaccine (RRT-TV) for prevention of gastroenteritis caused by rotavirus
Reevaluación de la vacuna tetravalente (RRV-TV) en el contexto de la prevención de la gastroenteritis por rotavirus
Consuelo Silva de OliveiraI; Alexandre da Costa LinharesI; Nildo Alves BatistaII
]]> IInstituto Evandro Chagas/SVS/MS, Ananindeua, Pará, BrasilEndereço para correspondência
Correspondence
Dirección para correspondencia
RESUMO
Os rotavírus são considerados a principal causa de gastrenterite grave em crianças abaixo de cinco anos, principalmente nos países em desenvolvimento. A vacinação nos primeiros meses de vida se constitui na medida mais efetiva em saúde pública para o controle e prevenção das infecções por tais agentes. Não obstante o recente licenciamento de duas vacinas para uso corrente em lactentes (Rotarix® e RotaTeq®), as pesquisas prosseguem com novas alternativas de prevenção e tratamento. Neste contexto, procedeu-se à reanálise da Rhesus-Human Reassortant Rotavirus Tetravalent Vaccine (RRV-TV), com ênfase à eficácia clínica frente aos parâmetros clínicos, aos sorotipos mais prevalentes na região, à ocorrência de eventos adversos graves (intussuscepção) e a proteção seletiva aos quadros de maior gravidade. Os dados clínicos e epidemiológicos foram obtidos das fichas clínicas de 91 episódios diarreicos em crianças no âmbito de uma investigação prévia conduzida em Belém, Pará. Foram considerados como indicadores de gravidade, os parâmetros clínicos e um sistema de escores, comumente aplicado aos estudos de eficácia da RRV-TV. Os resultados mais expressivos do estudo, como a significativa proteção (p < 0,05) conferida pela RRV-TV em cinco das sete condições clínicas avaliadas, a eficácia cumulativa de 100% contra os episódios com escore clínico >14 relacionados ao sorotipo G2, a eficácia de 75% contra os episódios mais graves, são discutidos à luz do contexto atual dos conhecimentos sobre o tema.
Palavras-chave: Vacinas contra Rotavirus; Gastroenterite; Infecções por Rotavírus.
Rotaviruses are considered the leading cause of severe gastroenteritis in children under five years of age, especially in developing countries. Vaccination in the first months is the most effective public health action for the control and prevention of infections by such agents. Despite the recent licensing of two vaccines for use in infants (Rotarix® and Rota Teq®), researchers continue to seek new alternatives for prevention and treatment. Herein, we provide a review of the Rhesus-Human Reassortant Rotavirus Tetravalent Vaccine (RRV-TV), with an emphasis on its clinical efficacy as regards clinical parameters, the most prevalent serotypes in the region, the occurrence of severe adverse events (e.g., intussusception), and selective protection in the most severe cases. The clinical and epidemiological data were obtained from medical records pertaining to 91 episodes of diarrhea among children in a previous investigation conducted in Belém, Pará State, Brazil. Clinical patterns and a scoring system commonly used in studies on the efficacy of RRV-TV were considered as indicators of severity. The most impressive results of this study, such as a significant protection (p < 0.05) by RRV-TV in five of the seven clinical conditions assessed, the cumulative efficacy rate of 100% against episodes with a clinical score of >14 related to serotype G2, a 75% efficacy rate against severe episodes, and the non-occurrence of intussusception, are discussed in the context of current knowledge on this issue.
Keywords: Rotavirus Vaccines; Gastroenteritis; Rotavirus Infections.
