Hemocyte production in Biomphalaria glabrata snails after exposure to different Schistosoma mansoni infection protocols
Produção de hemócitos de caramujos da espécie Biomphalaria glabrata após a exposição a diferentes protocolos de infecção por Schistosoma mansoni
Producción de hemocitos de caracoles de la especie Biomphalaria glabrata luego de la exposición a diferentes protocolos de infección por Schistosoma mansoni
Daniel Valle Vasconcelos SantosI; Marco Antonio Vasconcelos SantosII; Izabel Raimunda de Carvalho RodriguesII
]]> ILaboratório de Biofísica Celular, Instituto de Ciências Biológicas, Universidade Federal do Pará, Belém, Pará, BrasilEndereço para correspondência
Correspondence
Dirección para correspondencia
ABSTRACT
The objective of this work was to determine the profile of the cellular defense system during mansonic infection. Specifically, this study assessed the number of hemocytes that were produced and released into the hemolymph in response to the parasitic infection. The quantification of the Biomphalaria glabrata hemocytes was performed on groups of snails at 1, 5, 10, 15, 20 and 30 days post-infection that had been individually infected with 5, 10, 15 or 30 Schistosoma mansoni miracidia. The results revealed that B. glabrata possesses a cellular defense mechanism that is characterized by the release of hemocytes into the hemolymph. The maximum peak of cellular production occurred 24 hours after infection, and there was a significant reduction in the hemocyte concentration over the following 10 days. However, at 15 days post-infection, there was a second increase in the cellular hemocyte production, although this was not as strong as the primary peak. At 30 days post-infection, there was another moderate rise in the cellular hemocyte production. Based on this cellular response profile, the defense system of the snail appears to be effective immediately following infection, but the response does not ensure the destruction of all parasites during the course of the infection.
Keywords: Biomphalaria; Schistosoma mansoni; hemocytes.
O objetivo deste artigo foi determinar o perfil do sistema de defesa celular durante a infecção mansônica. Especificamente, este estudo avaliou o número de hemócitos produzidos e liberados na hemolinfa em resposta à infecção pelo parasita. A quantificação dos hemócitos de Biomphalaria glabrata foi realizada em grupos de caramujos previamente infectados com 5, 10, 15 ou 30 miracídios de Schistosoma mansoni nos dias 1, 5, 10, 15, 20 e 30 pós-infecção. Os resultados revelaram que B. glabrata possui um mecanismo de defesa celular caracterizado pela liberação de hemócitos na hemolinfa. O maior registro de produção celular ocorreu 24 h após a infecção e houve uma redução significante na concentração de hemócitos durante os 10 dias seguintes. No entanto, no dia 15 pós-infecção, houve um segundo aumento na produção de hemócitos, porém não tão acentuado como o primeiro pico. No dia 30 pós-infecção, foi observado outro aumento moderado da produção de hemócitos. Com base neste perfil de resposta celular, o sistema de defesa do caramujo aparenta ser eficiente nos momentos imediatamente posteriores à infecção, mas essa resposta não assegura a destruição de todos os parasitas no curso da infecção.
Palavras-chave: Biomphalaria; Schistosoma mansoni; hemócitos.
RESUMEN
El objetivo de este artículo fue el de determinar el perfil del sistema de defensa celular durante la infección mansónica. Específicamente, este estudio evaluó el número de hemocitos producidos y liberados en la hemolinfa como respuesta a la infección por el parásito. La cuantificación de los hemocitos de Biomphalaria glabrata se realizó en grupos de caracoles previamente infectados con 5, 10, 15 o 30 miracideos de Schistosoma mansoni en los días 1, 5, 10, 15, 20 y 30 pos infección. Los resultados revelaron que B. glabrata posee un mecanismo de defensa celular caracterizado por la liberación de hemocitos en la hemolinfa. El mayor registro de producción celular ocurrió 24 h luego de la infección y hubo una reducción significante en la concentración de hemocitos durante los 10 días siguientes. Sin embargo, al 15o día pos infección, hubo un segundo aumento en la producción de hemocitos, aunque no tan acentuado como el primer pico. Al 30o día pos infección, se verificó otro aumento moderado de la producción de hemocitos. Con base en este perfil de respuesta celular, el sistema de defensa del caracol aparenta ser eficiente en los momentos inmediatamente posteriores a la infección, pero esa respuesta no asegura la destrucción de todos los parásitos en el curso de la infección.