RESUMEN
El rotavirus es la principal causa de gastroenteritis aguda en niños menores de cinco años, especialmente en países en desarrollo. La vacunación en los primeros meses de vida constituye la medida más eficaz en materia de salud pública para el control y la prevención de las infecciones por estos agentes. A pesar de la reciente concesión de licencias para el uso de dos vacunas de rutina en recién nacidos (Rotarix® y Rota Teq®), la investigación continúa con nuevas alternativas para la prevención y tratamiento. En este contexto, se procedió a la revisión de la Rhesus-Human Reassortant Rotavirus Tetravalent Vaccine (RRV-TV), con énfasis en la eficacia clínica frente a los parámetros clínicos en los serotipos que predominan en la región, la ocurrencia de eventos adversos graves (invaginación intestinal) y la protección selectiva de mayor gravedad. Los datos clínicos y epidemiológicos se obtuvieron de los registros médicos de 91 episodios de diarrea en niños, en el ámbito de una investigación anterior realizada en Belém (estado de Pará). Se consideraron indicadores de gravedad los parámetros clínicos y un sistema de puntuación que se aplica comúnmente a los estudios de eficacia de la RRV-TV. Se examinan a la luz de los conocimientos actuales sobre el tema los resultados más llamativos del estudio y la significativa protección (p < 0,05) brindada por la RRV-TV en cinco de las siete condiciones clínicas evaluadas, la eficacia acumulada de 100% para los episodios con la puntuación clínica superior a 14 en relación con el serotipo G2, la eficiencia de 75% contra los episodios graves y la no ocurrencia de invaginación intestinal.
Palabras clave: Vacunas contra Rotavirus; Gastroenteritis; Infecciones por Rotavirus.
INTRODUÇÃO
As gastrenterites agudas por rotavírus constituem um grave problema de saúde pública, principalmente em países em desenvolvimento, onde a doença está associada a aproximadamente a 600 mil óbitos, por ano, de crianças abaixo de 5 anos de idade, com a maior concentração na África e Ásia. Nos Estados Unidos, apesar das baixas taxas de mortalidade (20 a 60 óbitos por ano), tais agentes são responsáveis por 30% a 70% de todas as hospitalizações por gastrenterites1.
]]> Com os estudos conduzidos por Bishop et al3 em Melbourne, Austrália, surgiram as primeiras evidências da diarreia aguda relacionada aos rotavírus. No Brasil, a partir dos achados pioneiros de Linhares et al18 na Região Norte, os estudos epidemiológicos se sucederam em outras regiões do País, confirmando a importância dos rotavírus na morbidade infantil por diarreia, com índices de 30% dos casos hospitalizados e 10% de ocorrência na comunidade22. As recentes investigações epidemiológicas conduzidas no Brasil revelam uma estimativa de 120.513 hospitalizações e 2.475 óbitos associados a estes vírus7.Os rotavírus apresentam uma grande diversidade antigênica, responsável pela multiplicidade de sorotipos, sendo os tipos de G1 a G4 e G9 os de maior prevalência e responsáveis por 95% dos episódios diarreicos em crianças no mundo21. No Brasil, merece destaque uma recente revisão conduzida por Leite et al12 em um número expressivo de amostras oriundas de várias regiões do País (2.691), no período de 1982 a 2007, em que se identificaram os sorotipos G1, G2, G3, G4, G5 e G9, como os de maior circulação no País.
As infecções por rotavírus exibem um padrão sazonal bem definido, particularmente nas regiões de clima temperado, onde se observa uma nítida prevalência dos casos nos meses mais frios. Nas áreas tropicais, a sazonalidade é menos pronunciada, com maior concentração dos casos nos meses mais secos do ano1.
Com base nas observações de que a melhoria das condições de saneamento não interfere nas elevadas taxas de prevalência de diarreia por rotavírus nos países desenvolvidos e naqueles em desenvolvimento, e diante das elevadas taxas de hospitalizações a despeito do uso em larga escala do soro de reidratação oral, a estratégia vacinal é considerada a medida mais efetiva de controle e prevenção das infecções por tais agentes.
A obtenção de uma vacina eficaz e inócua é prioridade no âmbito da Organização Mundial de Saúde (OMS), para ser administrada principalmente nas crianças até os dois primeiros anos de vida, as quais, quando adoecidas, podem evoluir para desidratação e óbito. Os estudos com as candidatas à vacina ideal evoluíram desde os chamados procedimentos Jennerianos - aqueles que utilizam cepas de rotavírus oriundos de animais -, seguido das vacinas de segunda geração, até estratégias que envolvem técnicas de engenharia genética.