Palabras clave: Biomphalaria; Schistosoma mansoni; hemocitos.
INTRODUCTION
The resistance of Biomphalaria snails to parasitic infection with Schistosoma mansoni is directly related to the hemocyte capacity of the vector to phagocytose and destroy newly penetrated parasites1,2,3,4. The hemocytes are the mollusk's main line of defense against parasites and bacteria5, and these cells are believed to originate from the amoebocyte-producing organ6, although some authors have suggested that these specialized cells may have a multicentric origin7,8.
]]> Miracidium encapsulation occurs immediately after parasite penetration and determines the life or death of the parasite in its host9,10. Basch11 demonstrated that this process can be altered by numerous factors, such as the vector's nutritional state, the virulence of the parasitic strain, the hemocyte quantity in the hemolymph, certain direct or indirect environmental conditions and the number of miracidia that manage to penetrate the host.The objective of the present study was to evaluate the hemocyte production profile of captive Biomphalaria glabrata snails after experimental infection with different numbers of S. mansoni miracidia. The characterization of the variability in hemocyte production is fundamental for understanding the resistance mechanisms of different Biomphalaria species for infection with S. mansoni.
MATERIAL AND METHODS
Brazilian albino (non-pigmented) and pigmented (wild type) strains of B. glabrata differ in their susceptibility to S. mansoni infection12. The B. glabrata specimens used in this study descended from snails captured in the Bragantine region in the state of Pará State, Brazil. This snail colony was kept in captivity and was bred in specialized tanks at the Laboratory of Intestinal Parasitosis and Malacology at the Instituto Evandro Chagas, (Pará State, Brazil). The S. mansoni strain used was extracted from infected mice at this laboratory, but it was initially isolated from infected snails captured at freshwater reservoirs in the City of Belém, Pará State, Brazil.
The livers of the infected mice were macerated, filtrated though gauze and subjected to spontaneous sedimentation. The sediment was exposed to light for 45 min, and the miracidia were collected with micropipettes using a stereomicroscope (Zeiss, Stemi SV 11).
A total of 752 snails, which were approximately 8 months of age and had a shell diameter of 1.5 cm, were individually infected in glass vials filled with 2 mL of declorinated freshwater that contained 5, 10, 15 or 30 miracidia. The infection was considered successful if no miracidia were visualized in the recipient after 90 minutes of light exposure. Following this procedure, the mollusks were transferred to special aquariums that were labeled with their infection protocol. The control group consisted of 52 uninfected snails from the same initial colony.
Hemolymph samples (20µL) were collected by direct puncture from each snail at 1, 5, 10, 15, 20 and 30 days post-infection to evaluate the quantity of the circulating hemocytes. The samples were mixed with Turk's solution (0.1% crystal violet in 1% glacial acetic acid) at a 1:1 ratio, and 5µL of the resulting solution were placed in each side of a Neubauer chamber. As a result, each hemolymph sample was analyzed in duplicate using an optical microscope (Leitz, Dialux 20 EB) at 100x and 400x magnification.
The results are presented as the mean values and the standard deviations. The differences between the values from each group were evaluated using an analysis of variance (ANOVA) and Student's † test (α = 0.05). All statistical analyses were performed using the BioEstat® 5.0 software (IDSM/MCT/CNPq).