Dentre as várias estratégias vacinais estudadas, destaca-se a Rhesus-human Reassortant Rotavirus Tetravalent Vaccine (RRV-TV), primeira vacina contra rotavírus avaliada no Brasil, em meados de 90, em Belém, Pará14. A referida vacina envolve rearranjo genético entre quatro cepas atenuadas de origens símia e humana (4x104 pfu/dose) e foi objeto de vários estudos clínicos, conduzidos em países desenvolvidos11 e na América do Sul (Peru, Brasil e Venezuela). Os resultados indicaram eficácia em relação aos episódios mais graves, alcançando índices comparáveis aos outros estudos com tal imunizante em concentrações mais elevadas10,24. A análise dos resultados alcançados nos vários ensaios clínicos ensejou o licenciamento da primeira vacina contra rotavírus nos EUA (Rotashield®) em julho de 1998, com a recomendação do esquema de três doses, aos 2, 4 e 6 meses de idade5. Entretanto, nove meses depois e com mais de 1 milhão de doses aplicadas em aproximadamente 500 mil crianças, a vacina foi retirada do programa de imunização, decisão fundamentada em ampla avaliação de casos notificados de intussuscepção associada à vacina6. As análises subsequentes dos dados registrados confirmaram esta correlação, porém com o risco estimado entre 1/10.000 a 1/32.000, e com maior ocorrência no terceiro a décimo quarto dia após a primeira dose, em crianças acima dos 3 meses de idade, o que evidenciou nítida associação com a faixa etária20. Vale ressaltar que os ensaios clínicos com a vacina, em curso no mundo, foram descontinuados.
Atualmente se acredita que houve uma superestimativa dos riscos de intussuscepção nos EUA, considerando que 35% das crianças receberam as primeiras doses em idade superior à indicada nos estudos atuais25. Portanto, admite-se que nos países em desenvolvimento os benefícios advindos desta vacina poderiam ser maiores do que o eventual risco de intussuscepção. Neste contexto, justificam-se as análises retrospectivas das experiências acumuladas com a vacina tetravalente, na tentativa de elucidar questões que permanecem obscuras, no esforço global de acelerar a introdução de novas vacinas nos programas de imunização, particularmente de países como a Índia, Indonésia e China, onde um terço dos óbitos são atribuídos aos rotavírus9.
MATERIAIS E MÉTODOS
O presente estudo procedeu a uma reanálise da vacina tetravalente RRV-TV produzida pelo laboratório Wyeth-Ayerst Research (Filadélfia, Pensilvânia, EUA) frente a 91 episódios diarreicos ocorridos em crianças participantes de uma investigação prévia conduzida em Belém, Pará. Tal investigação foi aprovada pelo Comitê de Ética Médica do Instituto Evandro Chagas, pelo Conselho Regional de Medicina do Pará, pela Secretaria Estadual de Saúde do Pará, pelo Ministério da Saúde do Brasil e pela Comissão de Ética da OMS, Genebra, Suiça.
]]> A referida investigação foi desenvolvida na Cidade de Belém, localizada no norte do Brasil, Amazônia Oriental. O estudo básico transcorreu ao longo de dois anos, de caráter prospectivo, randomizado, duplo-cego, em que se administrou vacina ou placebo, na proporção de 1:1, a 540 crianças. O esquema vacinal consistia de três doses, iniciando-se no primeiro mês de vida, seguida de duas outras no terceiro e quinto meses, abrangendo 540 crianças na primeira dose, 513 na segunda, 495 (92%) receberam as três doses do esquema vacinal e, destas, 466 (94%) foram acompanhadas até o final do estudo. As diferenças observadas no quantitativo de crianças estão relacionadas à obediência ao protocolo da pesquisa, como limites pré-definidos de faixa etária para cada uma das três doses preconizadas no esquema vacinal.Para o cálculo do percentual de eficácia da vacina foram considerados apenas os episódios de diarreia por rotavírus registrados depois de decorridas duas semanas após a administração da terceira dose, até completarem-se dois anos de observação.
Os parâmetros clínicos analisados foram obtidos a partir das fichas clínicas utilizadas na vigilância dos episódios diarreicos do referido estudo básico. Nessa investigação, cada criança, transcorrida a primeira semana após a vacinação, era submetida a uma vigilância sistemática dos episódios diarreicos, o que consistia de duas visitas semanais até o final do estudo, com a finalidade de determinar a incidência dos episódios diarreicos. Frente a um caso de diarreia, visitas diárias eram realizadas até o final do episódio. Os critérios clínicos indicativos de gravidade foram: a) diarreia - três ou mais evacuações líquidas ou semilíquidas em um período de 24 h (definição adotada pela OMS para estudos com a vacina contra os rotavírus); b) presença de vômitos; c) febre - temperatura retal ≥ 38o C; d) sinais de desidratação (critérios estabelecidos pela OMS); e) número máximo de evacuações líquidas ou semilíquidas ≥ 6 em 24 h; f) número médio de evacuações líquidas ou semilíquidas ≥ 6 em 24 h; e g) visita a hospitais ou centros de saúde. A par disso, foi utilizado na análise da eficácia um sistema de escore clínico8, comumente aplicado aos estudos de eficácia da RRV-TV e que reúne sinais e sintomas com a pontuação correspondente e somatória máxima de 20 pontos (Tabela 1). Episódios com escores clínicos de 0 a 8, 9 a 14 e acima de 14, foram definidos como leves, moderados a graves e muito graves, respectivamente.

Houve registro de 131 episódios diarreicos por rotavírus no período do estudo, porém somente 91 foram considerados, por traduzirem os episódios que iniciaram após 15 dias da terceira dose. Para a definição diagnóstica desses casos, os espécimes fecais e, em menor número, os swabs retais, foram obtidos logo após a detecção de um episódio diarreico e as amostras submetidas rotineiramente ao método ELISA para detecção de rotavírus (ou seus antígenos), utilizando-se o kit Dakopatts (Copenhague, Dinamarca), recomendado pela OMS.
RESULTADOS
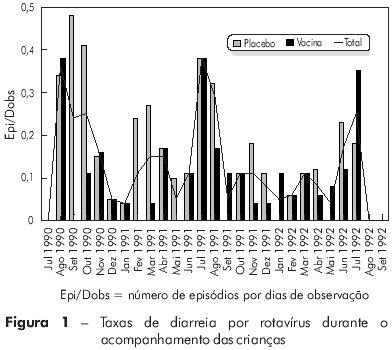
]]> A análise da incidência (número de episódios/dias de observação) dos 91 episódios diarreicos por rotavírus, nos três anos de acompanhamento, revelou índices significativamente superiores nos meses de junho a setembro (p = 0,007), se comparados aos de outros meses no mesmo período de estudo. Esse período corresponde aos meses mais secos do ano (Figura 1).
No tocante à eficácia da RRV-TV frente às condições clínicas relacionadas à gravidade, observou-se efeito protetor em quase todos os parâmetros, exceto frente à febre, em que a vacina não conferiu proteção nos dois anos de estudo (Tabela 2).

]]> A eficácia relativa da vacina em relação ao número máximo de evacuações superior a cinco, nos dois anos de estudo foi significativa frente a todos os episódios: 53% (p = 0,01%). No que se refere à duração dos vômitos, registrou-se total proteção (100%, p = 0,03) no segundo ano de acompanhamento, se considerados os casos de diarreia relacionados ao sorotipo G2.
Durante os dois anos de avaliação, observou-se expressiva eficácia da RRV-TV quanto à desidratação em todos os episódios diarreicos, similar ao observado quanto à necessidade de reidratação. Tais percentuais foram de 48% (p = 0,01) e 46 % (p = 0,01), respectivamente.