]]> RESULTS
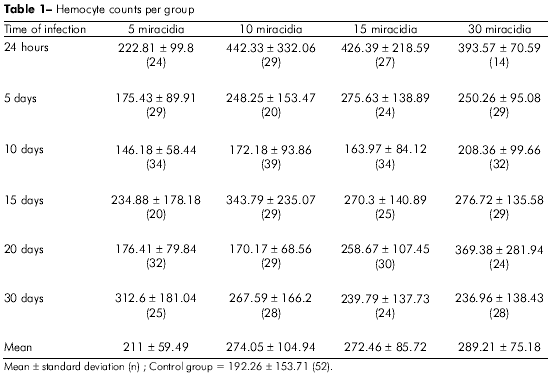
Of the 752 infected snails, 8% died during the course of the infection, and there was no correlation found between the mortality rate and the number of miracidia used for the infection. As a result of this snail death, 710 snails were used for the quantification of circulating hemocytes. These snails were divided into 24 groups of approximately 30 individuals, and the control group consisted of 52 specimens. A summary of these results is presented in table 1.

There was a discrete increase (9.7%) in the circulating hemocyte count in the group that was infected with five miracidia, but these values were not significant when they were compared with the control group. There were, however, very sharp increases in hemocyte production at 24 h post-infection in the groups that were infected with greater numbers of miracidia. The hemocyte production was increased by 130.07%, 121.78% and 104.71% in the groups receiving 10, 15 and 30 miracidia, respectively. After five days of infection, there was a significant reduction in the number of hemocytes in each of the snail groups, although the group that had been infected with five miracidia had a smaller production rate the other experimental groups (p < 0.05).
At 10 days post-infection, there were the fewest numbers of circulating hemocytes in all groups, and the group that had been infected with 30 miracidia experienced the smallest reduction.
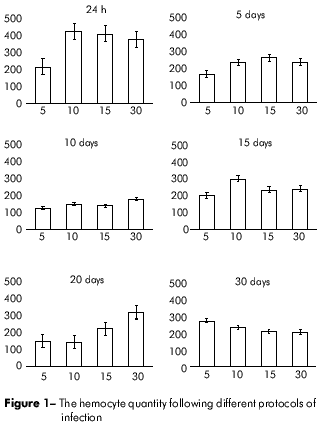
At 15 days post-infection, the circulating hemocyte count increased proportionally to the initial parasitic load, and this trend continued through day 20 post-infection. At day 30, the cellular production stabilized, and there were no significant differences between the experimental groups. These results are shown in figure 1.
]]>
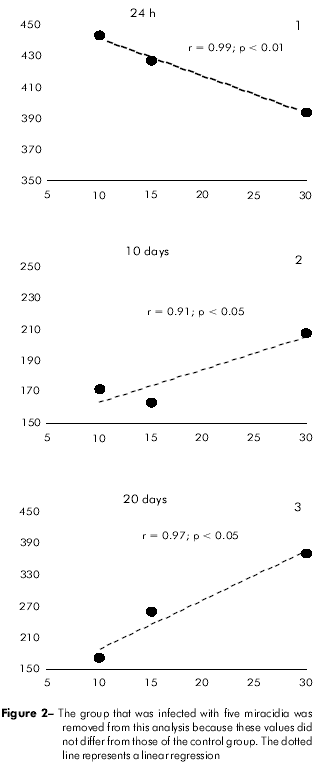
After 24 h of infection, there was a strong inverse correlation between the circulating hemocyte count and the number of miracidia used for the infection (r = 0.99; p < 0.01). This relationship was inverted at day 10 post-infection (r = 0.91; p < 0.05) and was maintained until the last phase of the infection (r = 0.97; p < 0.05) (Figure 2).
DISCUSSION
Hemocytes, which are mobile amoeboid cells, are critical constituents of the Biomphalaria snail's defense against infection with S. mansoni miracidia3,13. These cells form the primary barrier against invading parasites and bacteria13,14; their cytoskeletal mobility and the adaptations of their plasma membranes encapsulate foreign organisms15, and these cells also cooperate with several humoral defense factors. There have been many studies that have attempted to elucidate the complex parasite/host interaction in this model16,17,18,19.