A análise da eficácia da RRV-TV frente a todos os episódios diarreicos, de acordo com as faixas de gravidade clínica (Tabela 3) revelou que a vacina não exibiu níveis protetores contra os episódios classificados como de leve intensidade (escore de 0 a 8) e induziu moderada proteção contra todos os casos com escores clínicos de 9 a 14. Com relação aos quadros considerados muito graves (escore > 14) a RRV-TV conferiu níveis expressivos de proteção, 75% (p = 0,02).

A figura 2 exibe a eficácia da vacina frente a todos os casos associados ao sorotipo G2, observando-se que a curva não exibe variações no intervalo entre os escores clínicos de ≥ 2 a ≥ 5 (53%), com ascensão a partir do escore ≥ 6 até ≥ 10 (60% a 77%, respectivamente), decrescendo a seguir a níveis de proteção de 40% (≥ 13). A partir dos escores ≥ 14 até ≥ 16, a curva de eficácia volta a ascender, alcançando níveis máximos de 100% contra os casos revestidos de maior gravidade.
]]>

Quanto à ocorrência de evento adverso grave, não houve registro nos dois anos de acompanhamento.
DISCUSSÃO
Os rotavírus apresentam uma ampla distribuição em todo o mundo. No presente estudo, identificou-se um perfil similar ao descrito para as regiões tropicais, mas de caráter inusitado, se comparado com as observações anteriores que registravam a ocorrência dos episódios por rotavírus durante o todo o ano22. Este perfil, no entanto, pode sofrer modificações, segundo as recentes análises conduzidas pelo CDC, que correlacionam o padrão sazonal das infecções com o uso em larga escala de vacinas, situação atual do Brasil, com a recente introdução da vacina monovalente (Rotarix®) no Programa Nacional de Imunizações (PNI)4.
No que concerne à eficácia frente aos sorotipos prevalentes, os achados atuais demonstram que a proteção conferida pela vacina deve contemplar os sorotipos prevalentes na região. Neste particular, os estudos retrospectivos conduzidos por Linhares et al17 em populações urbanas da nossa região, ao longo da década de 80 até 1992, revelaram a maior prevalência do sorotipo 1, principalmente durante o primeiro ano de vida, seguido do tipo 2. No presente estudo observaram-se variações na curva de eficácia da RRV-TV frente a todos os casos associados ao sorotipo G2, que podem refletir os períodos de maior e menor circulação do sorotipo na região, e ficou demonstrada a expressiva proteção da vacina nos episódios com escores clínicos ≥ 14, definidos como os de maior gravidade. Entretanto, os números absolutos de pequena monta relacionados aos escores mais elevados podem delinear taxas aparentemente mais expressivas.
Ressalte-se que não foi possível, no estudo em análise, mostrar proteção aos sorotipos considerados emergentes (por exemplo: G9), que se constituem num grande desafio no cenário atual das vacinas licenciadas. Diante da ampla variação temporal e geográfica dos sorotipos, a emergência de novos tipos de relevância epidemiológica e a possível cocirculação em uma mesma região, é plenamente justificável o monitoramento sistemático dos sorotipos circulantes, principalmente nos estudos pós licenciamento21.
O risco de intussuscepção (IS) associado à vacina Rotashield tem estimulado ampla discussão e minuciosa revisão dos casos registrados, com os resultados preliminares identificando a idade do início da vacinação como fator de risco para a ocorrência de IS2. A experiência com esta vacina nos EUA seguiu a recomendação da primeira dose aos 2 meses de idade, seguidas de doses aos 4 e 6 meses de idade. As reavaliações subsequentes demonstraram, no entanto, que 61% das crianças iniciaram a vacinação aos 3 meses de idade ou mais e que 80% dos casos de IS associados à vacina, ocorreram nesta faixa etária, período de maior incidência do agravo na população em geral2. Admitindo-se tal associação, justifica-se a recomendação para, nos ensaios clínicos com vacinas, a redução do esquema vacinal para duas doses, com a primeira administrada ainda no período neonatal e o benefício adicional de atenuar outros eventos adversos, como a febre, comumente associada à vacina tetravalente28.
Acumulam-se evidências de que a vacina ideal contra os rotavírus deve assegurar proteção frente aos casos revestidos de maior gravidade, responsáveis pelos elevados índices de morbimortalidade infantil em todo o mundo. No estudo em questão observou-se uma expressiva proteção da vacina em cinco das sete condições clínicas indicadoras de gravidade, tais como: a duração da diarreia, o número máximo de evacuações e de vômitos, a desidratação e a necessidade de reidratação. Vale ressaltar que condições clínicas similares foram identificadas em pesquisas anteriores, ressaltando-se aquelas conduzidas em nossa região por Linhares et al13 em que os vômitos, o número de evacuações líquidas e a desidratação, associados aos episódios diarreicos por rotavírus, exibiram maior gravidade em comparação aos de outras etiologias. Estes índices de proteção foram comparáveis àqueles obtidos em pesquisas conduzidas em outras regiões do mundo com formulações mais concentradas10,24. A análise dos sinais e sintomas em conjunto, à luz de escores clínicos, demonstrou a proteção seletiva da vacina, induzindo proteção aos episódios considerados muito graves (75%, p = 0,02), em consonância com o objetivo principal da vacinação contra os rotavirus15. Tais achados se renovam nos estudos mais recentes que culminaram com o advento das vacinas Rota Teq® e Rotarix®, que estão agora licenciadas19,29.
]]> A eficácia clínica da vacina tetravalente registrada nesta pesquisa foi reproduzida em outros ensaios clínicos conduzidos na América Latina e Finlândia, e confirmada nos dois estudos pós-licenciamento da Rotashield®, que encontraram 100% de proteção nos quadros de maior gravidade, com o esquema de três doses23,26. O potencial protetor da vacina, uma vez introduzida em larga escala, ficou bem evidenciado no estudo conduzido por Tate et al27 nos EUA, com a análise retrospectiva dos dados de uma coorte de crianças vacinadas com a Rotashield®, de 1999 a 2000, com a vacina mostrando-se altamente eficaz na prevenção das hospitalizações e das visitas aos serviços de emergência por gastrenterite de todas as etiologias, ampliando as estimativas prévias do impacto da vacinação em larga escala.Conquanto tratar-se de uma vacina não mais recomendada para uso rotineiro, os achados oriundos com a RRV-TV, estabeleceram as bases para as estratégias futuras e determinantes do atual cenário satisfatório com as vacinas ora licenciadas30.
CONCLUSÃO
Os resultados alcançados confirmam a expressiva proteção da RRV-TV aos episódios diarreicos revestidos de maior gravidade e a eficácia significativa frente aos sorotipos mais prevalentes à época do estudo.
Os achados da presente reanálise somam-se àqueles oriundos dos estudos com a RRV-TV conduzidos na América Latina e nos Estados Unidos e constituíram a base das novas diretrizes para os ensaios clínicos da nova geração de vacinas, no esforço global de acelerar a introdução de vacinas contra os rotavírus nos programas de imunização dos países que apresentam os maiores índices de mortalidade infantil por estes agentes, com destaque para a Índia, Indonésia e China.
AGRADECIMENTO
Agradecimento à sra. Maria José Mateus (Biblioteca do Instituto Evandro Chagas) pelos relevantes serviços no acesso aos artigos científicos.
]]> REFERÊNCIAS
1 Bernstein DI. Rotavirus overview. Pediatr Infect Dis J. 2009 Mar;28(3 Suppl):S50-3. [ Links ]
2 Bines J. Rotavirus vaccines and intussusception risk. Curr Opin Gastroenterol. 2005 Jan;21(1):20-5. [ Links ]
3 Bishop RF, Davidson GP, Holmes IH, Ruck BJ. Virus particles in epithelial cells of duodenal mucosa from children with acute, nonbacterial gastroenteritis. Lancet. 1973 Dec;2(7841):1281-3.