]]> In this paper, we have evaluated variations in the circulating hemocyte counts of individual B. glabrata snails following exposure to diferent numbers of S. mansoni miracidia. The main finding of this study was that there was a significant increase in hemocyte production only when more than five miracidia penetrated the snail. This increase was followed by a dramatic decrease in the hemocyte count at 24 h, which persisted during the first 10 days of infection. At 15 days post-infection, a recovery in the hemocyte cell production was initiated, and the hemocyte level became stable at 30 days post-infection. Moreover, this was independent of the initial parasitic load. There was a strong negative correlation between the hemocyte count and the number of miracidia used for the infection at 24 h post-infection, but this relationship was inverted at 10 and 20 days post-infection.After 24 h of infection, the mean increase in the number of circulating hemocytes was 93.11%. Stumpf and Gilbertson19 reported a twofold increase in the B. glabrata hemocyte count at 2 h after exposure to the miracidia. Similar results were obtained by Joky et al20 and Jeong et al13; there was an increase in the B. glabrata hemocyte count immediately following exposure to a smaller number of miracidia, which was followed by the peak of the cellular production at the third and fourth days and a marked decrease in the hemocyte counts at the sixth and the seventh days. Martins-Souza et al21 characterized the live hemocytes present after schistosomal infection using a cytometric analysis and identified the peak hemocyte production to be 24 h post-infection.
These data support our results in regard to the rise in the hemocyte count during the first hours after miracidia penetration, as well as to the subsequent decline in the circulating hemocyte numbers22,23,24.
This study has also demonstrated that following exposure to S. mansoni, the hemocyte level in the hemolymph was maintained to a greater extent than in the control group and for a longer period of time than that reported by previous papers25. This finding supports the potential existence of a long-term effect on the hemocytes, which most likely consists of humoral factors, after the snail has reacted to the invading organisms.
Our study has also demonstrated that the number of infectious miracidia is a deterministic factor for the magnitude of the cellular response to schistosomal infection. For example, the number of hemocytes produced in the first 24 h after infection was much greater in mollusks infected with 10, 15 or 30 miracidia than in those infected with five of these larvae.
In regard to the dynamics of the snail's hemocytic cellular response to the invading miracidia, previous papers9,10 have reported that the hemocytes interact with or adhere to the parasite's surface 3 h after the penetration of the larvae. Seven and a half hours after this contact, the hemocytes phagocytize the microvilli on the parasite surface. Twenty-four hours later, there is great hemocyte activity, and encapsulation processes and the formation of large phagosomes have been observed. Forty-eight hours later, capsules are found along with large numbers of hemocytes that contain numerous phagosomes. At the fourth day, however, these capsules are much scarcer.
The results obtained by Cheng and Garrabrant12 on two diferent strains of B. glabrata (PR-albino and 10-R2) infected by S. mansoni demonstrated that the hemolymph acid phosphatase levels increased eightfold after 24 h of infection with the 10-R2 strain when compared to the non-infected control group, but that there was only a discrete increase in these levels at the same time period for the group infected with the PR-albino strain. In regard to the hemocyte numbers, the results were completely reversed. The PR-albino strain induced a peak of hemocyte production during the first 12 h of the infection, which was immediately followed by a marked reduction in the circulating hemocyte numbers, while the 10-R2 strain did not significantly alter the hemocyte count during the infectious process, which indicated that acid phosphatases may have an important role in the regulation of hemocyte production following parasite exposure.
CONCLUSION
The dynamics of the phagocytosis process, which were observed through the fourth day post-infection, suggest that the mobilization of these defensive cells is only effective during the first phase of the infectious process after a large number of parasites have penetrated the snail. Moreover, this mobilization does not guarantee the elimination of the larvae during the course of the infection. The mollusk's defense response against the parasitic larvae could be more effective if it were not only regulated by a cellular process, but also by biochemical mechanisms that would directly stimulate the hematopoietic organs to produce more hemocytes.
]]>ACKNOWLEDGEMENTS
We would like to thank Rosângela Barros do Nascimento, Carlos da Silva Faria and Bruno Silva de Lima for technical support throughout the course of our research. We also thank Kauê Machado Costa for critically reviewing the manuscript and his thoughtful suggestions.
FINANCIAL SUPPORT
Instituto Evandro Chagas / Fundação Nacional de Saúde.
REFERENCES
1 Newton W. The comparative tissue reaction of two strains of Australorbis glabratus to infection with Schistosomia mansoni. J Parasitol. 1952 Aug;38(4 Sec. A. 1):362-6.
2 Barbosa F, Coelho M, Dobbin J. Qualidade de vetor dos hospedeiros de Schistosoma mansoni no Nordeste do Brasil. II. Duração da infestação e eliminação de cercarias em Australorbis glabratus. Publ Av Inst Aggeu Magalhães. 1954;7:79-92.
3 Lie J, Heyneman D, Jeong K. Studies on resistance in snails. 4. Induction of ventricular capsules and changes in the amebocyte-producing organ during sensitization of Biomphalaria glabrata snails. J Parasitol. 1976 Apr;62(2):286-91. [Link]
4 Negrão-Corrêa D, Pereira C, Rosa F, Martins R, Martins-Souza RL, Andrade ZA, et al. Molluscan response to parasite: Biomphalaria and Schistosoma mansoni interaction. ISJ. 2007;4:101-11. [Link]
5 Granath Jr WO, Yoshino TP. Lysosomal enzyme activities in susceptible and refractory strains of Biomphalaria glabrata during the course of infection with Schistosoma mansoni. J Parasitol. 1983 Dec;69(6):1018-26. [Link]
6 Cheng TC, Auld KR. Hemocytes of the pulmonate gastropod Biomphalaria glabrata. J Invertebr Pathol. 1977 Jul;30(1):119-22.
7 Connors VA, Buron I, Granath Jr WO. Schistosoma mansoni: interleukin-1 increases phagocytosis and superoxide production by hemocytes and decreases output of cercariae in schistosome-susceptible Biomphalaria glabrata. Exp Parasitol. 1995 Feb;80:139-48. [Link]
8 Souza S, Andrade Z. On the origin of the Biomphalaria glabrata hemocytes. Mem Inst Oswaldo Cruz. 2006 Sep;101 Suppl 1:S213-8. [Link]
9 Loker ES, Bayne CJ, Buckley PM, Kruse KT. Ultrastructure of encapsulation of Schistosoma mansoni mother sporocysts by hemocytes of juveniles of the 10-R2 strain of Biomphalaria glabrata. J Parasitol. 1982 Feb;68(1):84-94.
10 Van der Knaap WP, Loker E. Immune mechanisms in trematod-snail interactions. Parasitol Today. 1990 Jun;6(6):175-82. Doi:10.1016/0169-4758(90)90349-9 [Link]
11 Basch PF. Intermediate host specificity in Schistosoma mansoni. Exp Parasitol. 1976 Feb;39(1):150-69.
12 Cheng T, Garrabrant T. Acid phosphatase in granulocytic capsules formed in strains of Biomphalaria glabrata totally and partially resistant to Schistosoma mansoni. Int J Parasitol. 1977 Dec;7(6):467-72.
13 Jeong KH, Lie KJ, Heyneman D. Leucocytosis in Biomphalaria glabrata sensitized and resensitized to Echinostoma lindoense. J Invertebr Pathol. 1980 Jan;35(1):9-13.