4 Centers for Disease Control and Prevention. Prevention of rotavirus gastroenteritis among infants and children recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2009 Feb;58(2):1-26. [ Links ]
5 Centers for Disease Control and Prevention. Rotavirus vaccine for the prevention of rotavirus gastroenteritis among children: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 1999 Mar;48(2):1-20. [ Links ]
6 Centers for Disease Control and Prevention. Withdrawal of rotavirus vaccine recommendation. MMWR Morb Mortal Wkly Rep. 1999 Nov;48(43):1007. [ Links ]
7 Constenla DO, Linhares AC, Rheingans RD, Antil LR, Waldman EA, Silva LJ. Economic impact of a rotavirus vaccine in Brazil. J Health Popul Nutr. 2008 Dec;26(4):388-96. [ Links ]
8 Flores J, Perez-Schael I, Gonzalez M, Garcia D, Perez M, Daoud N, et al. Protection against severe rotavirus diarrhoea by rhesus rotavirus vaccine in Venezuelan infants. Lancet. 1987 Apr;1(8538):882-4. [ Links ]
9 Glass RI, Bresee JS, Turcios R, Fischer TK, Parashar UD, Steele AD. Rotavirus vaccines: targeting the developing world. J Infect Dis. 2005 Sep;192 Suppl 1:S160-6. DOI: 10.1086/431504 [ Links ]
10 Joensuu J, Koskenniemi E, Pang XL, Vesikari T. Randomised placebo-controlled trial of rhesus-human reassortant rotavirus vaccine for prevention of severe rotavirus gastroenteritis. Lancet. 1997 Oct;350(9086):1205-9. [ Links ]
11 Kapikian AZ, Chanock RM. Rotaviruses. In: Fields BN, Knipe DM, editors. Fields virology. 3rd ed. Philadelphia: Raven Publishers; 1996. Vol. 2, p. 1657-708.
12 Leite JPG, Carvalho-Costa FA, Linhares AC. Group A rotavirus genotypes and the ongoing Brazilian experience - A Review. Mem Inst Oswaldo Cruz. 2008 Dec;103(8):745-53. DOI: 10.1590/S0074-02762008000800001 [ Links ]
13 Linhares AC, Gabbay YB, Freitas RB, Travassos da Rosa ES, Mascarenhas JD, Loureiro EC. Longitudinal study of rotavirus infection among children from Belém, Brazil. Epidemiol Infect. 1989 Feb;102(1):129-45. [ Links ]
14 Linhares AC, Gabbay YB, Mascarenhas JDP, Freitas RB, Oliveira CS, Bellesi N, et al. Immunogenicity, safety and efficacy of tetravalent rhesus-human, reassortant rotavirus vaccine in Belém, Brazil. Bull World Health Organ. 1996;74(5):491-500. [ Links ]
15 Linhares AC, Lanata CF, Hausdorff WP, Gabbay YB, Black RE. Reappraisal of the Peruvian and Brazilian lower-titer tetravalent rhesus-human reassortant rotavirus vaccine efficacy trials: analysis by severity of diarrhea. Pediatr Infect Dis J. 1999 Nov;18(11):1001-6. [ Links ]
16 Linhares AC, Monção HC, Gabbay YB, Araújo VL, Serruya AC, Loureiro ECB. Acute diarrhoea associated with rotavirus among children living in Belém, Brazil. Trans R Soc Trop Med Hyg. 1983;77(3):384-90. [ Links ]
17 Linhares AC, Moura JM, Gabbay YB, Mendes PS, Mascarenhas JD, Azevedo RC. Rotavirus serotypes and electrophoretypes among children attending three pediatric hospitals in Belém, Brazil. J Trop Pediatr. 1993 Jun;39(3):137-41. [ Links ]
18 Linhares AC, Pinheiro FP, Schmetz C, Muller G, Peters D. Duovírus (rotavírus) em Belém do Pará, Brasil (nota prévia). Rev Inst Med Trop Sao Paulo. 1977 jul-ago;19(4):278-9.