14 Matricon-Gondran M, Letocart M. Internal defenses of the snail Biomphalaria glabrata II. Defense cells have different phagocytic responses to various injected foreign materials. J Invertebr Pathol. 1999 Nov;74(3):235-47. Doi:10.1006/Jipa.1999.4877 [Link]
15 Loker ES, Bayne CJ, Yui MA. Echinostoma paraensei: hemocytes of Biomphalaria glabrata as targets of echinostome mediated interference with host snail resistance to Schistosoma mansoni. Exp Parasitol. 1986 Aug;62(1):149-54. [Link]
16 Barracco MA, Steil AA, Gargioni R. Morphological characterization of the hemocytes of the pulmonate snail Biomphalaria tenagophila. Mem Inst Oswaldo Cruz. 1993 Jan-Mar;88(1):73-83. http://dx.doi.org/10.1590/S0074-02761993000100012 [Link]
17 Bayne CJ, Buckley PM, DeWan PC. Schistosoma mansoni: cytotoxicity of hemocytes from susceptible snail hosts for sporocysts in plasma from resistant Biomphalaria glabrata. Exp Parasitol. 1980 Dec;50(3):409-16.
18 Bayne CJ, Loker ES, Yui MA, Stephens J. Immune-recognition of Schistosoma mansoni primary sporocysts may require specific receptors on Biomphalaria glabrata hemocytes. Parasite Immunol. 1984 Dec;6(6):519-28. DOI:10.1111/j.1365-3024.1984.tb00822.x [Link]
19 Stumpf JL, Gilbertson DE. Hemocytes of Biomphalaria glabrata: factors affecting variability. J Invertebr Pathol. 1978 Sep;32(2):177-81. Doi:10.1016/0022-2011(78)90027-7 [Link]
20 Joky A, Matricon-Gondran M, Benex J. Response to the amoebocyte-producing organ of sensitized Biomphalaria glabrata after exposure to Echinostoma caproni miracidia. J Invertebr Pathol. 1985 Jan;45(1):28-33. Doi:10.1016/0022-2011(85)90045-X [Link]
21 Martins-Souza RL, Pereira CA, Coelho PM, Martins-Filho O, Negrão-Corrêa D. Flow cytometry analysis of the circulating haemocytes from Biomphalaria glabrata and Biomphalaria tenagophila following Schistosoma mansoni infection. Parasitology. 2009 Jan;136(1):67-76. DOI:10.1017/S0031182008005155 [Link]
22 Matricon-Gondran M. The site of ultrafiltration in the kidney sac of the pulmonate gastropod Biomphalaria glabrata. Tissue & cell. 1990;22:911-23. Doi:10.1016/0040-8166(90)90052-B [Link]
23 Bayne CJ, Hahn UK, Bender RC. Mechanisms of molluscan host resistance and of parasite strategies for survival. Parasitology. 2001;123 Suppl:S159-67. [Link]
24 Martins-Souza RL, Pereira CA, Coelho PM, Negrão-Corrêa D. Silica treatment increases the susceptibility of the Cabo Frio strain of Biomphalaria tenagophila to Schistosoma mansoni infection but does not alter the natural resistance of the Taim strain. Parasitol Res. 2003 Dec;91(6):500-7. DOI: 10.1007/s00436-003-0985-4 [Link]
25 Oliveira AL, Silva D, Manzano BC, Abdel-Hamid AZ, Marcelino MY, Zanotti-Magalhaes EM, et al. Genetic differences between strains of Biomphalaria glabrata (Planorbidae) that are susceptible and unsusceptible to schistosomiasis. Genet Mol Res. 2010 Jul;9(3):1450-9. DOI 10.4238/vol9-3gmr821 [Link]
 Correspondence / Correspondência / Correspondencia:
Correspondence / Correspondência / Correspondencia:
Daniel Valle Vasconcelos Santos
Rua Arciprestes Manoel Teodoro, no 265 Bairro: Batista Campos
CEP: 66023700 Belém - Pará – Brasil
Tel.: 55 (91) 81299664 ]]>
E-mail: danielsantos@ufpa.br
Received / Recebido em / Recibido en: 27/5/2011
Accepted / Aceito em / Aceito en: 1/9/2011