19 Linhares AC, Velázquez RR, Pérez-Schael I, Sáez-Llorens X, Abate H, Espinoza F, et al. Efficacy and safety of an oral live attenuated human rotavirus vaccine against rotavirus gastroenteritis during the first 2 years of life in Latin American infants: a randomised, doubleblind, placebo-controlled phase III study. Lancet. 2008 Apr;371(9619):1181-9. [ Links ]
20 Murphy BR, Morens DM, Simonsen L, Chanock RM, La Montagne JR, Kapikian AZ. Reappraisal of the association of intussusception with the licensed live rotavirus vaccine challenges initial conclusions. J Infect Dis. 2003 Apr;187(8):1301-8. [ Links ]
21 O'Ryan M. The ever-changing landscape of rotavirus serotypes. Pediatr Infect Dis J. 2009 Mar;28(3 Suppl):S60-2. [ Links ]
22 Pereira HG, Linhares AC, Candeias JAN, Glass RI. National laboratory surveillance of viral agents of gastroenteritis in Brazil. Bull Pan Am Health Organ. 1993;27(3):224-33. [ Links ]
23 Pérez Mato S, Perrin K, Scardino D, Bégué RE. Evaluation of rotavirus vaccine effectiveness in a pediatric group practice. Am J Epidemiol. 2002 Dec;156(11):1049-55. [ Links ]
24 Pérez-Schael I, Guntiñas MJ, Pérez M, Pagone V, Rojas AM, González R, et al. Efficacy of the rhesus rotavirus-based quadrivalent vaccine in infants and young children in Venezuela. N Engl J Med. 1997 Oct;337(17):1181-7. [ Links ]
25 Simonsen L, Viboud C, Elixhauser A, Taylor RJ, Kapikian AZ. More on RotaShield and intussusception: the role of age at the time of vaccination. J Infect Dis. 2005 Sep;192 Suppl 1:S36-43. [ Links ]
26 Staat MA, Cortese MM, Bresee JS, Bégué RE, Vitek C, Rhodes P, et al. Rhesus rotavirus vaccine effectiveness and factors associated with receipt of vaccine. Pediatr Infect Dis J. 2006 Nov;25(11):1013-8. [ Links ]
27 Tate JE, Curns AT, Cortese MM, Weintraub ES, Hambidge S, Zangwill KM, et al. Burden of acute gastroenteritis hospitalizations and emergency department visits in US children that is potentially preventable by rotavirus vaccination: a probe study using the now-withdrawn RotaShield vaccine. Pediatrics. 2009 Mar;123(3):744-9. [ Links ]
28 Vesikari T, Karvonen A, Forrest BD, Hoshino Y, Chanock RM, Kapikian AZ. Neonatal administration of rhesus rotavirus tetravalent vaccine. Pediatr Infect Dis J. 2006 Feb;25(2):118-22. [ Links ]
29 Vesikari T, Matson DO, Dennehy P, Van Damme P, Santosham M, Rodriguez Z, et al. Safety and efficacy of a pentavalent human-bovine (WC3) reassortant rotavirus vaccine. N Engl J Med. 2006 Jan;354(1):23-33. [ Links ]
30 World Health Organization. Rotavirus vaccines. Wkly Epidemiol Rec. 2007 Aug;82(32):285-95.
 Correspondência/Correspondence/Correspondencia:
Correspondência/Correspondence/Correspondencia:
Consuelo Silva de Oliveira
Instituto Evandro Chagas, Seção de Virologia
Rodovia BR316, km 7, s/no, Levilândia
CEP: 67030-000 Ananindeua-Pará-Brasil
E-mail:consuelooliveira@iec.pa.gov.br
Recebido em/Received/Recibido en: 31/07/2009 ]]> Aceito em/Accepted/Aceito en: 24/09/2009
]]>